Abstract
Background
Adipogenous tissue derived stem cells (ASC) are available in abundance in the human body and can differentiate in the presence of lineage-specific induction factors, for example, in myogenic, adipogenic, chondrogenic and osteogenic cells. The aim of this study was to evaluate the impact of osteogenic induced ASC’s (O-ASC) on revascularization and cellular repopulation of avital cortical bone employing a vascularized bovine scaffold.
Methods
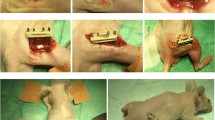
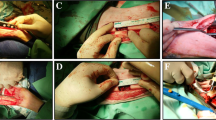
An inguinal arterio-venous bundle was dissected in the groin of female white New Zealand rabbits (n = 6) and placed centrally inside an O-ASC seeded scaffold via a central drill hole. In the same surgical session this construct was placed into a segment of avital cortical bone allograft from a donor rabbit. Unseeded scaffolds that were implanted and treated in the same fashion served as controls (n = 6). In order to prevent external revascularization, all constructs were wrapped in silicon foil and finally implanted in the rabbits’ groin. Three months later, the constructs were explanted and investigated for vascularization of (a) the scaffold (b) the surrounding bone allograft. Histological stainings to determine cell growth, cellular repopulation of the scaffold and the cortical bone matrix, as well as inflammatory parameters were carried out.
Results
O-ASC seeded scaffolds showed a significant increase in new vessel formation in the scaffold as well as in the bone allograft compared to unseeded scaffolds. Furthermore, new vital osteocytes as a sign of cellular repopulation inside the bone allograft were found only in the treatment group. Vital chondrocytes were only found in the O-ASC seeded scaffolds as well.
Conclusion
The presence of O-ASC significantly induce neo-vascularization and osteocytic repopulation of previously avital bone allograft as opposed to unseeded scaffolds in a rabbit model. Hence, this model might be of relevant value for future bone tissue engineering research and for re-vitalizing marginally nourished bone such as in avascular bone necrosis.









Similar content being viewed by others
References
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH (2001) Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7:211–228
Lee JA, Parrett BM, Conejero JA, Laser J, Chen J, Kogon AJ, Nanda D, Grant RT, Breitbart AS (2003) Biological alchemy: engineering bone and fat from fat-derived stem cells. Ann Plast Surg 50:610–617
Conejero JA, Lee JA, Parrett BM, Terry M, Wear-Maggitti K, Grant RT, Breitbart AS (2006) Repair of palatal bone defects using osteogenically differentiated fat-derived stem cells. Plast Reconstr Surg 117:857–863
Huang JI, Beanes SR, Zhu M, Lorenz HP, Hedrick MH, Benhaim P (2002) Rat extramedullary adipose tissue as a source of osteochondrogenic progenitor cells. Plast Reconstr Surg 109:1033–1041
Wood MB (1986) Free vascularized bone transfers for nonunions, segmental gaps, and following tumor resection. Orthopedics 9:810–816
de Boer HH, Wood MB, Hermans J (1990) Reconstruction of large skeletal defects by vascularized fibula transfer. Factors that influenced the outcome of union in 62 cases. Int Orthop 14:121–128
Shaffer JW, Field GA, Goldberg VM et al (1985) Fate of vascularized and nonvascularized autografts. Clin Orthop 197:32–43
Erdmann D, Giessler GA, Bergquist GE et al (2004) Free fibula transfer. Analysis of 76 consecutive microsurgical procedures and review of the literature. Chirurg 75:799–809
Follmar KE, Prichard HL, DeCroos FC, Wang HT, Levin LS, Klitzman B, Olbrich KC, Erdmann D (2007) Ann Plast Surg 58:561–565
De Ugarte DA, Ashjian PH, Elbarbary A, Hedrick MH (2003) Future of fat as raw material for tissue regeneration. Ann Plast Surg 50:215–219
Oswald J, Boxberger S, Jørgensen B, Feldmann S, Ehninger G, Bornhäuser M, Werner C (2004) Mesenchymal stem cells can be differentiated into endothelial cells in vitro. Stem Cells 22:377–384
Al-Khaldi A, Al-Sabti H, Galipeau J, Lachapelle K (2003) Therapeutic angiogenesis using autologous bone marrow stromal cells: improved blood flow in a chronic limb ischemia model. Ann Thorac Surg 75:204–209
