Abstract
Rapeseed (Brassica napus L.), which is the third leading source of vegetable oil, is sensitive to drought stress during the early vegetative growth stage. To investigate the initial response of rapeseed to drought stress, changes in the protein expression profiles of drought-sensitive (RGS-003) and drought-tolerant lines (SLM-003), and their F1 hybrid, were analyzed using a proteomics approach. Seven-day-old rapeseed seedlings were treated with drought stress by restricting water for 7 days, and proteins were extracted from roots and separated by two-dimensional polyacrylamide gel electrophoresis. In the sensitive rapeseed line, 35 protein spots were differentially expressed under drought stress, and proteins related to metabolism, energy, disease/defense, and transport were decreased. In the tolerant line, 32 protein spots were differentially expressed under drought stress, and proteins involved in metabolism, disease/defense, and transport were increased, while energy-related proteins were decreased. Six protein spots in F1 hybrid were common among expressed proteins in the drought-sensitive and -tolerant lines. Notably, tubulin beta-2 and heat shock protein 70 were decreased in the drought-sensitive line and hybrid F1 plants, while jasmonate-inducible protein and 20S proteasome subunit PAF1 were increased in the F1 hybrids and drought-tolerant line. These results indicate that (1) V-type H+ ATPase, plasma-membrane associated cation-binding protein, HSP 90, and elongation factor EF-2 have a role in the drought tolerance of rapeseed; (2) The decreased levels of heat shock protein 70 and tubulin beta-2 in the drought-sensitive and hybrid F1 lines might explain the reduced growth of these lines in drought conditions.






Similar content being viewed by others
Abbreviations
- 2-DE:
-
Two-dimensional polyacrylamide gel electrophoresis
- CBB:
-
Coomassie brilliant blue
- MS:
-
Mass spectrometry
- LC:
-
Liquid column
- pI:
-
Isoelectric point
- IEF:
-
Isoelectric focusing
References
Ali GM, Komatsu S (2006) Proteomic analysis of rice leaf sheath during drought stress. J Proteome Res 5:396–403
Asher G, Reuven N, Shaul Y (2006) 20S proteasomes and protein degradation by default. BioEssays 28:844–849
Bandehagh A, Salekdeh GH, Toorchi M, Mohammadi A, Komatsu S (2011) Comparative proteomic analysis of canola leaves under salinity stress. Proteomics 11:1965–1975
Benedetti CE, Costa CL, Turcinelli SR, Arruda P (1998) Differential expression of a novel gene in response to coronatine, methyl jasmonate and wounding in the coi1 mutant of Arabidopsis. Plant Physiol 116:1037–1042
Bevan M, Bancroft I, Bent E, Love K, Goodman H, Dean C, Bergkamp R, Dirkse W, Van Staveren M, Stiekema W (1998) Analysis of 1.9 Mb of contiguous sequence from chromosome 4 of Arabidopsis thaliana. Nature 391:485–488
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Carystinos GD, MacDonald HR, Monroy AF, Dhindsa RS, Poole RJ (1995) Vacuolar H+-translocating pyrophosphatase is induced by anoxia or chilling in seedlings of rice. Plant Physiol 108:641–649
Castillejo MA, Maldonado AM, Ogueta S, Jorrin JV (2008) Proteomic analysis of responses to drought stress in sunflower (Helianthus annuus) leaves by 2DE gel electrophoresis and mass spectrometry. Open Proteomics J 1:59–71
Chaves MM, Pereira JS, Maroco J, Rodrigues ML, Ricardo CPP, Osorio ML, Carvalho I, Faria T, Pinheiro C (2002) How plants cope with water stress in the field? Photosynthesis and growth. Ann Bot 89:907–916
Chaves MM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103:551–560
Demirevska K, Simova-Stoilova L, Vassileva V, Vaseva I, Grigorova B, Feller U (2008) Drought-induced leaf protein alterations in sensitive and tolerant wheat varieties. Gen Appl Plant Physiol 34:79–102
Dunn MA, Hughes MA, Pearce RS, Jack PL (1990) Molecular characterization of a barley gene induced by cold treatment. J Exp Bot 41:1405–1413
Gong H, Chen K, Chen G, Wang S, Zhang C (2003) Drought stress stimulates p-nitrophenyl phosphate hydrolysis rate of the plasma membrane H+-ATPase from wheat leaves. Plant Growth Regul 40:139–145
Grune T, Jung T, Merker K, Davies KJ (2004) Decreased proteolysis caused by protein aggregates, inclusion bodies, plaques, lipofuscin, ceroid and ‘aggresomes’ during oxidative stress, aging and disease. Int J Biochem Cell Biol 36:2519–2530
