Abstract
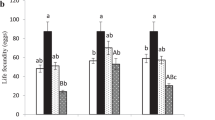
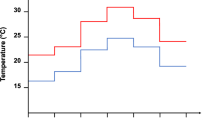
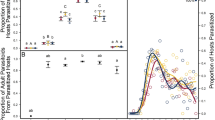
Coexistence of species sharing the same resources is often possible if species are phylogenetically divergent in resource acquisition and allocation traits, decreasing competition between them. Developmental and life-history traits related to resource use are influenced by environmental conditions such as temperature, but thermal trait responses may differ among species. An increase in ambient temperature may, therefore, affect trait divergence within a community, and potentially species coexistence. Parasitoids are interesting models to test this hypothesis, because multiple species commonly attack the same host, and employ divergent larval and adult host use strategies. In particular, development mode (arrested or continued host growth following parasitism) has been recognized as a major organiser of parasitoid life histories. Here, we used a comparative trait-based approach to determine thermal responses of development time, body mass, egg load, metabolic rate and energy use of the coexisting Drosophila parasitoids Asobara tabida, Leptopilina heterotoma, Trichopria drosophilae and Spalangia erythromera. We compared trait values between species and development modes, and calculated trait divergence in response to temperature, using functional diversity indices. Parasitoids differed in their thermal response for dry mass, metabolic rate and lipid use throughout adult life, but only teneral lipid reserves and egg load were affected by developmental mode. Species-specific trait responses to temperature were probably determined by their adaptations in resource use (e.g. lipogenesis or ectoparasitism). Overall, trait values of parasitoid species converged at the higher temperature. Our results suggest that local effects of warming could affect host resource partitioning by reducing trait diversity in communities.



Similar content being viewed by others
References
Angilletta MJ, Steury TD, Sears MW (2004) Temperature, growth rate, and body size in ectotherms: fitting pieces of a life-history puzzle. Int Comp Biol 44:498–509
Ashton IW, Miller AE, Bowman WD, Suding KN (2010) Niche complementarity due to plasticity in resource use: plant partitioning of chemical N forms. Ecology 91:3252–3260
Askew RR, Shaw MR (1986) Parasitoid communities: their size structure and development. In: Waage J, Greathead D (eds) Insect parasitoids. Academic, London, pp 225–264
Atkinson D (1994) Temperature and organism size—a biological law for ectotherms. Adv Ecol Res 25:1–58
Baffoe KO, Dalin P, Nordlander G, Stenberg JA (2012) Importance of temperature for the performance and biocontrol efficiency of the parasitoid Perilitus brevicollis (Hymenoptera: Braconidae) on Salix. Biocontrol 57:611–618
Bazzocchi GG, Lanzoni A, Burgio G, Fiacconi MR (2003) Effects of temperature and host on the pre-imaginal development of the parasitoid Diglyphus isaea (Hymenoptera: Eulophidae). Biol Control 26:74–82
Berg MP, Ellers J (2010) Trait plasticity in species interactions: a driving force of community dynamics. Evol Ecol 24:617–629
Berg MP, Kiers ET, Driessen G, Vanderheijden MW, Kooi BW, Kuenen F, Liefting M, Verhoef HA, Ellers J (2010) Adapt or disperse: understanding species persistence in a changing world. Glob Change Biol 16:587–598
Blackburn TM (1991a) A comparative examination of life span and fecundity in parasitoid Hymenoptera. J Anim Ecol 60:151–164
Blackburn TM (1991b) Evidence for a ‘fast-slow’ continuum of life–history traits among parasitoid Hymenoptera. Funct Ecol 5:65–74
Bochdanovits Z, De Jong G (2003) Temperature dependence of fitness components in geographical populations of Drosophila melanogaster: changing the association between size and fitness. Biol J Linn Soc 80:717–725
Boivin G (2010) Phenotypic plasticity and fitness in egg parasitoids. Neotrop Entomol 39:457–463
