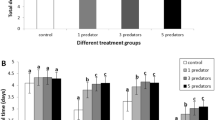
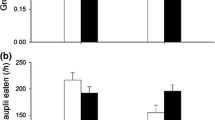
Abstract
Prey organisms are expected to use different short- and long-term responses to predation risk to avoid excessive costs. Contrasting both types of responses is important to identify chronic stress responses and possible compensatory mechanisms in order to better understand the full impact of predators on prey life history and population dynamics. Using larvae of the damselfly Enallagma cyathigerum, we contrasted the effects of short- and long-term predation risk, with special focus on consequences for body stoichiometry. Under short-term predation risk, larvae reduced growth rate, which was associated with a reduced food intake, increased metabolic rate and reduced glucose content. Under long-term predation risk, larvae showed chronic predator stress as indicated by persistent increases in metabolic rate and reduced food intake. Despite this, larvae were able to compensate for the short-term growth reduction under long-term predation risk by relying on physiological compensatory mechanisms, including reduced energy storage. Only under long-term predation risk did we observe an increase in body C:N ratio, as predicted under the general stress paradigm (GSP). Although this was caused by a predator-induced decrease in N content, there was no associated increase in C content. These stoichiometric changes could not be explained by GSP responses because, under chronic predation risk, there was no decrease in N-rich proteins or increase in C-rich fat and sugars; instead glycogen decreased. Our results highlight the importance of compensatory mechanisms and the value of explicitly integrating physiological mechanisms to obtain insights into the temporal dynamics of non-consumptive effects, including effects on body stoichiometry.



Similar content being viewed by others
References
Abrams PA, Rowe L (1996) The effects of predation on the age and size of maturity of prey. Evolution 50:1052–1061
Adamo SA, Baker JL (2011) Conserved features of chronic stress across phyla: the effects of long-term stress on behavior and the concentration of the neurohormone octopamine in the cricket, Gryllus texensis. Horm Behav 60:478–483. doi:10.1016/j.yhbeh.2011.07.015
Benard MF (2004) Predator-induced phenotypic plasticity in organisms with complex life histories. Annu Rev Ecol Evol Syst 35:651–673. doi:10.2307/annurev.ecolsys.35.021004.1124260
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B 57:289–300
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Phys 37:911–917
Boonstra R (2013) Reality as the leading cause of stress: rethinking the impact of chronic stress in nature. Funct Ecol 27:11–23. doi:10.1111/1365-2435.12008
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein, utilizing the principle of protein-dye landing. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Costello DM, Michel MJ (2013) Predator-induced defences in tadpoles confound body stoichiometry predictions of the general stress paradigm. Ecology 94:2229–2236
Crawley MJ (2007) The R book. Wiley, Chichester
Creel S, Christianson D (2008) Relationships between direct predation and risk effects. Trends Ecol Evol 23:194–201. doi:10.1016/j.tree.2007.12.004
Creel S, Winnie JA Jr, Christianson D (2009) Glucocorticoid stress hormones and the effect of predation risk on elk reproduction. Proc Natl Acad Sci USA 106:2388–12393. doi:10.1073/pnas.0902235106
Culler LE, McPeek MA, Ayres MP (2014) Predation risk shaping thermal physiology of a predaceous damselfly. Oecologia 176:653–660. doi:10.1007/s00442-014-3058-8
Dahl J, Peckarsky B (2002) Induced morphological defences in the wild: predator effect on a mayfly, Drunella coloradensis. Ecology 83:1620–1634
Dalton CM, Flecker AS (2014) Metabolic stoichiometry and ecology of fear in Trinidadian guppies: consequences for life histories and stream ecosystems. Oecologia 176:691–701. doi:10.1007/s00442-014-3084-6
De Block M, Stoks R (2004) Cannibalism-mediated life history plasticity to combined time and food stress. Oikos 106:587–597. doi:10.1111/j.0030-1299.2004.12974.x
