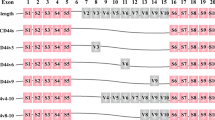
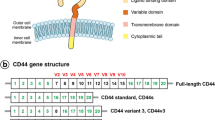
Abstract
The CD44 protein family spans a large group of transmembrane glycoproteins acquired by alternative splicing and post-translational modifications. The great heterogeneity in molecular structure is reflected in its various important functions: CD44 mediates (1) interaction between cell and extracellular matrix, (2) signal submission, e.g., by acting as co-receptor for membrane-spanning receptor tyrosine kinases or by association with intracellular molecules initiating several signaling pathways, and (3) anchor function connecting to the cytoskeleton via the ezrin-radixin-moesin protein family. The expression pattern of the different CD44 isoforms display strong variations dependent on cell type, state of activation, and differentiation stage. In hematopoietic cells, CD44 mediates interaction of progenitor cells and bone marrow stroma during hematopoiesis, regulates maturation, and activation-induced cell death in T cells, influences neutrophil and macrophage migration as well as cytokine production, and participates in lymphocyte extravasation and migration. CD44 is involved in development and progress of hematological neoplasias by enhancement of apoptotic resistance, invasiveness, as well as regulation of bone marrow homing, and mobilization of leukemia-initiating cells into the peripheral blood. Thereby altered CD44 expression functions as marker for worse prognosis in most hematological malignancies. Additionally, CD44 expression levels can be used to distinguish between different hematological neoplasias and subtypes. Concerning new treatment strategies, CD44 displays promising potential either by direct targeting of CD44 expressed on the malignant cells or reversing an acquired resistance to primary treatment mediated through altered CD44 expression. The former can be achieved by antibody or hyaluronan-based immunotherapy.


Similar content being viewed by others
References
Dalchau R, Kirkley J, Fabre JW (1980) Monoclonal antibody to a human leukocyte-specific membrane glycoprotein probably homologous to the leukocyte-common (L-C) antigen of the rat. Eur J Immunol 10(10):737–744
Ponta H, Wainwright D, Herrlich P (1998) The CD44 protein family. Int J Biochem Cell Biol 30(3):299–305
Screaton GR, Bell MV, Jackson DG, Cornelis FB, Gerth U, Bell JI (1992) Genomic structure of DNA encoding the lymphocyte homing receptor CD44 reveals at least 12 alternatively spliced exons. Proc Natl Acad Sci U S A 89(24):12160–12164
Günthert U (1993) CD44: a multitude of isoforms with diverse functions. Curr Top Microbiol Immunol 184:47–63
Naor D, Wallach-Dayan SB, Zahalka MA, Sionov RV (2008) Involvement of CD44, a molecule with a thousand faces, in cancer dissemination. Semin Cancer Biol 18(4):260–267
Fichter M, Hinrichs R, Eissner G, Scheffer B, Classen S, Ueffing M (1997) Expression of CD44 isoforms in neuroblastoma cells is regulated by PI 3-kinase and protein kinase C. Oncogene 14(23):2817–2824
Fitzgerald KA, O'Neill LA (1999) Characterization of CD44 induction by IL-1: a critical role for Egr-1. J Immunol 162(8):4920–4927
König H, Ponta H, Herrlich P (1998) Coupling of signal transduction to alternative pre-mRNA splicing by a composite splice regulator. EMBO J 17(10):2904–2913
Khan SA, Cook AC, Kappil M, Günthert U, Chambers AF, Tuck AB, Denhardt DT (2005) Enhanced cell surface CD44 variant (v6, v9) expression by osteopontin in breast cancer epithelial cells facilitates tumor cell migration: novel post-transcriptional, post-translational regulation. Clin Exp Metastasis 22(8):663–673
Zhang T, Huang X, Dong L, Hu D, Ge C, Zhan Y, Xu W, Yu M, Li W et al (2010) PCBP-1 regulates alternative splicing of the CD44 gene and inhibits invasion in human hepatoma cell line HepG2 cells. Mol Cancer 9:72
Yan C, Wu W, Li H, Zhang G, Duerksen-Hughes PJ, Zhu X, Yang J (2010) Benzo[a]pyrene treatment leads to changes in nuclear protein expression and alternative splicing. Mutat Res 686(1–2):47–56
Naor D, Sionov RV, Ish-Shalom D (1997) CD44: structure, function, and association with the malignant process. Adv Cancer Res 71:241–319
Ponta H, Sherman L, Herrlich PA (2003) CD44: from adhesion molecules to signalling regulators. Nat Rev Mol Cell Biol 4(1):33–45
Mori T, Kitano K, Terawaki S, Maesaki R, Fukami Y, Hakoshima T (2008) Structural basis for CD44 recognition by ERM proteins. J Biol Chem 283(43):29602–29612
Kajita M, Itoh Y, Chiba T, Mori H, Okada A, Kinoh H, Seiki M (2001) Membrane-type 1 matrix metalloproteinase cleaves CD44 and promotes cell migration. J Cell Biol 153(5):893–904