Canham CD, Kobe RK, Latty EF, Chazdon RL (1999) Interspecific and intraspecific variation in tree seedling survival: effects of allocation to roots versus carbohydrate reserves. Oecologia 121:1–11
Carmona CP, Azcárate FM, de Bello F, Ollero HS, Lepš J, Peco B (2012) Taxonomical and functional diversity turnover in Mediterranean grasslands: interactions between grazing, habitat type and rainfall. J Appl Ecol 49:1084–1093
Carton Y, Boulétreau M, van Alphen JJM, Van Lenteren JC (1986) The Drosophila parasitic wasps. In: Ashburner M, Carson HL (eds) The genetics and biology of Drosophila (volume 3). Academic, London, pp 347–394
Cavieres G, Sabat P (2008) Geographic variation in the response to thermal acclimation in rufous-collared sparrows: are physiological flexibility and environmental heterogeneity correlated? Funct Ecol 22:509–515
Chase JM, Leibold MA (2003) Ecological niches linking classical and contemporary approaches. University of Chicago Press, Chicago
Clarke A (1993) Seasonal acclimatization and latitudinal compensation in metabolism—Do they exist? Funct Ecol 7:139–149
Cohen JE, JonssonT Muller CB, Godfray HCJ, Savage VM (2005) Body sizes of hosts and parasitoids in individual feeding relationships. Proc Natl Acad Sci USA 102:684–689
Coleman JS, Bazzaz FA (1992) Effects of CO2 and temperature on growth and resource use of co-occurring C3 and C4 annuals. Ecology 73:1244–1259
Colinet H, Boivin G, Hance T (2007) Manipulation of parasitoid size using the temperature-size rule: fitness consequences. Oecologia 152:425–433
Dosdall LM, Zalucki MP, Tansey JA, Fur long MJ (2012) Developmental responses of the diamondback moth parasitoid Diadegma semiclausum (Hellén) (Hymenoptera: Ichneumonidae) to temperature and host plant species. B Entomol Res 102:373–384
Eijs IEM, Van Alphen JJM (1999) Life history correlations: why are hymenopteran parasitoids an exception? Ecol Lett 2:27–35
Eijs IEM, Ellers J, Van Duinen DG (1998) Feeding strategies in drosophilid parasitoids: the impact of natural food resources on energy reserves in females. Ecol Entomol 23:133–138
Ellers J (1996) Fat and eggs: an alternative method to measure the trade–off between survival and reproduction in insect parasitoids. Neth J Zool 46:227–235
Ellers J, van Alphen JJM (1997) Life history evolution in Asobara tabida: plasticity in allocation of fat reserves to survival and reproduction. J Evol Biol 10:771–785
Ellers J, van Alphen JJM, Sevenster JG (1998) A field study of size-fitness relationships in the parasitoid Asobara tabida. J Anim Ecol 67:318–324
Ellers J, Sevenster JG, Driessen G (2000) Egg load evolution in parasitoids. Am Nat 156:650–665
Elliott NC, Burd JD, Kindler SD, Lee JH (1995) Temperature effects on development of three cereal aphid parasitoids (Hymenoptera: Aphididae). Great Lakes Entomol 28:199–204
Fleury F, Gibert P, Ris N, Allemand R (2009) Ecology and life history evolution of frugivorous Drosophila parasitoids. Adv Parasitol 70:3–44
Foray V, Gibert P, Desouhant E (2011) Differential thermal performance curves in response to different habitats in the parasitoid Venturia canescens. Naturwissenschaften 98:683–691
Geister TL, Lorenz MW, Hoffmann KH, Fischer K (2009) Energetics of embryonic development: effects of temperature on egg and hatchling composition in a butterfly. J Comp Phys B 179:87–98
Gillooly JF, Brown JH, West GB, Savage VM, Charnov EL (2001) Effects of size and temperature on metabolic rate. Science 293:2248–2251
Godfray HJC (1994) Parasitoids behavioral and evolutionary ecology. Princeton University Press, Princeton
Hance T, van Baaren J, Vernon P, Boivin G (2007) Impact of extreme temperatures on parasitoids in a climate change perspective. Annu Rev Entom 52:107–126
Harvey JA (2005) Factors affecting the evolution of development strategies in parasitoid wasps: the importance of functional constraints and incorporating complexity. Entomol Exp Appl 117:1–13
