Abstract
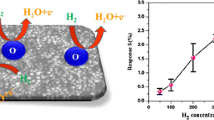
This work reports on the development of thin films of SnO2 doped with cerium and palladium and shows them to be viable materials for chemoresistive sensing of hydrogen (H2). The sensing material was synthesized by a hydrothermal route and with different weight percentage loadings of the dopants. The structural and morphological features were investigated by X-ray diffraction, field emission scanning electron microscopy, FTIR and X-ray photoelectron spectroscopy. Thin films were fabricated by spin coating on a ceramic substrate. The change in the resistance of the film was measured as a function of the concentration of H2. The results show that the amount of loading with Ce and Pd has a large effect on the performance. The Ce doped nanocomposite sensor has a lower detection limit of 50 ppm of H2 and covers the 50 to 500 ppm H2 concentration range if operated at the optimum temperature of 200 °C and a working voltage of 5 V.

Ce and Pd doped SnO2 based chemoresistive gas sensors were developed for H2 gas. The y show an appreciable detection limit, sensitivity and selectivity.









Similar content being viewed by others
References
Sahoo P, Dhara S, Dash S, Amirthapandian S, Arun KP, Tyagi AK (2013) Room temperature H2 sensing using functionalized GaN nanotubes with ultra-low activation energy. Int J Hydrog Energy 38:3513–3520
Eranna G, Joshi BC, Runthala DP, Gupta RP (2004) Oxide materials for development of integrated gas sensors-a comprehensive review. Crit Rev Solid State Mater Sci 29:111–188
Huang JR, Hsu WC, Chen HI, Liu WC (2007) Comparative study of hydrogen sensing characteristics of a Pd/GaN Schottky diode in air and N2 atmospheres. Sens Actuators B Chem 123:1040–1048
Soo MT, Cheong KY, Mohd Noor AF (2010) Advances of SiC-based MOS capacitor hydrogen sensors for harsh environment applications. Sens Actuators B Chem 151:39–55
Abdulah QN, Yam FK, Hassan JJ, Chin CW, Hassan Z, Bououdina M (2013) High performance room temperature GaN-nanowires hydrogen gas sensor fabricated by chemical vapor deposition (CVD) technique. Int J Hydrog Energy 38:14085–14101
Linke S, Dallmer M, Werner R, Moritz W (2012) Low energy hydrogen sensor. Int J Hydrog Energy 37:17523–17528
Korotcenkov G, Brinzari V, Cho BK (2016) Conductometric gas sensors based on metal oxides modified with gold nanoparticles: a review. Microchim Acta 183:1033–1054. https://doi.org/10.1007/s00604-015-1741-z
Kanan SM, El-Kadri OM, Abu-Yousef IA, Kanan MC (2009) Semiconducting metal oxide based sensors for selective gas pollutant detection. Sensors 9:8158–8196
Liwei W, Yanfei K, Xianghong L, Shoumin Z, Weiping H, Shurong W (2012) ZnO nanorod gas sensor for ethanol detection. Sens Actuators B Chem 162:237–243
Korotcenkov G, Brinzari V, Cho BK (2016) Conductometric gas sensors based on metal oxides modified with gold nanoparticles: a review. Microchim Acta 183:1033–1054
Batzill M, Diebold U (2005) The surface and materials science of tin oxide. Prog Surf Sci 79:47–154
Korotcenkov G (2007) Metal oxides for solid-state gas sensors: what determines our choice? Mater Sci Eng B 139:1–23
Brasan N, Stetter JR, Findlay M, Gopel W (2000) High performance gas sensing of CO: comparative tests for (SnO2-based) semiconducting and for electrochemical sensors. Sens Actuators B Chem 66:31–33
Zhang G, Liu M (2000) Effect of particle size and dopant on properties of SnO2-based gas sensors. Sens Actuators B Chem 69:144–152
Chowdhuri A, Gupta V, Sreenivas K (2003) Fast response H2S gas sensing characteristics with ultra-thin CuO is lands on sputtered SnO2. Sens Actuators B Chem 93:572–579
Jain K, Pant RP, Lakshmikumar ST (2006) Effect of Ni doping on thick film SnO2 gas sensor. Sens Actuators B Chem 113:823–829
Rahman MM, Jamal A, Khan SB, Faisal M (2011) Highly sensitive ethanol chemical sensor based on Ni-doped SnO2 nanostructure materials. Biosens Bioelectron 28:127–134
Wang Y, Wang YM, Cao JL, Kong FH, Xia HJ, Zhang J, Zhu BL, Wang SR, Wu SH (2008) Low-temperature H2S sensors based on ag-doped -Fe2O3 nanoparticles. Sens Actuators B Chem 131:183–189
