Abstract
Background
Optical biopsy methods such as probe-based confocal laser endomicroscopy (pCLE) provide useful intraoperative real-time information, especially during minimally invasive surgery with flexible endoscopic or robotic platforms. By translating the probe at constant pressure across the target tissue, undistorted “mosaics” can be produced. However, this poses ergonomic challenges with a conventional flexible endoscope.
Methods
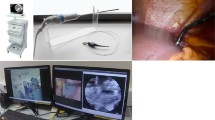
A 100 μm confocal depth pCLE probe was integrated into a previously described seven degrees-of-freedom articulated endoscopic robot. After estimating the average workspace created by a female pneumoperitoneum, the accessibility of the peritoneal cavity by the device for robot-assisted pCLE peritoneoscopy was calculated. To demonstrate its in vivo feasibility, the robot was inserted transvaginally in a pig, under laparoscopic vision. Optical biopsy was performed of several targets within the peritoneal cavity.
Results
The workspace analysis calculated that 88 % of the surface of an estimated average female pneumoperitoneum could be contacted by the probe using the robot transvaginally. In vivo, the robot was manoeuvred to provide views of all abdominal and pelvic organs. At each target there was robotic acquisition of still pCLE images, and slowly translating images for the construction of increased field-of-view mosaics up to 2 mm in length. Optical biopsies took 1–2 min per target, and at 3.5 μm lateral resolution, the mosaic images showed characteristic features of anterior abdominal wall, liver, and spleen.
Conclusion
In the porcine model, the robotically actuated method of performing peritoneoscopy and pCLE mosaicked optical biopsy is safe and provides a consistent means of acquiring near-histological grade images of submesothelial tissue. Clinical translation is likely to provide sufficient accessibility of the peritoneal cavity.




Similar content being viewed by others
References
Sutton C, Ewen S, Whitelaw N, Haines P (1994) Prospective, randomized, double-blind, controlled trial of laser laparoscopy in the treatment of pelvic pain associated with minimal, mild, and moderate endometriosis. Fertil Steril 62:696
Swank DJ, Swank-Bordewijk SCG, Hop WCJ, van Erp WFM, Janssen IMC, Bonjer HJ, Jeekel J (2003) Laparoscopic adhesiolysis in patients with chronic abdominal pain: a blinded randomised controlled multi-centre trial. Lancet 361:1247–1251
Brandt CP, Priebe PP, Eckhauser ML (1993) Diagnostic laparoscopy in the intensive care patient. Surg Endosc 7:168–172
Hariharan D, Constantinides VA, Froeling FE, Tekkis PP, Kocher HM (2010) The role of laparoscopy and laparoscopic ultrasound in the preoperative staging of pancreatico-biliary cancers—a meta-analysis. Eur J Surg Oncol 36:941–948
Hazey J, Narula V, Renton D, Reavis K, Paul C, Hinshaw K, Muscarella P, Ellison E, Melvin W (2008) Natural-orifice transgastric endoscopic peritoneoscopy in humans: initial clinical trial. Surg Endosc 22:16–20
Voermans RP, Sheppard B, van Berge Henegouwen MI, Fockens P, Faigel DO (2009) Comparison of transgastric NOTES and laparoscopic peritoneoscopy for detection of peritoneal metastases. Ann Surg 250:255–259
Steele K, Schweitzer MA, Lyn-Sue J, Kantsevoy SV (2008) Flexible transgastric peritoneoscopy and liver biopsy: a feasibility study in human beings (with videos). Gastrointest Endosc 68:61–66
Voermans RP, van Berge Henegouwen MI, Bemelman WA, Fockens P (2009) Feasibility of transgastric and transcolonic natural orifice transluminal endoscopic surgery peritoneoscopy combined with intraperitoneal EUS. Gastrointest Endosc 69:e61–e67
Mieog J, Hutteman M, van der Vorst J, Kuppen P, Que I, Dijkstra J, Kaijzel E, Prins F, Lowik C, Smit V, van de Velde C, Vahrmeijer A (2011) Image-guided tumor resection using real-time near-infrared fluorescence in a syngeneic rat model of primary breast cancer. Breast Cancer Res Treat 128:679–689
Cahill RA, Asakuma M, Trunzo J, Schomisch S, Wiese D, Saha S, Dallemagne B, Marks J, Marescaux J (2010) Intraperitoneal virtual biopsy by fibered optical coherence tomography (OCT) at natural orifice transluminal endoscopic surgery (NOTES). J Gastrointest Surg 14:732–738