Harvey JA (2008) Comparing and contrasting development and reproductive strategies in the pupal hyperparasitoids Lysibia nana and Gelis agilis (Hymenoptera: Ichneumonidae). Evol Ecol 22:153–166
Harvey JA, Witjes LMA (2005) Comparing and contrasting life history and development strategies in the pupal hyperparasitoids Lysibia nana and Gelis agilis (Hymenoptera: Ichneumonidae). Appl Entomol Zool 40:309–316
Harvey JA, Bezemer TM, Gols R, Nakamatsu Y, Tanaka T (2008) Comparing the physiological effects and function of larval feeding in closely-related endoparasitoids (Braconidae: Microgastrinae). Physiol Entomol 33:217–225
Harvey JA, Poelman EH, Tanaka T (2013) Intrinsic inter- and intraspecific competition in parasitoid wasps. Annu Rev Entom 58:333–351
Hochachka PW, Somero GN (2002) Biochemical adaptation: mechanism and process in physiological evolution. Oxford University Press, Oxford
IPCC (2007) Working group I report, climate change 2007: “the physical science basis”. The fourth assessment report of the intergovernmental panel on climate change, Paris
Irz P, Michonneau F, Oberdorff T, Whittier TR, Lamouroux N, Mouillot D, Argillier C (2007) Fish community comparisons along environmental gradients in lakes of France and northeast USA. Glob Ecol Biogeogr 16:350–366
Jervis MA, Heimpel GE, Ferns PN, Harvey JA, Kidd NAC (2001) Life-history strategies in parasitoid wasps: a comparative analysis of “ovigeny”. J Anim Ecol 70:442–458
Jervis MA, Ellers J, Harvey JA (2008) Resource acquisition, allocation, and utilization in parasitoid reproductive strategies. Annu Rev Entomol 53:361–385
Jervis MA, Moe A, Heimpel GE (2012) The evolution of parasitoid fecundity: a paradigm under scrutiny. Ecol Lett 15:357–364
Laliberté E, Shipley B (2011) FD: measuring functional diversity from multiple traits, and other tools for functional ecology. R Package 10–11
Le Lann C, Roux O, Serain N, van Alphen JJM, Vernon P, van Baaren J (2011a) Thermal tolerance of sympatric hymenopteran parasitoid species: does it match their seasonal activities? Physiol Entomol 36:21–28
Le Lann C, Wardziak T, van Baaren J, van Alphen JJM (2011b) Plasticity in metabolic rates and life history traits affects foraging behaviour in a parasitic wasp. Funct Ecol 25:641–651
Le Lann C, Visser B, van Baaren J, van Alphen JJM, Ellers J (2012) Comparing resource exploitation and allocation of two closely related aphid parasitoids sharing the same host. Evol Ecol 26:79–94
Li B, Mills N (2004) The influence of temperature on size as an indicator of host quality for the development of a solitary koinobiont parasitoid. Entomol Exp Appl 110:249–256
Luo LG, Ding GH, Ji X (2010) Income breeding and temperature-induced plasticity in reproductive traits in lizards. J Exp Biol 213:2073–2078
MacArthur RH, Levins R (1967) The limiting similarity convergence and divergence of coexisting species. Am Nat 101:377–385
Mayhew PJ, Blackburn TM (1999) Does parasitoid development mode organize life-history traits in the parasitoid Hymenoptera? J Anim Ecol 68:906–916
Mitchell SE, Halves J, Lampert W (2004) Coexistence of similar genotypes of Daphnia magna in intermittent populations: response to thermal stress. Oikos 106:469–478
Nealis VG, Jones RE, Wellington WG (1984) Temperature and development in host–parasite relationship. Oecologia 61:224–229
Pavoine S, Bonsall M (2011) Measuring biodiversity to explain community assembly: a unified approach. Biol Rev 86:792–812
Pelosse P, Bernstein C, Desouhant E (2007) Differential energy allocation as an adaptation to different habitats in the parasitic wasp Venturia canescens. Evol Ecol 21:669–685
Petchey OL, Beckerman AP, Riede JO, Warren PH (2008) Size, foraging, and food web structure. Proc Natl Acad Sci USA 105:4191–4196
Price PW (1972) Parasitoids utilizing the same host: adaptive nature of differences in size and form. Ecology 53:190–195