Liu X, Zhang J, Guo X, Wu S, Wang S (2010) Amino acid-assisted one pot assembly of au, Pt nanoparticles onto one-dimensional ZnO microrods. Nano 2:1178–1184
Liu X, Zhang J, Guo X, Wu S, Wang S (2011) Enhanced sensor response of Ni-doped SnO2 hollow spheres. Sens Actuators B Chem 152:162–167
Wang Y, Wang SR, Zhao YQ, Zhu BL, Kong FH, Wang D, Wu SH, Huang WP, Zhang SM (2007) H2S sensing characteristics of Pt-doped Fe2O3 thick film sensors. Sens Actuators B Chem 125:79–84
Jiang ZW, Guo Z, Sun B, Jia Y, Li MQ, Liu JH (2010) Highly sensitive and selective butanone sensors based on cerium-doped SnO2 thin films. Sens Actuators B Chem 145:667–673
Liu D, Liu T, Zhang H, Chengling LV, Zeng W, Zhang J (2012) Gas sensing mechanism and properties of Ce-doped SnO2 sensors for volatile organic compounds. Mater Sci Semicond Process 15:438–444
Bittencourt C, Llobet E, Silva MAP, Landers R, Nieto L, Vicaro KO, Sueiras JE, Calderer J, Correig X (2003) Influence of the deposition method on the morphology and elemental composition of SnO2 films for gas sensing: atomic force and X-ray photoemission spectroscopy analysis. Sens Actuators B Chem 92:67–72
Ghimbeu CM, Schoonman J, Martine L, Maryam S (2007) Electrostatic spray deposited zinc oxide films for gas sensor applications. Appl Surf Sci 253:7483–7489
Ferro RJ, Rodriguez A, Jimenez I, Cirera A, Cerda J, Morante JR (2005) Gas- sensing properties of sprayed films of (CdO) x (ZnO)1-x mixed oxide. IEEE Sensors J 5:48–52
Gong H, Hu JQ, Wang JH, Ong CH, Zhu FR (2006) Nano-crystalline cu-doped ZnO thin film gas sensor for CO. Sens Actuators B Chem 115:247–251
Raghu S, Santhosh PN, Ramaprabhu S (2016) Nanostructured palladium modified graphitic carbon nitride–high performance room temperature hydrogen sensor. Int J Hydrog Energy 41(45):20779–20786
Lange U, Hirsch T, Mirsky VM, Wolfbeis OS (2011) Hydrogen sensor based on a graphene–palladium nanocomposite. Electrochim Acta 56(10):3707–3712
Hong J, Lee S, Seo J, Pyo S, Kim J, Lee T (2015) A highly sensitive hydrogen sensor with gas selectivity using a PMMA membrane-coated Pd nanoparticle/single-layer graphene hybrid. ACS Appl Mater Interfaces 7(6):3554–3561
Van der Bent JF, Puik E, Tong HD, van Rijn CJM (2015) Temperature balanced hydrogen sensor system with coupled palladium nanowires. Sensors Actuators A Phys 226:98–106
Raghu S, Santhosh PN, Ramaprabhu S (2016) Nanostructured palladium modified graphitic carbon nitride high performance room temperature hydrogen sensor. Int J Hydrog Energy 41:20779–20786
Hong J, Lee S, Seo J, Pyo S, Kim J, Lee T (2015) A highly sensitive hydrogen sensor with gas selectivity using a PMMA membrane-coated Pd nanoparticle/single-layer graphene hybrid. ACS Appl Mater Interfaces 7:3554–3561
Chung MG, Kim DH, Seo DK, Kim T, Im HU, Lee HM, Yoo JB, Hong SH, Kang TJ, Kim YH (2012) Flexible hydrogen sensors using graphene with palladium nanoparticle decoration. Sensors Actuators B 169:387–392
Majumdar S, Nag P, Devi PS (2014) Enhanced performance of CNT/SnO2 thick film gas sensors towards hydrogen. Mater Chem Phys 147:79–85
El-Maghraby EM, Qurashi A, Yamazaki T (2013) Synthesis of SnO2−nanowires their structural and H2 gas sensing properties. Ceram Int 39:8475–8480
Sripada R, Parambath VB, Baro M, Nair SPN, Sundara R (2015) Platinum and platinum-iron alloy nanoparticles dispersed nitrogen-doped graphene as high-performance room temperature hydrogen sensor. Int J Hydrog Energy 40:10346–10353
Dhall S, Jaggi N (2015) Room temperature hydrogen gas sensing properties of Pt sputtered F-MWCNTs/SnO2 network. Sensors Actuators B 210:742–747
Acknowledgements
This research work was supported by Uma.V and Pradeep.N, Department of Nanoscience and Technology, Mount Carmel College, Bengaluru, India, who provided expertise which greatly assisted the research.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 5.69 mb)
Rights and permissions
About this article
Cite this article
Deivasegamani, R., Karunanidhi, G., Santhosh, C. et al. Chemoresistive sensor for hydrogen using thin films of tin dioxide doped with cerium and palladium. Microchim Acta 184, 4765–4773 (2017). https://doi.org/10.1007/s00604-017-2514-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-017-2514-7