De Coen WM, Janssen CR (2003) The missing biomarker: relationships between effects on the cellular energy allocation biomarker of toxicant-stressed Daphnia magna and corresponding population characteristics. Environ Toxicol Chem 22:1632–1641. doi:10.1002/etc.5620220727
Dmitriew CM (2011) The evolution of growth trajectories: what limits growth rate? Biol Rev 86:97–116. doi:10.1111/j.1469-185X.2010.00136.x
Hawlena D, Schmitz OJ (2010a) Physiological stress as a fundamental mechanism linking predation to ecosystem functioning. Am Nat 176:537–556. doi:10.1086/656495
Hawlena D, Schmitz OJ (2010b) Herbivore physiological response to predation risk and implications for ecosystem nutrient dynamics. Proc Natl Acad Sci USA 107:15503–15507. doi:10.1073/pnas.1009300107
Hawlena D, Kress H, Dufresne ER, Schmitz OJ (2011) Grasshoppers alter jumping biomechanics to enhance escape performance under chronic predation risk of spider predation. Funct Ecol 25:279–288. doi:10.1111/j.1365-2435.2010.01767.x
Hawlena D, Strickland MS, Bradford MA, Schmitz OJ (2012) Fear of predation slows litter decomposition. Science 336:1434–1438. doi:10.1126/science.1220097
Hik DS, McColl CJ, Boonstra R (2001) Why are Arctic ground squirrels more stressed in the boreal forest than in the alpine meadows? Ecoscience 8:275–288
Janssens L, Stoks R (2013) Synergistic effects between pesticide stress and predator cues: conflicting results from life history and physiology in the damselfly Enallagma cyathigerum. Aquat Toxicol 132–133:92–99. doi:10.1016/j.aquatox.2013.02.003
Janssens L, Van Dievel M, Stoks R (in press) Warming reinforces non-consumptive predator effects on prey growth, physiology and body stoichiometry. Ecology
Lima SL (1998) Stress and decision making under the risk of predation: recent developments from behavior, reproductive, and ecological perspectives. Adv Stud Behav 27:215–290
Lima SL, Bednekoff PA (1999) Temporal variation in danger drives antipredator behavior: the predation risk allocation hypothesis. Am Nat 153:649–659. doi:10.1086/303202
Manoj K, Senthamarai Kannan K (2013) Comparison of methods for detecting outliers. Int J Sci Eng Res 4:709–714
Manzur T, Vidal F, Pantoja JF, Fernández M, Navarrete SA (2014) Behavioural and physiological responses of limpet prey to a seastar predator and their transmission to basal trophic levels. J Anim Ecol 83:923–933. doi:10.1111/1365-2656.12199
McPeek MA (2004) The growth/predation risk trade-off: so what is the mechanism? Am Nat 163:E88–E111. doi:10.1086/382755
McPeek MA, Grace M, Richardson JML (2001) Physiological and behavioral responses to predators shape the growth/predation risk trade-off in damselflies. Ecology 82:1535–1545
Mortensen L, Richardson JML (2008) Effects of chemical cues on foraging in damselfly larvae, Enallagma antennatum. J Insect Behav 21:285–295. doi:10.1007/s10905-008-9125-4
Peacor SD, Pangle KL, Schiesari L, Werner EE (2012) Scaling-up-antipredator phenotypic responses of prey: impacts over multiple generations in a complex aquatic community. Proc R Soc B 279:122–128. doi:10.1098/rspb.2011.0606
Preisser EL (2009) The physiology of predator stress in free-ranging prey. J Anim Ecol 78:1103–1105. doi:10.1111/j.1365-2656.2009.01602.x
Preisser EL, Bolnick DI, Benard MF (2005) Scared to death? The effects of intimidation and consumption in predator-prey interactions. Ecology 86:501–509
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, New York
Sapolsky RM (2002) Endocrinology of the stress-response. In: Becker JB, Breedlove SM, Crews D, McCarthy MM (eds) Behavioural endocrinology. MIT, Cambridge, pp 409–450
Sheriff MJ, Krebs CJ, Boonstra R (2011) From processes to pattern: how fluctuating predation risk impacts the stress axis of snowshoe hares during the 10-year cycle. Oecologia 166:593–605. doi:10.1007/s00442-011-1907-2
Slos S, Stoks R (2008) Predation risk induces stress proteins and reduces antioxidant defense. Funct Ecol 22:637–642. doi:10.1111/j.1365-2435.2008.01424.x
Slos S, De Meester L, Stoks R (2009) Food level and sex shape predator-induced physiological stress: immune defence and antioxidant defence. Oecologia 161:461–467. doi:10.1007/s00442-009-1401-2
Sörensen JG, Kristensen TN, Loeschcke V (2003) The evolutionary and ecological role of heat shock proteins. Ecol Lett 6:1025–1037. doi:10.1046/j.1461-0248.2003.00528.x