Kim Y, Kim H, Cho H, Bae Y, Suh K, Jung J (2007) Direct comparison of human mesenchymal stem cells derived from adipose tissues and bone marrow in mediating neovascularization in response to vascular ischemia. Cell Physiol Biochem 20:867–876
Kaigler D, Krebsbach PH, Polverini PJ, Mooney DJ (2003) Role of vascular endothelial growth factor in bone marrow stromal cell modulation of endothelial cells. Tissue Eng 9:95–103
Lee RH, Kim B, Choi I, Kim H, Choi HS, Suh K, Bae YC, Jung JS (2004) Characterization and expression analysis of mesenchymal stem cells from human bone marrow and adipose tissue. Cell Physiol Biochem 14:311–324
Kim SJ, Cho HH, Kim YJ, Seo SY, Kim HN, Lee JB, Kim JH, Chung JS, Jung JS (2005) Human adipose stromal cells expanded in human serum promote engraftment of human peripheral blood hematopoietic stem cells in NOD/SCID mice. Biochem Biophys Res Commun 329:25–31
Miranville A, Heeschen C, Sengenès C, Curat CA, Busse R, Bouloumié A (2004) Improvement of postnatal neovascularization by human adipose tissue-derived stem cells. Circulation 110:349–355
Planat-Benard V, Silvestre JS, Cousin B, André M, Nibbelink M, Tamarat R, Clergue M, Manneville C, Saillan-Barreau C, Duriez M, Tedgui A, Levy B, Pénicaud L, Casteilla L (2004) Plasticity of human adipose lineage cells toward endothelial cells: physiological and therapeutic perspectives. Circulation 109:656–663
Rehman J, Traktuev D, Li J, Merfeld-Clauss S, Temm-Grove CJ, Bovenkerk JE, Pell CL, Johnstone BH, Considine RV, March KL (2004) Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation 109:1292–1298
Moon MH, Kim SY, Kim YJ, Kim SJ, Lee JB, Bae YC, Jung JS (2006) Human adipose tissue-derived mesenchymal stem cells improve postnatal neovascularization in a mouse model of hindlimb ischemia. Cell Physiol Biochem 17:279–290
Dufourcq P, Descamps B, Tojais NF, Leroux L, Oses P, Daret D, Moreau C, Lamazière JM, Couffinhal T, Duplàa C (2008) Secreted frizzled-related protein-1 enhances mesenchymal stem cell function in angiogenesis and contributes to neovessel maturation. Stem Cells 26:2991–3001
Nakagami H, Maeda K, Morishita R, Iguchi S, Nishikawa T, Takami Y, Kikuchi Y, Saito Y, Tamai K, Ogihara T, Kaneda Y (2005) Novel autologous cell therapy in ischemic limb disease through growth factor secretion by cultured adipose tissue-derived stromal cells. Arterioscler Thromb Vasc Biol 25:2542–2547
Constantin G, Marconi S, Rossi B, Angiari S, Calderan L, Anghileri E, Gini B, Bach SD, Martinello M, Bifari F, Galiè M, Turano E, Budui S, Sbarbati A, Krampera M, Bonetti B (2009) Adipose-derived mesenchymal stem cells ameliorate chronic experimental autoimmune encephalomyelitis. Stem Cells 27:2624–2635
González MA, Gonzalez-Rey E, Rico L, Büscher D, Delgado M (2009) Treatment of experimental arthritis by inducing immune tolerance with human adipose- derived mesenchymal stem cells. Arthritis Rheum 60:1006–1019
Gonzalez-Rey E, Anderson P, González MA, Rico L, Büscher D, Delgado M (2009) Human adult stem cells derived from adipose tissue protect against experimental colitis and sepsis. Gut 58:929–939
González MA, Gonzalez-Rey E, Rico L, Büscher D, Delgado M (2009) Adipose- derived mesenchymal stem cells alleviate experimental colitis by inhibiting inflammatory and autoimmune responses. Gastroenterology 136:978–989
Gonzalez-Rey E, Gonzalez MA, Varela N, O’Valle F, Hernandez-Cortes P, Rico L, Büscher D, Delgado M (2010) Human adipose-derived mesenchymal stem cells reduce inflammatory and T cell responses and induce regulatory T cells in vitro in rheumatoid arthritis. Ann Rheum Dis 69:241–248
Acknowledgments
This study was supported by a grant from the “Dietmar-Hopp-Stiftung”, Raiffeisenring 51, 68789 St. Leon-Rot, Germany; http://www.dietmar-hopp-stiftung.de. We gratefully acknowledge the laboratory work of Monika Engstner, Regina Beck and Anna Dyjas.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kloeters, O., Berger, I., Ryssel, H. et al. Revitalization of cortical bone allograft by application of vascularized scaffolds seeded with osteogenic induced adipose tissue derived stem cells in a rabbit model. Arch Orthop Trauma Surg 131, 1459–1466 (2011). https://doi.org/10.1007/s00402-011-1306-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-011-1306-5