Price PW (1973) Reproductive strategies in parasitoid wasps. Am Nat 107:684–693
Price PW (1974) Strategies for egg production. Evolution 28:76–84
Qu YF, Li H, Gao JF, Ji X (2011) Embryonic thermosensitivity and hatchling morphology differ between two coexisting lizards. Acta Oecol 37:375–380
R Development Core Team (2010) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria ISBN 3–900051–07–0 [WWW document]. Available from http://wwwR-projectorg [Accessed Nov 2010]
Reale D, McAdam AG, Boutin S, Berteaux D (2003) Genetic and plastic responses of a northern mammal to climate change. Proc R Soc Lond B 270:591–596
Romani R, Isidoro N, Bin F, Vinson SB (2002) Host recognition in the pupal parasitoid Trichopria drosophilae: a morpho-functional approach. Entomol Exp Appl 105:119–128
Tilman D (1982) Ressource competition and community structure. Princeton University Press, Princeton
Traynor RE, Mayhew PJ (2005) A comparative study of body size and clutch size across the parasitoid Hymenoptera. Oïkos 109:305–316
Valladares F, Wright SJ, Lasso E, Kitajima K, Pearcy RW (2000) Plastic phenotypic response to light of 16 congeneric shrubs from a Panamanian rainforest. Ecology 81:1925–1936
van Baaren J, Le Lann C, van Alphen JJM (2010) Consequences of climate change on aphid-based multi-trophic systems. In: Kindlmann P, Dixon AFG, Michaud JP (eds) Aphid biodiversity under environmental change: Patterns and processes. Springer, Dordrecht, pp 55–68
van Strien-van Liempt WTFH (1983) The competition between Asobara Tabida Nees Von Esenbeck, 1834 and Leptopilina Heterotoma (Thomson, 1862) in multiparasitized hosts. Neth J Zool 33:125–163
Villéger S, Mason NWH, Mouillot D (2008) New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology 89:2290–2301
Vinson SB (1988) Physiological studies of parasitoids reveal new approaches to the biological control of insect pests. ISI Atlas of Science 1:25–32
Violle C, Enquist BJ, McGill BJ, Jiang L, Albert CH, Hulshof C, Jung V, Messier J (2012) The return of the variance: intraspecific variability in community ecology. Trends Ecol Evol 27:244–252
Visser B, Ellers J (2008) Lack of lipogenesis in parasitoids: a review of physiological mechanisms and evolutionary implications. J Insect Physiol 54:1315–1322
Visser B, Le Lann C, den Blanken FJ, Harvey JA, van Alphen JJM, Ellers J (2010) Loss of lipid synthesis as an evolutionary consequence of a parasitic lifestyle. Proc Natl Acad Sci USA 107:8677–8682
Visser B, Roelofs D, Hahn DA, Teal PEA, Mariën J, Ellers J (2012) Transcriptional changes associated with lack of lipid synthesis in parasitoids. Gen Biol Evol 4:864–874
Wu GM, Barrette M, Boivin G, Brodeur J, Giraldeau LA, Hance T (2011) Temperature influences the handling efficiency of an aphid parasitoid through body size-mediated effects. Environ Entomol 40:737–742
Acknowledgments
We thank André T.C. Dias for helpful advice on earlier drafts of this manuscript. This research was supported by grants to C.L.L. from the Ministère de l’Enseignement Supérieur et de la Recherche and Rennes Métropole (France), by the COMPAREVOL program (Marie Curie Excellence Chair, http://comparevol.univ-rennes1.fr/) and by the ECOCLIM program founded by the Région Bretagne (France). B.V. was funded by Netherlands Organisation for Scientific Research (NWO) ALW Grant 816.01.013. The authors declare that they have no conflict of interest. The experiments comply with the current laws of the countries (The Netherlands and France) in which they were performed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Thomas S. Hoffmeister.
Rights and permissions
About this article
Cite this article
Le Lann, C., Visser, B., Mériaux, M. et al. Rising temperature reduces divergence in resource use strategies in coexisting parasitoid species. Oecologia 174, 967–977 (2014). https://doi.org/10.1007/s00442-013-2810-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-013-2810-9