Abstract
Non-modified molybdenum disulfide (MoS2) is known to adsorb heavy metal ions. However, in case of very small particle sizes and high dispersibility, ordinary centrifugation and filtration fail to separate MoS2 from aqueous media, and this limits its applicability to solid-phase extraction. To overcome this shortcoming, the authors have prepared a nanocomposite consisting of MoS2 and sheets of reduced graphene oxide (r–GO). The material was obtained by a hydrothermal reaction between sodium molybdate and L-cysteine in the presence of GO sheets. The results of characterizations show the MoS2 nanosheets to be well dispersed on the surface of r-GO sheets. The nanocomposite was applied to the preconcentration of Pb(II) and Ni(II) ions as representative examples of heavy metal ions. The adsorption capacities are 322 and 294 mg.g−1 for Pb(II) and Ni(II), respectively. The ions were quantified by atomic absorption spectrometry, and the respective detection limits are 0.71 and 0.21 μg⋅L−1, respectively. The relative standard deviations are ±2.2 % for both ions. The method was successfully applied to the preconcentration of the analytes in (spiked) urine, saliva and water samples.

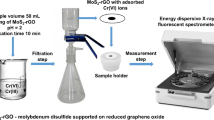
Schematic illustration of the preparation of nanocomposite consisting of MoS2 and sheets of reduced graphene oxide (MoS2–rGO).


Similar content being viewed by others
References
Maijenburg AW, Regis M, Hattori AN, Tanaka H, Choi KS, Elshof JE (2014) MoS2 nanocube structures as catalysts for electrochemical H2 evolution from acidic aqueous solutions. ACS Appl Mater Interfaces 6:2003–2010
Pumera M, Loo AH (2014) Layered transition-metal dichalcogenides (MoS2 and WS2) for sensing and biosensing. TrAC Trend Analyt Chem 61:49–53
Parlak O, Incel A, Uzun L, Turner, AP, Tiwari, A (2016) Structuring Au nanoparticles on two-dimensional MoS2 nanosheets for electrochemical glucose biosensors. Biosens Bioelectronic. doi:10.1016/j.bios.2016.03.024
Tenne R (2002) Inorganic nanotubes and fullerene-like materials. Chem A Eur J 8:5296–5304
Radisavljevic B, Radenovic A, Brivio J, Giacometti V, Kis A (2011) Single-layer MoS2 transistors. Nat Nanotechnol 6:147–150
Xiao J, Choi D, Cosimbescu L, Koech P, Liu J, Lemmon JP (2010) Exfoliated MoS2 nanocomposite as an anode material for lithium ion batteries. Chem Mater 22:4522–4524
Li H, Lu G, Yin Z, He Q, Li H, Zhang Q, Zhang H (2012) Optical identification of single-and few-layer MoS2 sheets. Small 8:682–686
Komarneni M, Sand A, Burghaus U (2009) Adsorption of thiophene on inorganic MoS2 fullerene-like nanoparticles. Catal Lett 129:66–70
Chao Y, Zhu W, Wu X, Hou F, Xun S, Wu P, Ji H, Xu H, Li H (2014) Application of graphene-like layered molybdenum disulfide and its excellent adsorption behavior for doxycycline antibiotic. Chem Eng J 243:60–67
Song H, You S, Jia X (2015) Synthesis of fungus-like MoS2 nanosheets with ultrafast adsorption capacities toward organic dyes. Appl Phys A 121:541–548
Rao CNR, Biswas K, Subrahmanyam KS, Govindaraj A (2009) Graphene, the new nanocarbon. J Mater Chem 19:2457–2469
Wang Y, Ke X, Zhou X, Li J, Ma J (2016) Graphene for separation and preconcentration of trace amounts of cobalt in water samples prior to flame atomic absorption spectrometry. J Saudi Chem Soc 20:145–152
Wang J, Chen B (2015) Adsorption and coadsorption of organic pollutants and a heavy metal by graphene oxide and reduced graphene materials. Chem Eng J 281:379–388
Yuan G, Wang G, Wang H, Bai J (2016) Half-cell and full-cell investigations of 3D hierarchical MoS2-graphene composite on anode performance in lithium-ion batteries. J Alloys Compd 660:62–72
Yan H, Song P, Zhang S et al (2016) Facile synthesis, characterization and gas sensing performance of ZnO nanoparticles-coated MoS2 nanosheets. J Alloys Compd 662:118–125
Al-Daous MA (2015) Graphene-MoS2 composite: hydrothermal synthesis and catalytic property in hydrodesulfurization of dibenzothiophene. Catal Commun 72:180–184
