Abstract
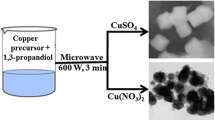
In this work, we demonstrate that using a glycolic medium, both stable CuO and Cu2O nanoparticles can be elaborated from Cu colloidal particles by adjusting their chemical environment, in particular the acido-basicity of the solution. In this context, the effect of pH on the sol–gel synthesis of Cu2O/CuO NPs was investigated. XRD results confirmed the formation of pure Cu2O with a cubic structure at lower pH (pH ≤ 6), whereas the pure monoclinic CuO was formed at higher pH (pH ≥ 12). TEM image indicates that the as-formed CuO NPs in basic pH are spherical in shape and their average size is found to be in the range of 4.5 nm. However, the as-obtained Cu2O NPs in acid pH are cubical, with an average diameter of about 3 nm, and agglomerated into large spherical particles under the effect of ethylene glycol. Using the UV–Vis spectroscopy, the measured band gap energies of the prepared Cu2O and CuO NPs are 2.07 and 4.08 eV respectively. FTIR results confirm the purity of the synthesized CuO and Cu2O nanoparticles.






Similar content being viewed by others
References
M.A. El-Sayed, Acc. Chem. Res. 37, 326 (2004)
A.P. Alivisatos, Endeavour 21, 56 (1997)
A.P. Alivisatos, Science (1996). doi:10.1126/science.271.5251.933
X. Chen, S.S. Mao, Chem. Rev. 107, 2959 (2007)
Z.L. Wang, J. Phy. Condens. Matter 16, 858 (2004)
H. Zheng, J.Z. Ou, M.S. Strano, R.B. Kaner, A. Mitchell, K. Kalantar-zadeh, Adv. Funct. Mater. (2011). doi:10.1002/adfm.201002477
J. Pan, H. Shen, S. Mathur, J. Nanotechnol. (2012). doi:10.1155/2012/917320
A. Cruccolini, R. Narducci, R. Palombari, Sens. Actuators B: Chemical (2004). doi:10.1016/j.snb.2003.10.012
A.O. Musa, T. Akomolafe, M.J. Carter, Sol. Energy Mater. Sol. Cells 51, 305 (1998)
X.G. Zheng, C.N. Xu, Y. Tomokiyo, E. Tanaka, H. Yamada, Y. Soejima, Phys. Rev. Lett. 85, 5170 (2000)
X. Zhang, D. Zhang, X. Ni, H. Zheng, Solid-State Electronics 52, 245 (2008)
L.P. Zhou, B.X. Wang, X.F. Peng, X.Z. Du, Y.P. Yang, Adv. Mech. Eng. (2010). doi:10.1155/2010/172085
S. Seung-Deok, J. Yun-Ho, L. Seung-Hun, S. Hyun-Woo, K. Dong-Wan, Nanoscale Res. Lett. 6, 2 (2011)
Y. Jiang, S. Decker, C. Mohs, K.J. Klabunde, J. Catal. 180, 24 (1998)
C.H.B. Ng, W.Y. Fan, J. Phys. Chem. B 110, 20801 (2006)
C.-H. Kuo, M.H. Huang, J. Phys. Chem. C 112, 18355 (2008)
J.-Y. Ho, M.H. Huang, J. Phys. Chem. C 113, 14159 (2009)
C.-H. Kuo, C.-H. Chen, M.H. Huang, Adv. Funct. Mater. 17, 3773 (2007)
B. White, M. Yin, A. Hall, D. Le, S. Stolbov, T. Rahman, N. Turro, S. O’Brien, Nano Lett. 6, 2095 (2006)
M. Hara, T. Kondo, M. Komoda, S. Ikeda, J.N. Kondo, K. Domen, K. Shinohara, A. Tanaka, Chem. Commun. (1998). doi:10.1039/A707440I
C.M. McShane, K.S. Choi, J. Am. Chem. Soc. 131, 2561 (2009)
Z. Yang, C.K. Chiang, H.T. Chang, Nanotechnology (2008). doi:10.1088/0957-4484/19/02/025604
C. Xu, Y. Liu, G. Xu, G. Wang, Mater. Res. Bull. 37, 2365 (2002)
R.V. Kumar, Y. Diamant, A. Gendanken, Chem. Mater. 12, 2301 (2000)
R.V. Kumar, Y. Diamant, A. Gendanken, R. Elgamiel, Langmuir 17, 1406 (2001)
H. Xu, W. Wang, W. Zhu, Micro. Meso. Mater. 95, 321 (2006)
L. Gou, C. Murphy, J. Nano Lett. 3, 231 (2003)
J. Zhang, J. Liu, Q. Peng, X. Wang, Y. Li, Chem. Mater. 18, 867 (2006)
J.C. Park, J. Kim, H. Kwon, H. Song, Adv. Mater. 21, 803 (2009)
Brinker J, Scherer GW (1990) Academic Press, New York
W.Z. Wang, O.K. Varghese, C.M. Ruan, M. Paulose, C.A. Grimes, J. Mater. Res. 18, 2756 (2003)
A. Li, P. Li, J. Hu, W. Zhang, J. Mater. Sci.: Mater. Electron. 26, 5071 (2015)
X. Deng, Q. Zhang, Q. Zhao, L. Ma, M. Ding, X. Xu, Nanoscale Res. Lett. 10, 2 (2015)
B. Jaber, L. Laânab, Mater. Sci. Semicond. Process. 27, 446 (2014)
M. Cao, C. Hu, Y. Wang, Y. Guo, C. Guo, E. Wang, Chem. Commun. 15, 1884 (2003)
S. Pande, S. Jana, A.K. Sinha, A. Datta, T. Pal, J. Phys. Chem. C 112, 3619 (2008)
M. Kooti, L. Matouri, Tran. F : Nanotechnology 17, 73 (2010)
G. Kliche, Z.V. Popovic, Phys Rev B 42, 10060 (1990)
Y.C. Zhang, J.Y. Tang, G.L. Wang, M. Zhang, X.Y. Hu, J. Cryst. Growth 294, 278 (2006)
I. Prakash, P. Muralidharan, N. Nallamuthu, M. Venkateswarlu, N. Satyanarayana, Mater. Res. Bull. 42, 1619 (2007)
O. Game, U. Singh, A.A. Gupta, A. Suryawanshi, A. Banpurkar, S. Ogale, J. Mater. Chem. 22, 17302 (2012)
A. Patra, K. Rajesh, T.P. Radhakrishnan, Bull. Mater. Sci. 31, 421 (2008)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zayyoun, N., Bahmad, L., Laânab, L. et al. The effect of pH on the synthesis of stable Cu2O/CuO nanoparticles by sol–gel method in a glycolic medium. Appl. Phys. A 122, 488 (2016). https://doi.org/10.1007/s00339-016-0024-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-016-0024-9