Cichy J, Puré E (2004) Cytokines regulate the affinity of soluble CD44 for hyaluronan. FEBS Lett 556(1–3):69–74
Murai T, Miyauchi T, Yanagida T, Sako Y (2006) Epidermal growth factor-regulated activation of Rac GTPase enhances CD44 cleavage by metalloproteinase disintegrin ADAM10. Biochem J 395(1):65–71
Mackay CR, Terpe HJ, Stauder R, Marston WL, Stark H, Günthert U (1994) Expression and modulation of CD44 variant isoforms in humans. J Cell Biol 124(1–2):71–82
Legras S, Günthert U, Stauder R, Curt F, Oliferenko S, Kluin-Nelemans HC, Marie JP, Proctor S, Jasmin C et al (1998) A strong expression of CD44-6v correlates with shorter survival of patients with acute myeloid leukemia. Blood 91(9):3401–3413
Günthert U, Hofmann M, Rudy W, Reber S, Zöller M, Haussmann I, Matzku S, Wenzel A, Ponta H et al (1991) A new variant of glycoprotein CD44 confers metastatic potential to rat carcinoma cells. Cell 65(1):13–24
Heider K, Kuthan H, Stehle G, Munzert G (2004) CD44v6: a target for antibody-based cancer therapy. Cancer Immunol Immunother 53(7):567–579
Kai K, Arima Y, Kamiya T, Saya H (2010) Breast cancer stem cells. Breast Cancer 17(2):80–85
Todaro M, Francipane MG, Medema JP, Stassi G (2010) Colon cancer stem cells: promise of targeted therapy. Gastroenterology 138(6):2151–2162
Lee CJ, Dosch J, Simeone DM (2008) Pancreatic cancer stem cells. J Clin Oncol 26(17):2806–2812
Zou G (2010) Liver cancer stem cells as an important target in liver cancer therapies. Anticancer Agents Med Chem 10(2):172–175
Perschl A, Lesley J, English N, Trowbridge I, Hyman R (1995) Role of CD44 cytoplasmic domain in hyaluronan binding. Eur J Immunol 25(2):495–501
Cichy J, Pure E (2000) Oncostatin M and transforming growth factor-beta 1 induce post-translational modification and hyaluronan binding to CD44 in lung-derived epithelial tumor cells. J Biol Chem 275(24):18061–18069
Katoh S, Zheng Z, Oritani K, Shimozato T, Kincade PW (1995) Glycosylation of CD44 negatively regulates its recognition of hyaluronan. J Exp Med 182(2):419–429
Puré E, Assoian RK (2009) Rheostatic signaling by CD44 and hyaluronan. Cell Signal 21(5):651–655
Fujiwara T, Kawakatsu T, Tayama S, Kobayashi Y, Sugiura N, Kimata K, Takai Y (2008) Hyaluronan-CD44 pathway regulates orientation of mitotic spindle in normal epithelial cells. Genes Cells 13(7):759–770
Legras S, Levesque JP, Charrad R, Morimoto K, Le Bousse C, Clay D, Jasmin C, Smadja-Joffe F (1997) CD44-mediated adhesiveness of human hematopoietic progenitors to hyaluronan is modulated by cytokines. Blood 89(6):1905–1914
Levesque MC, Haynes BF (1999) TNFalpha and IL-4 regulation of hyaluronan binding to monocyte CD44 involves posttranslational modification of CD44. Cell Immunol 193(2):209–218
Brown KL, Maiti A, Johnson P (2001) Role of sulfation in CD44-mediated hyaluronan binding induced by inflammatory mediators in human CD14(+) peripheral blood monocytes. J Immunol 167(9):5367–5374
DeGrendele HC, Kosfiszer M, Estess P, Siegelman MH (1997) CD44 activation and associated primary adhesion is inducible via T cell receptor stimulation. J Immunol 159(6):2549–2553
Verfaillie CM, Benis A, Iida J, McGlave PB, McCarthy JB (1994) Adhesion of committed human hematopoietic progenitors to synthetic peptides from the C-terminal heparin-binding domain of fibronectin: cooperation between the integrin alpha 4 beta 1 and the CD44 adhesion receptor. Blood 84(6):1802–1811
Nandi A, Estess P, Siegelman M (2004) Bimolecular complex between rolling and firm adhesion receptors required for cell arrest; CD44 association with VLA-4 in T cell extravasation. Immunity 20(4):455–465
Lee J, Wang M, Sudhir P, Chen G, Chi C, Chen J (2007) Osteopontin promotes integrin activation through outside-in and inside-out mechanisms: OPN–CD44V interaction enhances survival in gastrointestinal cancer cells. Cancer Res 67(5):2089–2097
Wang H, Hung Y, Su C, Peng S, Guo Y, Lai M, Liu C, Hsu J (2005) CD44 cross-linking induces integrin-mediated adhesion and transendothelial migration in breast cancer cell line by up-regulation of LFA-1 (alpha L beta2) and VLA-4 (alpha4beta1). Exp Cell Res 304(1):116–126
Torre C, Wang SJ, Xia W, Bourguignon LYW (2010) Reduction of hyaluronan-CD44-mediated growth, migration, and cisplatin resistance in head and neck cancer due to inhibition of Rho kinase and PI-3 kinase signaling. Arch Otolaryngol Head Neck Surg 136(5):493–501
Baronas-Lowell D, Lauer-Fields JL, Borgia JA, Sferrazza GF, Al-Ghoul M, Minond D, Fields GB (2004) Differential modulation of human melanoma cell metalloproteinase expression by alpha2beta1 integrin and CD44 triple-helical ligands derived from type IV collagen. J Biol Chem 279(42):43503–43513