Ding Y, Zhou Y, Nie W, Chen P (2015) MoS2-GO nanocomposites synthesized via a hydrothermal hydrogel method for solar light photocatalytic degradation of methylene blue. Appl Surf Sci 357:1606–1612
Duran A, Tuzen M, Soylak M (2009) Preconcentration of some trace elements via using multiwalled carbon nanotubes as solid phase extraction adsorbent. J Hazard Mater 169:466–471
Asgharinezhad AA, Ebrahimzadeh H (2015) Coextraction of acidic, basic and amphiprotic pollutants using multiwalled carbon nanotubes-magnetite nanoparticles@ polypyrrole composite. J Chromatogr A 1412:1–11
Ghaedi M, Ahmadi F, Tavakoli Z, Montazerozohori M, Khanmohammadi A, Soylak M (2008) Three modified activated carbons by different ligands for the solid phase extraction of copper and lead. J Hazard Mater 152:1248–1255
Tuzen M, Soylak M (2009) Column solid-phase extraction of nickel and silver in environmental samples prior to their flame atomic absorption spectrometric determinations. J Hazard Mater 164:1428–1432
Özdemir S, Okumus V, Dündar A, Kilinc E (2013) Preconcentration of metal ions using microbacteria. Microchim Acta 180:719–739
Habila MA, ALOthman ZA, El-Toni AM, Labis JP, Xiaomi LZ, Soylak, M (2016) Mercaptobenzothiazole-functionalized magnetic carbon nanospheres of type Fe3O4@SiO2@C for the preconcentration of nickel, copper and lead prior to their determination by ICP-MS. Microchim Acta 1–8
Huang KJ, Wang L, Zhang JZ, Wang LL, Mo YP (2014) One-step preparation of layered molybdenum disulfide/multi-walled carbon nanotube composites for enhanced performance supercapacitor. Energy 67:234–240
Paulchamy B, Arthi G, Lignesh BD (2015) A simple approach to stepwise synthesis of graphene oxide nanomaterial. J Nanomed Nanotechnol 6:1
Wang Z, Chen T, Chen W, Chang K, Ma L, Huang G, Chen D, Lee JY (2013) CTAB-assisted synthesis of single-layer MoS2-graphene composites as anode materials of Li-ion batteries. J Mater Chem A 1:2202–2210
Concha BN, Eugenio C, Carlos MG, Sánchez-Royo JF, Maribel GG (2012) Influence of the pH on the synthesis of reduced graphene oxide under hydrothermal conditions. Nanoscale 4:3977–3982
Pimenta MA, Dresselhaus G, Dresselhaus MS, Cancado LG, Jorio A, Saito R (2007) Studying disorder in graphite-based systems by Raman spectroscopy. Phys Chem Chem Phys 9:1276–1290
Karimi MA, Kafi M (2015) Removal, preconcentration and determination of Ni(II) from different environmental samples using modified magnetite nanoparticles prior to flame atomic absorption spectrometry. Arab J Chem 8:812–820
Tavallali, H and Malekzadeh, H and Karimi, MA, Payehghadr M, Deilamy-Rad, G, Tabandeh M (2014) Chemically modified multiwalled carbon nanotubes as efficient and selective sorbent for separation and preconcentration of trace amount of Co(II), Cd(II), Pb(II) and Pd(II). Arab J Chem. doi:10.1016/j.arabjc.2014.10.034
Kardar ZS, Beyki MH, Shemirani F (2016) Takovite-aluminosilicate@ MnFe2O4 nanocomposite, a novel magnetic adsorbent for efficient preconcentration of lead ions in food samples. Food Chem 209:241–247
Shahamirifard SAR, Ghaedi M, Rahimi MR, Hajati S, Montazerozohori M, Soylak M (2016) Simultaneous extraction and preconcentration of Cu2+, Ni2+ and Zn2+ ions using Ag nanoparticle-loaded activated carbon: Response surface methodology. Advance Powder Technol 27:426–435
Hajiaghababaei L, Ghasemi B, Badiei A, Goldooz H, Ganjali MR, Ziarani GM (2012) Aminobenzenesulfonamide functionalized SBA-15 nanoporous molecular sieve: a new and promising adsorbent for preconcentration of lead and copper ions. J Environ Sci 24:1347–1354
Acknowledgments
Financial support from University of Tehran is gratefully acknowledged. Also, this work was supported by Science and Technology Park of University of Tehran (Grant No. 94038).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCM 58 kb)
Rights and permissions
About this article
Cite this article
Aghagoli, M.J., Shemirani, F. Hybrid nanosheets composed of molybdenum disulfide and reduced graphene oxide for enhanced solid phase extraction of Pb(II) and Ni(II). Microchim Acta 184, 237–244 (2017). https://doi.org/10.1007/s00604-016-2000-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-016-2000-7