Rao G, Mansard M, Rebala P, Sekaran A, Reddy D (2009) Endocytoscopy assisted laparoscopic intraoperative diagnosis of disseminated malignancy. Surg Endosc 23:2395–2396
Pohl H, Rosch T, Vieth M, Koch M, Becker V, Anders M, Khalifa AC, Meining A (2008) Miniprobe confocal laser microscopy for the detection of invisible neoplasia in patients with Barrett’s oesophagus. Gut 57:1648–1653
Newton RC, Kemp SV, Yang G-Z, Darzi A, Sheppard MN, Shah PL (2011) Tracheobronchial Amyloidosis and Confocal Endomicroscopy. Respiration 82(2):209–211
Newton R, Kemp S, Zoumot Z, Yang GZ, Darzi A, Shah PL (2010) An unusual case of haemoptysis. Thorax 65:309, 353
Newton RC, Kemp SV, Yang G-Z, Ellson D, Darzi A, Shah P (2012) Imaging parenchymal lung diseases with confocal endomicroscopy. Respiratory Medicine 106(1):127–137
Newton R, Kemp S, Shah P, Elson D, Darzi A, Shibuya K, Mulgrew S, Yang GZ (2011) Progress toward optical biopsy: bringing the microscope to the patient. Lung 189:111–119
Becker V, Vercauteren T, von Weyhern CH, Prinz C, Schmid RM, Meining A (2007) High-resolution miniprobe-based confocal microscopy in combination with video mosaicing (with video). Gastrointest Endosc 66:1001–1007
Buchner AM, Shahid MW, Heckman MG, Krishna M, Ghabril M, Hasan M, Crook JE, Gomez V, Raimondo M, Woodward T, Wolfsen HC, Wallace MB (2009) Comparison of probe-based confocal laser endomicroscopy with virtual chromoendoscopy for classification of colon polyps. Gastroenterology 138:834–842
Ji R, Zuo XL, Li CQ, Zhou CJ, Li YQ (2011) Confocal endomicroscopy for in vivo prediction of completeness after endoscopic mucosal resection. Surg Endosc 25:1933–1938
von Delius S, Feussner H, Wilhelm D, Karagianni A, Henke J, Schmid RM, Meining A (2007) Transgastric in vivo histology in the peritoneal cavity using miniprobe-based confocal fluorescence microscopy in an acute porcine model. Endoscopy 39:407–411
Becker V, Wallace MB, Fockens P, von Delius S, Woodward TA, Raimondo M, Voermans RP, Meining A (2010) Needle-based confocal endomicroscopy for in vivo histology of intra-abdominal organs: first results in a porcine model (with videos). Gastrointest Endosc 71:1260–1266
Goetz M, Deris I, Vieth M, Murr E, Hoffman A, Delaney P, Galle PR, Neurath MF, Kiesslich R (2010) Near-infrared confocal imaging during mini-laparoscopy: a novel rigid endomicroscope with increased imaging plane depth. J Hepatol 53:84–90
Nguyen NQ, Biankin AV, Leong RW, Chang DK, Cosman PH, Delaney P, Kench JG, Merrett ND (2009) Real time intraoperative confocal laser microscopy-guided surgery. Ann Surg 249:735–737
Latt WT, Newton R, Visentini-Scarzanella M, Payne CJ, Noonan DP, Shang J, Yang GZ (2011) A hand-held instrument to maintain steady tissue contact during probe-based confocal laser endomicroscopy. IEEE Trans Biomed Eng 58:2694–2703
Ti Y, Lin WC (2008) Effects of probe contact pressure on in vivo optical spectroscopy. Opt Express 16:4250–4262
Newton RC, Noonan D, Payne C, Andreyev J, Di Marco A, Scarzanella MV, Darzi A, Yang GZ (2011) Probe tip contact force and bowel distension affect crypt morphology during confocal endomicroscopy. Gut 60:A12–A13
Clark J, Sodergren M, Noonan D, Shang J, Payne C, James D, Athanasiou T, Teare J, Darzi A, Yang GZ (2010) A novel articulated robotic laparoscope for single incision and natural orifice surgical applications: preliminary usability trials in a live porcine model. Surg Endosc 24(1):690
Shang J, Noonan D, Payne C, Clark J, Sodergren M, Darzi A, Yang GZ (2011) An articulated universal joint based flexible access robot for minimally invasive surgery. International conference on robotics and automation, IEEE, Shanghai, pp 1147–1152
Noonan DP, Payne CJ, Shang J, Sauvage V, Newton R, Elson D, Darzi A, Yang GZ (2010) Force adaptive multi-spectral imaging with an articulated robotic endoscope. Med Image Comput Comput Assist Interv 13:245–252
Sánchez-Margallo F, Moyano-Cuevas J, Latorre R, Maestre J, Correa L, Pagador J, Sánchez-Peralta L, Sánchez-Margallo J, Usón-Gargallo J (2011) Anatomical changes due to pneumoperitoneum analyzed by MRI: an experimental study in pigs. Surg Radiol Anat 33:389–396
Andersson LE, Baath M, Thorne A, Aspelin P, Odeberg-Wernerman S (2005) Effect of carbon dioxide pneumoperitoneum on development of atelectasis during anesthesia, examined by spiral computed tomography. Anesthesiology 102:293–299