Yu Q, Stamenkovic I (1999) Localization of matrix metalloproteinase 9 to the cell surface provides a mechanism for CD44-mediated tumor invasion. Genes Dev 13(1):35–48
Lefebvre DC, Lai JCY, Maeshima N, Ford JL, Wong ASL, Cross JL, Johnson P (2010) CD44 interacts directly with Lck in a zinc-dependent manner. Mol Immunol 47(10):1882–1889
Bourguignon LY, Zhu H, Shao L, Chen YW (2001) CD44 interaction with c-Src kinase promotes cortactin-mediated cytoskeleton function and hyaluronic acid-dependent ovarian tumor cell migration. J Biol Chem 276(10):7327–7336
Ilangumaran S, Arni S, van Echten-Deckert G, Borisch B, Hoessli DC (1999) Microdomain-dependent regulation of Lck and Fyn protein-tyrosine kinases in T lymphocyte plasma membranes. Mol Biol Cell 10(4):891–905
McClatchey AI (2003) Merlin and ERM proteins: unappreciated roles in cancer development? Nat Rev Cancer 3(11):877–883
Terawaki S, Kitano K, Mori T, Zhai Y, Higuchi Y, Itoh N, Watanabe T, Kaibuchi K, Hakoshima T (2010) The PHCCEx domain of Tiam1/2 is a novel protein- and membrane-binding module. EMBO J 29(1):236–250
Wang KX, Denhardt DT (2008) Osteopontin: role in immune regulation and stress responses. Cytokine Growth Factor Rev 19(5–6):333–345
Tanikawa R, Tanikawa T, Hirashima M, Yamauchi A, Tanaka Y (2010) Galectin-9 induces osteoblast differentiation through the CD44/Smad signaling pathway. Biochem Biophys Res Commun 394(2):317–322
Peterson RS, Andhare RA, Rousche KT, Knudson W, Wang W, Grossfield JB, Thomas RO, Hollingsworth RE, Knudson CB (2004) CD44 modulates Smad1 activation in the BMP-7 signaling pathway. J Cell Biol 166(7):1081–1091
Bourguignon LYW, Wong G, Earle C, Krueger K, Spevak CC (2010) Hyaluronan-CD44 interaction promotes c-Src-mediated twist signaling, MicroRNA-10b expression and RhoA/RhoC upregulation leading to Rho-kinase-associated cytoskeleton activation and breast tumor cell invasion. J Biol Chem 285(47):36721–36735
Bretscher A, Edwards K, Fehon RG (2002) ERM proteins and merlin: integrators at the cell cortex. Nat Rev Mol Cell Biol 3(8):586–599
Turley EA, Noble PW, Bourguignon LYW (2002) Signaling properties of hyaluronan receptors. J Biol Chem 277(7):4589–4592
Yu Q, Stamenkovic I (2004) Transforming growth factor-beta facilitates breast carcinoma metastasis by promoting tumor cell survival. Clin Exp Metastasis 21(3):235–242
Tian Y, Phillips AO (2003) TGF-beta1-mediated inhibition of HK-2 cell migration. J Am Soc Nephrol 14(3):631–640
Oliferenko S, Kaverina I, Small JV, Huber LA (2000) Hyaluronic acid (HA) binding to CD44 activates Rac1 and induces lamellipodia outgrowth. J Cell Biol 148(6):1159–1164
Subramaniam V, Vincent IR, Gardner H, Chan E, Dhamko H, Jothy S (2007) CD44 regulates cell migration in human colon cancer cells via Lyn kinase and AKT phosphorylation. Exp Mol Pathol 83(2):207–215
Shi X, Leng L, Wang T, Wang W, Du X, Li J, McDonald C, Chen Z, Murphy JW et al (2006) CD44 is the signaling component of the macrophage migration inhibitory factor-CD74 receptor complex. Immunity 25(4):595–606
Mielgo A, Brondani V, Landmann L, Glaser-Ruhm A, Erb P, Stupack D, Günthert U (2007) The CD44 standard/ezrin complex regulates Fas-mediated apoptosis in Jurkat cells. Apoptosis 12(11):2051–2061
Okamoto I, Kawano Y, Murakami D, Sasayama T, Araki N, Miki T, Wong AJ, Saya H (2001) Proteolytic release of CD44 intracellular domain and its role in the CD44 signaling pathway. J Cell Biol 155(5):755–762
Kim Y, Lee Y, Choe J, Lee H, Kim Y, Jeoung D (2008) CD44-epidermal growth factor receptor interaction mediates hyaluronic acid-promoted cell motility by activating protein kinase C signaling involving Akt, Rac1, Phox, reactive oxygen species, focal adhesion kinase, and MMP-2. J Biol Chem 283(33):22513–22528
Puré E, Cuff CA (2001) A crucial role for CD44 in inflammation. Trends Mol Med 7(5):213–221
Lapidot T, Dar A, Kollet O (2005) How do stem cells find their way home? Blood 106(6):1901–1910
Ohata S, Nawa M, Kasama T, Yamasaki T, Sawanobori K, Hata S, Nakamura T, Asaoka Y, Watanabe T et al (2009) Hematopoiesis-dependent expression of CD44 in murine hepatic progenitor cells. Biochem Biophys Res Commun 379(4):817–823
Chen K, Liu J, Heck S, Chasis JA, An X, Mohandas N (2009) Resolving the distinct stages in erythroid differentiation based on dynamic changes in membrane protein expression during erythropoiesis. Proc Natl Acad Sci U S A 106(41):17413–17418
Vaskova M, Fronkova E, Starkova J, Kalina T, Mejstrikova E, Hrusak O (2008) CD44 and CD27 delineate B-precursor stages with different recombination status and with an uneven distribution in nonmalignant and malignant hematopoiesis. Tissue Antigens 71(1):57–66