Guy CL, Neimi KJ, Brambl R (1985) Altered gene expression during cold acclimation of spinach. Proc Natl Acad Sci USA 82:3673–3677
Hajheidari M, Abdollahian-Noghabi M, Askari H, Heidai M, Sadeghian SY, Ober ES, Salekdeh GH (2005) Proteome analysis of sugar beet leaves under drought stress. Proteomics 5:950–960
Hajheidari M, Eivazi A, Buchanan BB, Wong JH, Majidi I, Salekdeh GH (2007) Proteomics uncovers a role for redox in drought tolerance in wheat. J Proteome Res 6:1451–1460
Harris D, Tripathi RS, Joshi A (2002) On-farm seed priming to improve crop establishment and yield in dry direct-seeded rice. In: Pandey S, Mortimer M, Wade L, Tuong TP, Lopes K, Hardy B (eds) Direct Seeding: Research Strategies and Opportunities. International Research Institute, Manila, pp 231–240
Hashimoto M, Larisa K, Sawa S, Furukawa T, Komatsu S, Koshiba T (2004) A novel rice PR10 protein, RSOsPR10, specifically induced in roots by biotic and abiotic stresses, possibly via the jasmonic acid signaling pathway. Plant Cell Physiol 45:550–559
Hosseini SM, Hassibi P (2011) Effects of water deficit stress on several quantitative and qualitative characteristics of canola (Brassica napus L.) cultivars. Nat Sci Biol 3:120–125
Jain D, Chattopadhyay D (2010) Analysis of gene expression in response to water deficit of chickpea (Cicer arietinum L.) varieties differing in drought tolerance. BMC Plant Biol 10:24
Jangpromma N, Kitthaisong S, Lomthaisong K, Daduang S, Jaisil P, Thammasirirak S (2010) A Proteomics analysis of drought stress-responsive proteins as biomarker for drought-tolerant sugarcane cultivars. Am J Biochem Biotech 6:89–102
Jensen CR, Mogensen VO, Mortensen G, Andersen MN, Schjoerring JK, Thage JH, Koribidis J (1996) Leaf photosynthesis and drought adaptation in field grown oilseed rape (Brassica napus L.). Aust J Plant Physiol 23:631–644
Johnson-Flanagan AM, Singh J (1987) Alteration of gene expression during the induction of freezing tolerance in Brassica napus suspension cultures. Plant Physiol l85:699–705
Kaya MD, Okçub G, Ataka M, Çikili Y, Kolsarca Ö (2006) Seed treatments to overcome salt and drought stress during germination in sunflower (Helianthus annuus L.). Eur J Agron 24:291–295
Komatsu S, Konishi H, Shen S, Yang G (2003) Rice proteomics: a step toward functional analysis of the rice genome. Mol Cell Proteomics 2:2–10
Kregel KC (2002) Heat shock proteins: modifying factors in physiological stress responses and acquired thermo tolerance. J Appl Physiol 92:2177–2186
Kurkela S, Franck M (1990) Cloning and characterization of a cold and ABA-inducible Arabidopsis gene. Plant Mo1 Biol 15:137–144
Low R, Rockel B, Kirsch M, Ratajczak R, Hortensteiner S, Martinoia E, Liittge U, Rausch T (1996) Early salt stress effects on the differential expression of vacuolar H+-ATPase genes in roots and leaves of Mesembryanthemum crystallinum. Plant Physiol 110:259–265
Magnotta SM, Gogarten JP (2002) Multi site polyadenylation and transcriptional response to stress of a vacuolar type H+-ATPase subunit A gene in Arabidopsis thaliana. BMC Plant Biol 2:3
Mason HS, Mullet JE (1990) Expression of two soybean vegetative storage protein genes during development and in response to water deficit, wounding, and jasmonic acid. Plant Cell 2:569–579
Mittler R (2006) Abiotic stress, the field environment and stress combination. Trends Plant Sci 11:15–19
Mohapatra SS, Poole RJ, Dhindsa RS (1987) Changes in protein patterns and translatable messenger RNA populations during cold acclimation of alfalfa. Plant Physiol 84:1172–1176
Narasimhan ML, Binzel ML, Perez-Prat E, Chen Z, Nelson DE, Singh NK, Bressan RA, Hasegawa PM (1991) NaCl regulation of tonoplast ATPase 70 kilodalton subunit mRNA in tobacco cells. Plant Physiol 97:562–568
Nicot N, Hausman J, Hoffmann L, Evers D (2005) Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic stress. J Exp Bot 56:2907–2914
Nouri MZ, Komatsu S (2010) Comparative analysis of soybean plasma membrane proteins under osmotic stress using gel-based and LC MS/MS-based proteomics approaches. Proteomics 10:1930–1945
Ohashi Y, Nakayama N, Saneoka H, Fujita K (2006) Effects of drought stress on photosynthetic gas exchange, chlorophyll fluorescence and stem diameter of soybean plants. Biol Plant 50:138–141
Okcu G, Kaya MD, Atak M (2005) Effects of salt and drought stresses on germination and seedling growth of pea (Pisum sativum L.). Turk J Agric For 29:237–242