Shamiyeh A, Glaser K, Kratochwill H, Hörmandinger K, Fellner F, Wayand W, Zehetner J (2009) Lifting of the umbilicus for the installation of pneumoperitoneum with the Veress needle increases the distance to the retroperitoneal and intraperitoneal structures. Surg Endosc 23:313–317
Becker V, van den Broek FJ, Buchner AM, Dekker E, Wallace MB, von Delius S, Schneider A, Schmid RM, Meining A (2011) Optimal fluorescein dose for intravenous application in miniprobe-based confocal laser scanning microscopy in pigs. J Biophotonics 4:108–113
Becker V, von Delius S, Bajbouj M, Karagianni A, Schmid RM, Meining A (2008) Intravenous application of fluorescein for confocal laser scanning microscopy: evaluation of contrast dynamics and image quality with increasing injection-to-imaging time. Gastrointest Endosc 68:319–323
Hollenstein M, Nava A, Valtorta D, Snedeker J, Mazza E, (2006) Mechanical characterization of the liver capsule and parenchyma. In: Biomedical simulation. Lecture notes in computer science, vol 4072. Springer, Berlin, pp 150–158
Kuntz E, Kuntz HD (2006) Hepatology: principles and practice: history, morphology, biochemistry, diagnostics, clinic, therapy, 2nd edn. Springer, Heidelberg
Tehver J, Grahame T (1931) The capsule and trabeculae of the spleens of domestic mammals. J Anat 65:473–481
De Palma GD, Staibano S, Siciliano S, Persico M, Masone S, Maione F, Siano M, Mascolo M, Esposito D, Salvatori F, Persico G (2010) In vivo characterisation of superficial colorectal neoplastic lesions with high-resolution probe-based confocal laser endomicroscopy in combination with video-mosaicing: a feasibility study to enhance routine endoscopy. Dig Liver Dis 42:791–797
Ignat M, Aprahamian M, Lindner V, Altmeyer A, Perretta S, Dallemagne B, Mutter D, Marescaux J (2009) Feasibility and reliability of pancreatic cancer staging using fiberoptic confocal fluorescence microscopy in rats. Gastroenterology 137(1584–1592):e1581
Hsiung PL, Hardy J, Friedland S, Soetikno R, Du CB, Wu AP, Sahbaie P, Crawford JM, Lowe AW, Contag CH, Wang TD (2008) Detection of colonic dysplasia in vivo using a targeted heptapeptide and confocal microendoscopy. Nat Med 14:454–458
von Delius S, Schorn A, Grimm M, Schneider A, Wilhelm D, Schuster T, Stangassinger M, Feussner H, Schmid RM, Meining A (2011) Natural-orifice transluminal endoscopic surgery: low-pressure pneumoperitoneum is sufficient and is associated with an improved cardiopulmonary response (PressurePig Study). Endoscopy 43:808–815
Sugrue M (2005) Abdominal compartment syndrome. Curr Opin Crit Care 11:333–338
Summers AM, Clancy MJ, Syed F, Harwood N, Brenchley PE, Augustine T, Riad H, Hutchison AJ, Taylor P, Pearson R, Gokal R (2005) Single-center experience of encapsulating peritoneal sclerosis in patients on peritoneal dialysis for end-stage renal failure. Kidney Int 68:2381–2388
Williams JD, Craig KJ, Topley N, Von Ruhland C, Fallon M, Newman GR, Mackenzie RK, Williams GT (2002) Morphologic changes in the peritoneal membrane of patients with renal disease. J Am Soc Nephrol 13:470–479
Mettang T, Krumme B, Wilhelm A, Duschka L (2010) Endoscopic transluminal insertion of a peritoneal dialysis catheter. Perit Dial Int 30:63–65
Goetz M, Vieth M, Kanzler S, Galle PR, Delaney P, Neurath MF, Kiesslich R (2008) In vivo confocal laser laparoscopy allows real time subsurface microscopy in animal models of liver disease. J Hepatol 48:91–97
Henrikson RC, Kaye GI, Mazurkiewicz JE (1997) Histology, 1st edn. Williams & Wilkins, Baltimore
Acknowledgments
This work was supported by the Wellcome Trust [(GR083689)], and an RT-ISIS grant from the EPSRC. We are grateful to the staff at the Department of Surgical Research, Northwick Park Institute for Medical Research, and also to Su-lin Lee and Ka Wai Kwok for helping to create the mesh.
Disclosures
Mr. Newton, Dr. Noonan, Dr. Vitiello, Mr. Clark, Mr. Payne, Dr. Shang, Mr. Sodergren, Professor Darzi, and Professor Yang have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Newton, R.C., Noonan, D.P., Vitiello, V. et al. Robot-assisted transvaginal peritoneoscopy using confocal endomicroscopy: a feasibility study in a porcine model. Surg Endosc 26, 2532–2540 (2012). https://doi.org/10.1007/s00464-012-2228-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-012-2228-1