Rajasagi M, Vitacolonna M, Benjak B, Marhaba R, Zöller M (2009) CD44 promotes progenitor homing into the thymus and T cell maturation. J Leukoc Biol 85(2):251–261
Baaten BJG, Li C, Deiro MF, Lin MM, Linton PJ, Bradley LM (2010) CD44 regulates survival and memory development in Th1 cells. Immunity 32(1):104–115
Nakano K, Saito K, Mine S, Matsushita S, Tanaka Y (2007) Engagement of CD44 up-regulates Fas ligand expression on T cells leading to activation-induced cell death. Apoptosis 12(1):45–54
Ruffell B, Johnson P (2008) Hyaluronan induces cell death in activated T cells through CD44. J Immunol 181(10):7044–7054
Ogino S, Nishida N, Umemoto R, Suzuki M, Takeda M, Terasawa H, Kitayama J, Matsumoto M, Hayasaka H et al (2010) Two-state conformations in the hyaluronan-binding domain regulate CD44 adhesiveness under flow condition. Structure 18(5):649–656
Napier SL, Healy ZR, Schnaar RL, Konstantopoulos K (2007) Selectin ligand expression regulates the initial vascular interactions of colon carcinoma cells: the roles of CD44v and alternative sialofucosylated selectin ligands. J Biol Chem 282(6):3433–3441
Zhu B, Suzuki K, Goldberg HA, Rittling SR, Denhardt DT, McCulloch CAG, Sodek J (2004) Osteopontin modulates CD44-dependent chemotaxis of peritoneal macrophages through G-protein-coupled receptors: evidence of a role for an intracellular form of osteopontin. J Cell Physiol 198(1):155–167
Ashkar S, Weber GF, Panoutsakopoulou V, Sanchirico ME, Jansson M, Zawaideh S, Rittling SR, Denhardt DT, Glimcher MJ et al (2000) Eta-1 (osteopontin): an early component of type-1 (cell-mediated) immunity. Science 287(5454):860–864
Muto J, Yamasaki K, Taylor KR, Gallo RL (2009) Engagement of CD44 by hyaluronan suppresses TLR4 signaling and the septic response to LPS. Mol Immunol 47(2–3):449–456
Hoffmann U, Heilmann K, Hayford C, Stallmach A, Wahnschaffe U, Zeitz M, Günthert U, Wittig BM (2007) CD44v7 ligation downregulates the inflammatory immune response in Crohn's disease patients by apoptosis induction in mononuclear cells from the lamina propria. Cell Death Differ 14(8):1542–1551
McKee CM, Penno MB, Cowman M, Burdick MD, Strieter RM, Bao C, Noble PW (1996) Hyaluronan (HA) fragments induce chemokine gene expression in alveolar macrophages. The role of HA size and CD44. J Clin Invest 98(10):2403–2413
Alstergren P, Zhu B, Glogauer M, Glougauer M, Mak TW, Ellen RP, Sodek J (2004) Polarization and directed migration of murine neutrophils is dependent on cell surface expression of CD44. Cell Immunol 231(1–2):146–157
Takazoe K, Tesch GH, Hill PA, Hurst LA, Jun Z, Lan HY, Atkins RC, Nikolic-Paterson DJ (2000) CD44-mediated neutrophil apoptosis in the rat. Kidney Int 58(5):1920–1930
Weiss JM, Renkl AC, Maier CS, Kimmig M, Liaw L, Ahrens T, Kon S, Maeda M, Hotta H et al (2001) Osteopontin is involved in the initiation of cutaneous contact hypersensitivity by inducing Langerhans and dendritic cell migration to lymph nodes. J Exp Med 194(9):1219–1229
Takano H, Nakazawa S, Shirata N, Tamba S, Furuta K, Tsuchiya S, Morimoto K, Itano N, Irie A et al (2009) Involvement of CD44 in mast cell proliferation during terminal differentiation. Lab Invest 89(4):446–455
Magyarosy E, Sebestyén A, Timár J (2001) Expression of metastasis associated proteins, CD44v6 and NM23-H1, in pediatric acute lymphoblastic leukemia. Anticancer Res 21(1B):819–823
Bendall LJ, Nilsson SK, Khan NI, James A, Bonnet C, Lock RB, Papa R, Bradstock KF, Gottlieb DJ (2004) Role of CD44 variant exon 6 in acute lymphoblastic leukaemia: association with altered bone marrow localisation and increased tumour burden. Leukemia 18(7):1308–1311
Khan NI, Cisterne A, Devidas M, Shuster J, Hunger SP, Shaw PJ, Bradstock KF, Bendall LJ (2008) Expression of CD44, but not CD44v6, predicts relapse in children with B cell progenitor acute lymphoblastic leukemia lacking adverse or favorable genetics. Leuk Lymphoma 49(4):710–718
Vaskova M, Mejstrikova E, Kalina T, Martinkova P, Omelka M, Trka J, Stary J, Hrusak O (2005) Transfer of genomics information to flow cytometry: expression of CD27 and CD44 discriminates subtypes of acute lymphoblastic leukemia. Leukemia 19(5):876–878
Charrad RS, Li Y, Delpech B, Balitrand N, Clay D, Jasmin C, Chomienne C, Smadja-Joffe F (1999) Ligation of the CD44 adhesion molecule reverses blockage of differentiation in human acute myeloid leukemia. Nat Med 5(6):669–676