Steiner UK, Van Buskirk J (2009) Predator-induced changes in metabolism cannot explain the growth/predation risk tradeoff. PLoS One 4:e6160. doi:10.1371/journal.pone.0006160
Sterner RW, Elser JJ (2002) Ecological stoichiometry: the biology of elements from molecules to the biosphere. Princeton University Press, Princeton
Stoks R, De Block M (2011) Rapid growth reduces cold resistance: evidence from latitudinal variation in growth rate, cold resistance and stress proteins. PLoS One 6:e16935. doi:10.1371/journal.pone.0016935
Stoks R, McPeek MA (2003) Antipredator behavior and physiology determine Lestes species turnover along the pond-permanence gradient. Ecology 84:3327–3338
Stoks R, McPeek MA, Mitchell JL (2003) Evolution of prey behavior in response to changes in predation regime: damselflies in fish and dragonfly lakes. Evolution 57:574–585. doi:10.1111/j.0014-3820.2003.tb01549.x
Stoks R, Nystrom JL, May ML, McPeek MA (2005a) Parallel evolution in ecological and reproductive traits to produce cryptic damselfly species across the Holarctic. Evolution 59:1976–1988. doi:10.1111/j.0014-3820.2005.tb01067.x
Stoks R, De Block M, McPeek MA (2005b) Alternative growth and energy storage responses to mortality threats in damselflies. Ecol Lett 8:1307–1316. doi:10.1111/j.1461-0248.2005.00840.x
Stoks R, De Block M, Van De Meutter F, Johansson F (2005c) Predation cost of rapid growth: behavioural coupling and physiological decoupling. J Anim Ecol 74:708–715. doi:10.1111/j.1365-2656.2005.00969.x
Stoks R, De Block M, Slos S, Van Doorslaer W, Rolff J (2006a) Time constraints mediate predator-induced plasticity in immune function, condition, and life history. Ecology 87:809–815
Stoks R, De Block M, McPeek MA (2006b) Physiological costs of compensatory growth in a damselfly. Ecology 87:1566–1574
Strobbe F, McPeek MA, De Block M, Stoks R (2010) Survival selection imposed by predation on a physiological trait underlying escape speed. Funct Ecol 24:1306–1312. doi:10.1111/j.1365-2435.2010.01752.x
Thaler JS, McArt SH, Kaplan I (2012) Compensatory mechanisms for ameliorating the fundamental trade-off between predator avoidance and foraging. Proc Natl Acad Sci USA 109:12075–12080. doi:10.1073/pnas.1208070109
Thaler JS, Contreras J, Davidowitz G (2014) Effects of predation risk and plant resistance on Manduca sexta caterpillar feeding behaviour and physiology. Ecol Entomol 39:210–216. doi:10.1111/een.12086
Trussell GC, Ewanchuk PJ, Matassa CM (2006) The fear of being eaten reduces energy transfer in a simple food chain. Ecology 87:2979–2984
Trussell GC, Ewanchuk PJ, Matassa CM (2008) Resource identity modifies the influence of predation risk on ecosystem function. Ecology 89:2798–2807
Werner EE, Anholt BR (1993) Ecological consequences of trade-off between growth and mortality-rates mediated by foraging activity. Am Nat 142:242–272
Zanette LY, Clincy M, Suraci JP (2014) Diagnosing predation risk effects on demography: can measuring physiology provide the means? Oecologia 176:637–651. doi:10.1007/s00442-014-3057-9
Acknowledgments
The authors declare that they have no conflict of interest. We appreciate the constructive feedback of Jill Lancaster and two anonymous reviewers that considerably improved our manuscript. L. J. is a postdoctoral fellow of FWO-Flanders and benefited from a PDM fellowship of the KU Leuven. Financial support came from the Belspo project SPEEDY, KU Leuven Excellence Center Financing PF/2010/07 and FWO research Grant G.0943.15.
Author contribution statement
MVD, LJ and RS conceived and designed the experiment. MVD and LJ performed the experiment. MVD, LJ and RS analysed the data. LJ, MVD and RS wrote the manuscript. MVD and LJ are joint first author.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jill Lancaster.
Rights and permissions
About this article
Cite this article
Van Dievel, M., Janssens, L. & Stoks, R. Short- and long-term behavioural, physiological and stoichiometric responses to predation risk indicate chronic stress and compensatory mechanisms. Oecologia 181, 347–357 (2016). https://doi.org/10.1007/s00442-015-3440-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-015-3440-1