Abstract
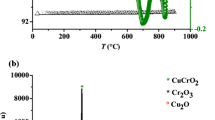
CuFeO2 single crystal, synthesized by the flux method, is a narrow band gap semiconductor crystallizing in the delafossite structure with a direct optical transition of 1.63 eV. The oxide exhibits a good chemical stability; the semi-logarithmic plot gave an exchange current density of 0.60 µA cm−2 in KCl (0.5 M) electrolyte. CuFeO2 shows p-type conductivity; the origin of acceptors Cu2+ results from oxygen insertion in the layered lattice where most of excess holes are trapped in surface-polaron states. The electrochemical study is confined in the (a,b) plane and reversible oxygen intercalation is evidenced from the intensity potential characteristics. The detailed photoelectrochemical studies have been reported for the first time on the single crystal. The photocurrent is ascribed to the transfer Cu+:3d→3d. The capacitance measurement (C−2–V) shows a linear behavior from which a flat band potential of +0.54 VSCE and a density N A of 1.60 × 1018 cm−3 were determined. The valence band, located at 5.33 eV below vacuum, is made up of Cu-3d orbital typical of delafossite oxides. The Nyquist plot shows a semicircle attributed to a capacitive behavior with a low density of surface states within the gap. The centre is localized below the real axis with an angle of 16.2° ascribed to a constant phase element (CPE), a single barrier of the junction CuFeO2/electrolyte and one relaxation time of the electrical equivalent circuit.







Similar content being viewed by others
Notes
Calculated from the lattice constant (0.3885 nm) of iron crystallizing in a face centered cubic lattice.
χ is the geometrical mean of EA values of the constituent atoms, \(\chi \left( {{\text{CuFeO}}_2 } \right) = \left\{ {\left( {\chi {\text{Cu}}} \right)\left( {\chi {\text{Fe}}} \right)\left( {\chi {\text{O}}} \right)^2 } \right\}^{{1 \mathord{\left/ {\vphantom {1 4}} \right. \kern-\nulldelimiterspace} 4}} \). For a neutral atom, χ is given by \({{\left( {A_f + I_1 } \right)} \mathord{\left/ {\vphantom {{\left( {A_f + I_1 } \right)} 2}} \right. \kern-\nulldelimiterspace} 2}\) where A f is the electron affinity and I 1 the first ionization energy, the values were taken from [23].
References
Hu CC, Nian JN, Teng H (2008) Sol Energy Mater Sol Cells 92:1071. doi:10.1016/j.solmat.2008.03.012
Tai Y-W, Chen J-S, Yang C-C, Wan B-Z (2004) Catal Today 90:95. doi:10.1016/j.cattod.2004.04.054
Gondal MA, Hameed A, Yamani ZH, Arfaj A (2004) Chem Phys Lett 385:111. doi:10.1016/j.cplett.2003.12.066
Claverie J, Campet G, Conte D, Le flem G, Hagenmuller P (1983) Phys Status Solidi (a) 77:603. doi:10.1002/pssa.2210770224
Saadi S, Bouguelia A, Derbal A, Trari M (2007) J photochem photobiol 187:97
Elazhari M, Ammar A, Elaatmani M, Trari M, Doumerc JP (1997) Eur J Solid State Inorg Chem 34:503
Prewitt CT, Shannon RD, Rogers DB (1971) Inorg Chem 10:719. doi:10.1021/ic50098a012
Younsi M, Aider A, Bouguelia A, Trari M (2005) Sol Energy 78:574. doi:10.1016/j.solener.2004.01.012
Omeiri S, Gabès Y, Bouguelia A, Trari M (2008) J Electroanal Chem 614:31. doi:10.1016/j.jelechem.2007.11.002
Trari M, Bouguelia A, Bessekhouad Y (2006) Sol Energy Mater Sol Cells 90:190. doi:10.1016/j.solmat.2005.03.003
Doumerc JP, Wichainchai A, Ammar A, Pouchar M, Hagenmuller P (1986) Mater Res Bull 121:745. doi:10.1016/0025-5408(86)90155-8
Zhao TR, Takei H (1997) Mater Res Bull 32:1377. doi:10.1016/S0025-5408(97)00126-8
Dordor P, Chaminade JP, Wichainchai A, Marquestaut E, Doumerc JP, Pouchard P, Hagenmuller P (1988) J Solid State Chem 75:105. doi:10.1016/0022-4596(88)90307-6
Cubicciotti D (1988) NACE 44:875
Mugnier E, Barnabé A, Tailhades P (2006) Solid State Ion 177:607. doi:10.1016/j.ssi.2005.11.026
Ong KP, Bai K, Wu P (2008) J Alloy Comp 449(1-2):366. doi:10.1016/j.jallcom.2006.01.141
Trari M, Topfer J, Dordor P, Grenier JC, Pouchard M, Doumerc JP (2005) J Solid State Chem 178:2751. doi:10.1016/j.jssc.2005.06.009
Grenier JC, Wattiaux A, Lagueyte N, Park JC, Marquestaut E, Etourneau J, Pouchard M (1991) Physica C 173:139. doi:10.1016/0921-4534(91)90360-B
Tauc J (1974) Amorphous and liquid semiconductors. Plenum, London
Pollert E, Hejtmaneck J, Doumerc JP, Claverie J, Hagenmuller P (1983) J Phys Chem Solids 44:273. doi:10.1016/0022-3697(83)90095-1
Xia S-J, Zhou W-F (1995) Electrochim Acta 40:175. doi:10.1016/0013-4686(94)00280-E
Butler MA, Ginley DS (1978) J Electrochem Soc 125:228. doi:10.1149/1.2131419
Lide DR (1997, 1998) Handbook of chemistry and physics, 78th edn. McGraw-Hill, New York
Barsoukov E, Macdonald JR (2005) Impedance spectroscopy, theory, experiment and applications, 2nd edn. Wiley, New York
Acknowledgement
The authors are grateful to M. Younsi for valuable suggestions regarding to the corrosion measurements. Financial support of this work was provided the Faculty of Chemistry (Algiers).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Omeiri, S., Bellal, B., Bouguelia, A. et al. Electrochemical and photoelectrochemical characterization of CuFeO2 single crystal. J Solid State Electrochem 13, 1395–1401 (2009). https://doi.org/10.1007/s10008-008-0703-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-008-0703-3