Charrad R, Gadhoum Z, Qi J, Glachant A, Allouche M, Jasmin C, Chomienne C, Smadja-Joffe F (2002) Effects of anti-CD44 monoclonal antibodies on differentiation and apoptosis of human myeloid leukemia cell lines. Blood 99(1):290–299
Song G, Liao X, Zhou L, Wu L, Feng Y, Han ZC (2004) HI44a, an anti-CD44 monoclonal antibody, induces differentiation and apoptosis of human acute myeloid leukemia cells. Leuk Res 28(10):1089–1096
Bourcier S, Sansonetti A, Durand L, Chomienne C, Robert-Lézénés J, Smadja-Joffe F (2010) CD44-ligation induces, through ERK1/2 pathway, synthesis of cytokines TNF-alpha and IL-6 required for differentiation of THP-1 monoblastic leukemia cells. Leukemia 24(7):1372–1375
Delaunay J, Lecomte N, Bourcier S, Qi J, Gadhoum Z, Durand L, Chomienne C, Robert-Lézénès J, Smadja-Joffe F (2008) Contribution of GM-CSF and IL-8 to the CD44-induced differentiation of acute monoblastic leukemia. Leukemia 22(4):873–876
Nervi B, Link DC, DiPersio JF (2006) Cytokines and hematopoietic stem cell mobilization. J Cell Biochem 99(3):690–705
Telen MJ (2005) Erythrocyte adhesion receptors: blood group antigens and related molecules. Transfus Med Rev 19(1):32–44
Jin L, Hope KJ, Zhai Q, Smadja-Joffe F, Dick JE (2006) Targeting of CD44 eradicates human acute myeloid leukemic stem cells. Nat Med 12(10):1167–1174
Funayama K, Shimane M, Nomura H, Asano S (2010) An evidence for adhesion-mediated acquisition of acute myeloid leukemic stem cell-like immaturities. Biochem Biophys Res Commun 392(3):271–276
Abecassis I, Maes J, Carrier J, Hillion J, Goodhardt M, Medjber K, Wany L, Lanotte M, Karniguian A (2008) Re-expression of DNA methylation-silenced CD44 gene in a resistant NB4 cell line: rescue of CD44-dependent cell death by cAMP. Leukemia 22(3):511–520
Peterson LF, Wang Y, Lo M, Yan M, Kanbe E, Zhang D (2007) The multi-functional cellular adhesion molecule CD44 is regulated by the 8;21 chromosomal translocation. Leukemia 21(9):2010–2019
Attarbaschi A, Mann G, Schumich A, König M, Pickl WF, Haas OA, Gadner H, Dworzak MN (2007) CD44 deficiency is a consistent finding in childhood Burkitt's lymphoma and leukemia. Leukemia 21(5):1110–1113
Rodig SJ, Vergilio J, Shahsafaei A, Dorfman DM (2008) Characteristic expression patterns of TCL1, CD38, and CD44 identify aggressive lymphomas harboring a MYC translocation. Am J Surg Pathol 32(1):113–122
Schniederjan SD, Li S, Saxe DF, Lechowicz MJ, Lee KL, Terry PD, Mann KP (2010) A novel flow cytometric antibody panel for distinguishing Burkitt lymphoma from CD10+ diffuse large B-cell lymphoma. Am J Clin Pathol 133(5):718–726
Stauder R, Eisterer W, Thaler J, Günthert U (1995) CD44 variant isoforms in non-Hodgkin's lymphoma: a new independent prognostic factor. Blood 85(10):2885–2899
Ristamäki R, Joensuu H, Söderström KO, Jalkanen S (1995) CD44v6 expression in non-Hodgkin's lymphoma: an association with low histological grade and poor prognosis. J Pathol 176(3):259–267
Drillenburg P, Wielenga VJ, Kramer MH, van Krieken JH, Kluin-Nelemans HC, Hermans J, Heisterkamp S, Noordijk EM, Kluin PM (1999) CD44 expression predicts disease outcome in localized large B cell lymphoma. Leukemia 13(9):1448–1455
Inagaki H, Banno S, Wakita A, Ueda R, Eimoto T (1999) Prognostic significance of CD44v6 in diffuse large B-cell lymphoma. Mod Pathol 12(5):546–552
Tzankov A, Pehrs A, Zimpfer A, Ascani S, Lugli A, Pileri S, Dirnhofer S (2003) Prognostic significance of CD44 expression in diffuse large B cell lymphoma of activated and germinal centre B cell-like types: a tissue microarray analysis of 90 cases. J Clin Pathol 56(10):747–752
Hu X, Chen Y, Liang ACT, Au W, Wong K, Wan TSK, Wong MLY, Shen L, Chan K et al (2010) CD44 activation in mature B-cell malignancies by a novel recurrent IGH translocation. Blood 115(12):2458–2461
Zarcone D, De Rossi G, Tenca C, Marroni P, Mauro FR, Cerruti GM, Albi N, Fiorucci S, Velardi A et al (1998) Functional and clinical relevance of CD44 variant isoform expression on B-cell chronic lymphocytic leukemia cells. Haematologica 83(12):1088–1098
Bairey O, Zimra Y, Rabizadeh E, Shaklai M (2004) Expression of adhesion molecules on leukemic B cells from chronic lymphocytic leukemia patients with predominantly splenic manifestations. Isr Med Assoc J 6(3):147–151
Molica S, Vitelli G, Levato D, Giannarelli D, Gandolfo GM (2001) Elevated serum levels of soluble CD44 can identify a subgroup of patients with early B-cell chronic lymphocytic leukemia who are at high risk of disease progression. Cancer 92(4):713–719
Eisterer W, Bechter O, Söderberg O, Nilsson K, Terol M, Greil R, Thaler J, Herold M, Finke L et al (2004) Elevated levels of soluble CD44 are associated with advanced disease and in vitro proliferation of neoplastic lymphocytes in B-cell chronic lymphocytic leukaemia. Leuk Res 28(10):1043–1051