Orr W, White TC, Iu B, Robert L, Singh J (1995) Characterization of a low temperature induced cDNA from winter Brassica napus encoding the 70 kDa subunit of tonoplast ATPase. Plant Mol Biol 28:943–948
Parsell DA, Lindquist S (1993) The function of heat shock proteins in stress tolerance: degradation and reactivation of damaged proteins. Annu Rev Genet 27:437–496
Peng Z, Wang M, Li F, Lv H, Li C, Xia G (2009) A proteomic study of the response to salinity and drought stress in an introgression strain of bread wheat. Mol Cell Proteomics 8:2676–2686
Reuveni M, Bennett AB, Bressan RA, Hasegawa PM (1990) Enhanced H+ transport capacity and ATP hydrolysis activity of the tonoplast H+-ATPase after NaCl adaptation. Plant Physiol 94:525–530
Riccardi F, Gazeau P, de Vienne D, Zivy M (1998) Protein changes in response to progressive water deficit in maize. Quantitative variation and polypeptide identification. Plant Physiol 117:1253–1263
Rizhsky LH, Liang H, Shuman J, Shulaev V, Davletova S, Mittler R (2004) When defense pathways collide: the response of Arabidopsis to a combination of drought and heat stress. Plant Physiol 134:1683–1696
Salekdeh GH, Siopongco J, Wade LJ, Ghareyazie B, Bennett J (2002) Proteomics analysis of rice leaves during drought stress and recovery. Proteomics 2:1131–1145
Sangtarash MH, Qaderi MM, Chinnappa CC, Reid DM (2009) Differential sensitivity of canola (Brassica napus) seedlings to ultraviolet-B radiation, water stress and abscisic acid. Environ Exp Bot 66:212–219
Sasaki Y, Asamizu D, Shibata Y, Nakamura T, Kaneko K, Awai T, Masuda H, Shimada K, Takamiya S, Ohta H (2000) Genome-wide expression-monitoring of jasmonate-responsive genes of Arabidopsis using cDNA arrays. Biochem Soc Trans 28:863–864
ShiraniRad AH, Naeemi M, Esfahani SN (2010) Evaluation of terminal drought stress tolerance in spring and winter rapeseed genotypes. Iran J Crop Sci 12:112–126
Tripathy JN, Zhang J, Robin S, Nguyen TT, Nguyen HT (2000) QTLs for cell-membrane stability mapped in rice (Oryza sativa L.) under drought stress. Theor Appl Genet 100:1197–1202
Turner JG, Ellis C, Devoto A (2002) The jasmonate signal pathway. Plant Cell 14:153–164
Voss P, Grune T (2007) The nuclear proteasome and the degradation of oxidatively damaged proteins. Amino Acids 32:527–534
Wasternack C, Hause B (2002) Jasmonates and octadecanoids: signals in plant stress responses and development. Prog Nucleic Acid Res Mol Biol 72:165–221
Whittier JE, Xiong Y, Rechsteiner MC, Squier TC (2004) Hsp90 enhances degradation of oxidized calmodulin by the 20 S proteasome. J Biol Chem 279:35–42
Yoshimura K, Yabuta Y, Ishikawa T, Shigeoka S (2000) Expression of spinach ascorbate peroxidase isoenzymes in response to oxidative stresses. Plant Physiol 123:223–233
Zang X, Komatsu S (2007) A proteomics approach for identifying osmotic-stress-related proteins in rice. Phytochemistry 68:426–437
Zeid IM, Shedeed ZA (2006) Response of alfalfa to putrescine treatment under drought stress. Biol Plant 50:635–640
Acknowledgments
The authors thank Dr. Yohei Nanjo and Dr. Keito Nishazawa for their valuable discussion.
Conflict of interest
This work was supported by the Scholarship Section of the Ministry of Science, Research and Technology and Tarbiat Modares University of I. R. Iran and National Institute of Crop Science of Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
726_2012_1299_MOESM1_ESM.pptx
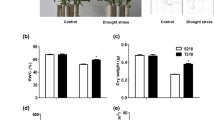
Effect of drought stress on rapeseed roots, hypocotyls, and leaves. Seven-day-old rapeseed seedlings were treated without (white) or with (black) drought stress for 1, 2, 3, 4, 5, 6, and 7 days. Leaf weight, hypocotyl weight, hypocotyl length, and root number were measured daily. Three rapeseed plants were used for each experiment and three independent experiments were performed. Values are presented as the mean ± SE and were compared using the Student t test (no significant changes were detected) (PPTX 90 kb)
Rights and permissions
About this article
Cite this article
Mohammadi, P.P., Moieni, A. & Komatsu, S. Comparative proteome analysis of drought-sensitive and drought-tolerant rapeseed roots and their hybrid F1 line under drought stress. Amino Acids 43, 2137–2152 (2012). https://doi.org/10.1007/s00726-012-1299-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-012-1299-6