Pedersen IM, Kitada S, Leoni LM, Zapata JM, Karras JG, Tsukada N, Kipps TJ, Choi YS, Bennett F et al (2002) Protection of CLL B cells by a follicular dendritic cell line is dependent on induction of Mcl-1. Blood 100(5):1795–1801
Redondo-Muñoz J, Ugarte-Berzal E, García-Marco JA, del Cerro MH, Van den Steen PE, Opdenakker G, Terol MJ, García-Pardo A (2008) Alpha4beta1 integrin and 190-kDa CD44v constitute a cell surface docking complex for gelatinase B/MMP-9 in chronic leukemic but not in normal B cells. Blood 112(1):169–178
Kim I, Uchiyama H, Chauhan D, Anderson KC (1994) Cell surface expression and functional significance of adhesion molecules on human myeloma-derived cell lines. Br J Haematol 87(3):483–493
Van Driel M, Günthert U, van Kessel AC, Joling P, Stauder R, Lokhorst HM, Bloem AC (2002) CD44 variant isoforms are involved in plasma cell adhesion to bone marrow stromal cells. Leukemia 16(1):135–143
Asosingh K, Günthert U, Bakkus MH, De Raeve H, Goes E, Van Riet I, Van Camp B, Vanderkerken K (2000) In vivo induction of insulin-like growth factor-I receptor and CD44v6 confers homing and adhesion to murine multiple myeloma cells. Cancer Res 60(11):3096–3104
Caers J, Günthert U, De Raeve H, Van Valckenborgh E, Menu E, Van Riet I, Van Camp B, Vanderkerken K (2006) The involvement of osteopontin and its receptors in multiple myeloma cell survival, migration and invasion in the murine 5T33MM model. Br J Haematol 132(4):469–477
Stauder R, Van Driel M, Schwärzler C, Thaler J, Lokhorst HM, Kreuser ED, Bloem AC, Günthert U, Eisterer W (1996) Different CD44 splicing patterns define prognostic subgroups in multiple myeloma. Blood 88(8):3101–3108
Eisterer W, Bechter O, Hilbe W, van Driel M, Lokhorst HM, Thaler J, Bloem AC, Günthert U, Stauder R (2001) CD44 isoforms are differentially regulated in plasma cell dyscrasias and CD44v9 represents a new independent prognostic parameter in multiple myeloma. Leuk Res 25(12):1051–1057
Liebisch P, Eppinger S, Schöpflin C, Stehle G, Munzert G, Döhner H, Schmid M (2005) CD44v6, a target for novel antibody treatment approaches, is frequently expressed in multiple myeloma and associated with deletion of chromosome arm 13q. Haematologica 90(4):489–493
Ma Y, Visser L, Roelofsen H, de Vries M, Diepstra A, van Imhoff G, van der Wal T, Luinge M, Alvarez-Llamas G et al (2008) Proteomics analysis of Hodgkin lymphoma: identification of new players involved in the cross-talk between HRS cells and infiltrating lymphocytes. Blood 111(4):2339–2346
Kim Y, Seo D, Kong S, Lee J, Lee E, Stetler-Stevenson M, Stetler-Stevenson WG (2008) TIMP1 induces CD44 expression and the activation and nuclear translocation of SHP1 during the late centrocyte/post-germinal center B cell differentiation. Cancer Lett 269(1):37–45
Beham-Schmid C, Heider KH, Hoefler G, Zatloukal K (1998) Expression of CD44 splice variant v10 in Hodgkin's disease is associated with aggressive behaviour and high risk of relapse. J Pathol 186(4):383–389
Heider KH, Sproll M, Susani S, Patzelt E, Beaumier P, Ostermann E, Ahorn H, Adolf GR (1996) Characterization of a high-affinity monoclonal antibody specific for CD44v6 as candidate for immunotherapy of squamous cell carcinomas. Cancer Immunol Immunother 43(4):245–253
Börjesson PKE, Postema EJ, Roos JC, Colnot DR, Marres HAM, van Schie MH, Stehle G, de Bree R, Snow GB et al (2003) Phase I therapy study with (186)Re-labeled humanized monoclonal antibody BIWA 4 (bivatuzumab) in patients with head and neck squamous cell carcinoma. Clin Cancer Res 9(10 Pt 2):3961S–3972S
Colnot DR, Wilhelm AJ, Cloos J, Roos JC, de Bree R, Quak JJ, Snow GB, van Dongen GA (2001) Evaluation of limited blood sampling in a preceding 99mTc-labeled diagnostic study to predict the pharmacokinetics and myelotoxicity of 186Re-cMAb U36 radioimmunotherapy. J Nucl Med 42(9):1364–1367
Colnot DR, Ossenkoppele GJ, Roos JC, Quak JJ, de Bree R, Börjesson PK, Huijgens PC, Snow GB, van Dongen GAMS (2002) Reinfusion of unprocessed, granulocyte colony-stimulating factor-stimulated whole blood allows dose escalation of 186Relabeled chimeric monoclonal antibody U36 radioimmunotherapy in a phase I dose escalation study. Clin Cancer Res 8(11):3401–3406
Stroomer JW, Roos JC, Sproll M, Quak JJ, Heider KH, Wilhelm BJ, Castelijns JA, Meyer R, Kwakkelstein MO et al (2000) Safety and biodistribution of 99mTechnetium-labeled anti-CD44v6 monoclonal antibody BIWA 1 in head and neck cancer patients. Clin Cancer Res 6(8):3046–3055
Colnot DR, Roos JC, de Bree R, Wilhelm AJ, Kummer JA, Hanft G, Heider K, Stehle G, Snow GB et al (2003) Safety, biodistribution, pharmacokinetics, and immunogenicity of 99mTc-labeled humanized monoclonal antibody BIWA 4 (bivatuzumab) in patients with squamous cell carcinoma of the head and neck. Cancer Immunol Immunother 52(9):576–582
Tijink BM, Buter J, de Bree R, Giaccone G, Lang MS, Staab A, Leemans CR, van Dongen GAMS (2006) A phase I dose escalation study with anti-CD44v6 bivatuzumab mertansine in patients with incurable squamous cell carcinoma of the head and neck or esophagus. Clin Cancer Res 12(20 Pt 1):6064–6072
Koppe M, Schaijk FV, Roos J, Leeuwen PV, Heider K, Kuthan H, Bleichrodt R (2004) Safety, pharmacokinetics, immunogenicity, and biodistribution of (186)Re-labeled humanized monoclonal antibody BIWA 4 (Bivatuzumab) in patients with early-stage breast cancer. Cancer Biother Radiopharm 19(6):720–729
Börjesson PKE, Jauw YWS, de Bree R, Roos JC, Castelijns JA, Leemans CR, van Dongen GAMS, Boellaard R (2009) Radiation dosimetry of 89Zr-labeled chimeric monoclonal antibody U36 as used for immuno-PET in head and neck cancer patients. J Nucl Med 50(11):1828–1836
Börjesson PKE, Jauw YWS, Boellaard R, de Bree R, Comans EFI, Roos JC, Castelijns JA, Vosjan MJWD, Kummer JA et al (2006) Performance of immuno-positron emission tomography with zirconium-89-labeled chimeric monoclonal antibody U36 in the detection of lymph node metastases in head and neck cancer patients. Clin Cancer Res 12(7 Pt 1):2133–2140
Coradini D, Zorzet S, Rossin R, Scarlata I, Pellizzaro C, Turrin C, Bello M, Cantoni S, Speranza A et al (2004) Inhibition of hepatocellular carcinomas in vitro and hepatic metastases in vivo in mice by the histone deacetylase inhibitor HA-But. Clin Cancer Res 10(14):4822–4830
Li S, Howell SB (2010) CD44-targeted microparticles for delivery of cisplatin to peritoneal metastases. Mol Pharm 7(1):280–290
Peer D, Margalit R (2004) Loading mitomycin C inside long circulating hyaluronan targeted nano-liposomes increases its antitumor activity in three mice tumor models. Int J Cancer 108(5):780–789
Saravanakumar G, Choi KY, Yoon HY, Kim K, Park JH, Kwon IC, Park K (2010) Hydrotropic hyaluronic acid conjugates: synthesis, characterization, and implications as a carrier of paclitaxel. Int J Pharm 394(1–2):154–161
Auzenne E, Ghosh SC, Khodadadian M, Rivera B, Farquhar D, Price RE, Ravoori M, Kundra V, Freedman RS et al (2007) Hyaluronic acid-paclitaxel: antitumor efficacy against CD44(+) human ovarian carcinoma xenografts. Neoplasia 9(6):479–486
Yang K, Tang Y, Habermehl GK, Iczkowski KA (2010) Stable alterations of CD44 isoform expression in prostate cancer cells decrease invasion and growth and alter ligand binding and chemosensitivity. BMC Cancer 10:16
Cordo Russo RI, García MG, Alaniz L, Blanco G, Alvarez E, Hajos SE (2008) Hyaluronan oligosaccharides sensitize lymphoma resistant cell lines to vincristine by modulating P-glycoprotein activity and PI3K/Akt pathway. Int J Cancer 122(5):1012–1018
Nagel S, Hirschmann P, Dirnhofer S, Günthert U, Tzankov A (2010) Coexpression of CD44 variant isoforms and receptor for hyaluronic acid-mediated motility (RHAMM, CD168) is an International Prognostic Index and C-MYC gene status-independent predictor of poor outcome in diffuse large B-cell lymphomas. Exp Hematol 38(1):38–45
Ohwada C, Nakaseko C, Koizumi M, Takeuchi M, Ozawa S, Naito M, Tanaka H, Oda K, Cho R et al (2008) CD44 and hyaluronan engagement promotes dexamethasone resistance in human myeloma cells. Eur J Haematol 80(3):245–250
Tajima K, Ohashi R, Sekido Y, Hida T, Nara T, Hashimoto M, Iwakami S, Minakata K, Yae T et al (2010) Osteopontin-mediated enhanced hyaluronan binding induces multidrug resistance in mesothelioma cells. Oncogene 29(13):1941–1951
Runnels HA, Weber GL, Min J, Kudlacz EM, Zobel JF, Donovan CB, Thiede MA, Zhang J, Alpert RB et al (2010) PF-03475952: a potent and neutralizing fully human anti-CD44 antibody for therapeutic applications in inflammatory diseases. Adv Ther 27(3):168–180
Grosso S, Puissant A, Dufies M, Colosetti P, Jacquel A, Lebrigand K, Barbry P, Deckert M, Cassuto JP et al (2009) Gene expression profiling of imatinib and PD166326-resistant CML cell lines identifies Fyn as a gene associated with resistance to BCR-ABL inhibitors. Mol Cancer Ther 8(7):1924–1933
Bourguignon LYW, Singleton PA, Diedrich F (2004) Hyaluronan-CD44 interaction with Rac1-dependent protein kinase N-gamma promotes phospholipase Cgamma1 activation, Ca(2+) signaling, and cortactin-cytoskeleton function leading to keratinocyte adhesion and differentiation. J Biol Chem 279(28):29654–29669
Lin YH, Yang-Yen HF (2001) The osteopontin-CD44 survival signal involves activation of the phosphatidylinositol 3-kinase/Akt signaling pathway. J Biol Chem 276(49):46024–46030
Hamilton SR, Fard SF, Paiwand FF, Tolg C, Veiseh M, Wang C, McCarthy JB, Bissell MJ, Koropatnick J et al (2007) The hyaluronan receptors CD44 and Rhamm (CD168) form complexes with ERK1,2 that sustain high basal motility in breast cancer cells. J Biol Chem 282(22):16667–16680
Eshkar Sebban L, Ronen D, Levartovsky D, Elkayam O, Caspi D, Aamar S, Amital H, Rubinow A, Golan I et al (2007) The involvement of CD44 and its novel ligand galectin-8 in apoptotic regulation of autoimmune inflammation. J Immunol 179(2):1225–1235
Sherman L, Wainwright D, Ponta H, Herrlich P (1998) A splice variant of CD44 expressed in the apical ectodermal ridge presents fibroblast growth factors to limb mesenchyme and is required for limb outgrowth. Genes Dev 12(7):1058–1071
Orian-Rousseau V, Chen L, Sleeman JP, Herrlich P, Ponta H (2002) CD44 is required for two consecutive steps in HGF/c-Met signaling. Genes Dev 16(23):3074–3086
Recio JA, Merlino G (2003) Hepatocyte growth factor/scatter factor induces feedback up-regulation of CD44v6 in melanoma cells through Egr-1. Cancer Res 63(7):1576–1582
Damm S, Koefinger P, Stefan M, Wels C, Mehes G, Richtig E, Kerl H, Otte M, Schaider H (2010) HGF-promoted motility in primary human melanocytes depends on CD44v6 regulated via NF-kappa B, Egr-1, and C/EBP-beta. J Invest Dermatol 130(7):1893–1903
Bennett KL, Jackson DG, Simon JC, Tanczos E, Peach R, Modrell B, Stamenkovic I, Plowman G, Aruffo A (1995) CD44 isoforms containing exon V3 are responsible for the presentation of heparin-binding growth factor. J Cell Biol 128(4):687–698
Yu W, Woessner JF, McNeish JD, Stamenkovic I (2002) CD44 anchors the assembly of matrilysin/MMP-7 with heparin-binding epidermal growth factor precursor and ErbB4 and regulates female reproductive organ remodeling. Genes Dev 16(3):307–323
Monaghan M, Mulligan KA, Gillespie H, Trimble A, Winter P, Johnston PG, McCormick D (2000) Epidermal growth factor up-regulates CD44-dependent astrocytoma invasion in vitro. J Pathol 192(4):519–525
Tremmel M, Matzke A, Albrecht I, Laib AM, Olaku V, Ballmer-Hofer K, Christofori G, Héroult M, Augustin HG et al (2009) A CD44v6 peptide reveals a role of CD44 in VEGFR-2 signaling and angiogenesis. Blood 114(25):5236–5244
Bourguignon LY, Zhu H, Chu A, Iida N, Zhang L, Hung MC (1997) Interaction between the adhesion receptor, CD44, and the oncogene product, p185HER2, promotes human ovarian tumor cell activation. J Biol Chem 272(44):27913–27918
Bourguignon LYW, Peyrollier K, Gilad E, Brightman A (2007) Hyaluronan-CD44 interaction with neural Wiskott–Aldrich syndrome protein (N-WASP) promotes actin polymerization and ErbB2 activation leading to beta-catenin nuclear translocation, transcriptional up-regulation, and cell migration in ovarian tumor cells. J Biol Chem 282(2):1265–1280
Ito T, Williams JD, Fraser D, Phillips AO (2004) Hyaluronan attenuates transforming growth factor-beta1-mediated signaling in renal proximal tubular epithelial cells. Am J Pathol 164(6):1979–1988
Takahashi K, Eto H, Tanabe KK (1999) Involvement of CD44 in matrix metalloproteinase-2 regulation in human melanoma cells. Int J Cancer 80(3):387–395
Thanakit V, Sampatanukul P, Ruangvejvorachai P, Keelawat S (2005) The association of co-expression of CD44v4/MMP-9 with different nodal status in high-grade breast carcinoma patients. J Med Assoc Thai 88(Suppl 4):S30–S35
Julovi SM, Ito H, Nishitani K, Jackson CJ, Nakamura T (2010) Hyaluronan inhibits matrix metalloproteinase-13 in human arthritic chondrocytes via CD44 and P38. J Orthop Res 2:258–264
Denning SM, Le PT, Singer KH, Haynes BF (1990) Antibodies against the CD44 p80, lymphocyte homing receptor molecule augment human peripheral blood T cell activation. J Immunol 144(1):7–15
Zhang L, Ma H, Greyner HJ, Zuo W, Mummert ME (2010) Inhibition of cell proliferation by CD44: Akt is inactivated and EGR-1 is down-regulated. Cell Prolif 43(4):385–395
Gore Y, Starlets D, Maharshak N, Becker-Herman S, Kaneyuki U, Leng L, Bucala R, Shachar I (2008) Macrophage migration inhibitory factor induces B cell survival by activation of a CD74–CD44 receptor complex. J Biol Chem 283(5):2784–2792
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hertweck, M.K., Erdfelder, F. & Kreuzer, KA. CD44 in hematological neoplasias. Ann Hematol 90, 493–508 (2011). https://doi.org/10.1007/s00277-011-1161-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-011-1161-z