Abstract
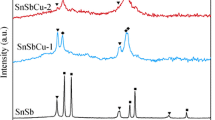
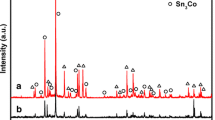
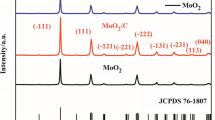
Nanostructured hollow Sn–Cu multi-phase composite nanoparticles anode that contains Sn and Cu6Sn5 was synthesized via galvanic replacement reaction using Sn nanoparticles as a sacrificial template. The sacrificial oxidation of Sn and simultaneous reduction of Cu on the surface because of the redox potential difference is proposed to account for the formation of hollow Sn–Cu nanostructures. The structural evolution of the Sn–Cu hollow nanoparticle, in the process of galvanic replacement and structure, composition changes during charge/discharge processes were studied based on scanning electron microscope, X-ray powder diffraction, transmission electron microscopy, and energy dispersive X-ray spectroscopy investigations. The electrochemical properties of the samples were evaluated by galvanostatic discharge–charge cycling, cyclic voltammetry, and electrochemical impedance spectroscopy. Compared with solid Sn–Cu nanoparticles, hollow Sn–Cu nanoparticles showed better capacity retention. The improved electrochemical performance may be attributed to the stable hollow structure and the combination of Cu6Sn5. The facile solution-based process and excellent cycling stability show great potential of the multi-phase Sn–Cu hollow composite nanoparticles as an anode material for lithium-ion batteries.








Similar content being viewed by others
References
Luo B, Wang B, Liang MH, Ning J, Li XL, Zhi LJ (2012) Reduced graphene oxide-mediated growth of uniform tin-core/carbon-sheath coaxial nanocables with enhanced lithium ion storage properties. Adv Mater 24:1405–1409
Winter M, Besenhard JO (1999) Electrochemical lithiation of tin and tin-based intermetallics and composites. Electrochim Acta 45:31–50
Lee KT, Jung YS, Oh SM (2003) Synthesis of tin-encapsulated spherical hollow carbon for anode material in lithium secondary batteries. J Am Chem Soc 125:5652–5653
Sun YG, Xia YN (2003) Alloying and dealloying processes involved in the preparation of metal nanoshells through a galvanic replacement reaction. Nano Lett 3:1569–1572
Hassoun J, Derrien G, Panero S, Scrosati B (2008) A nanostructured Sn-C composite lithium battery electrode with unique stability and high electrochemical performance. Adv Mater 20:3169–3175
Xu YH, Liu Q, Zhu YJ, Liu YH, Langrock A, Zachariah MR, Wang CS (2013) Uniform nano-Sn/C composite anodes for lithium ion batteries. Nano Lett 13:470–474
Wolfenstine J, Foster D, Read J, Behl WK, Luecke W (2000) Experimental confirmation of the model for microcracking during lithium charging in single-phase alloys. J Power Sources 87:1–3
Wolfenstine J (1999) Critical grain size for microcracking during lithium insertion. J Power Sources 79:111–113
Dimitrijevic BJ, Aifantis KE, Hackl K (2012) The influence of particle size and spacing on the fragmentation of nanocomposite anodes for Li batteries. J Power Sources 206:343–348
Chen JZ, Yang L, Fang SH, Hirano S (2012) Synthesis of mesoporous Sn-Cu composite for lithium ion batteries. J Power Sources 209:204–208
Gonzalez JR, Alcantara R, Nacimiento F, Tirado JL (2012) Optimization of tin intermetallics and composite electrodes for lithium-ion batteries obtained by sonochemical synthesis. J Solid State Electrochem 17:2495–2501
Nacimiento F, Lavela P, Tirado JL, Jimenez-Mateos JM (2012) A facile carbothermal preparation of Sn-Co-C composite electrodes for Li-ion batteries using low-cost carbons. J Solid State Electrochem 16:953–962
Shin NR, Kang YM, Song MS, Kim DY, Kwon HS (2009) Effects of Cu substrate morphology and phase control on electrochemical performance of Sn-Ni alloys for Li-ion battery. J Power Sources 186:201–205
Lee JM, Chang WS, Yu BC, Kim H, Im D, Doo SG, Sohn HJ (2012) Enhancement of cyclability using recombination reaction of Cu for Sn2Fe nanocomposite anode for lithium-ion batteries. Electrochem Commun 12:928–932
Kepler KD, Vaughey JT, Thackeray MM (1999) Li x Cu6Sn5 (0 < x < 13): an intermetallic insertion electrode for rechargeable lithium batteries. Electrochem Solid-State Lett 2:307–309
Larcher D, Beaulieu LY, Macneil DD, Dahn JR (2000) In situ X-ray study of the electrochemical reaction of Li with η΄-Cu6Sn5. J Electrochem Soc 147:1658–1662
Hu RZ, Zeng MQ, Zhu M (2009) Cyclic durable high-capacity Sn/Cu6Sn5 composite thin film anodes for lithium ion batteries prepared by electron-beam evaporation deposition. Electrochim Acta 54:2843–2850
Tamura N, Kato Y, Mikami A, Kamino M, Matsuta S, Fujitani S (2006) Study on Sn-Co alloy electrodes for lithium secondary batteries. J Electrochem Soc 153:A2227–A2231
Yang S, Feng X, Zhi L, Cao Q, Maier J, Müllen K (2010) Nanographene-constructed hollow carbon spheres and their favorable electroactivity with respect to lithium storage. Adv Mater 22:838–842
Tang YF, Yang L, Fang SH, Qiu Z (2009) Li4Ti5O12 hollow microspheres assembled by nanosheets as an anode material for high-rate lithium ion batteries. Electrochim Acta 54:6244–6249
Ma H, Cheng FY, Chen J, Zhao JZ, Li CS, Tao ZL, Liang J (2007) Nest-like silicon nanospheres for high-capacity lithium storage. Adv Mater 19:4067–4070
Kim H, Cho J (2008) Template synthesis of hollow Sb nanoparticles as a high-performance lithium battery anode material. Chem Mater 20:1679–1681
Ye JF, Zhang HJ, Yang R, Li XG, Qi LM (2010) Morphology-controlled synthesis of SnO2 nanotubes by using 1D silica mesostructures as sacrificial templates and their applications in lithium-ion batteries. Small 6:296–306
Park MH, Cho YH, Kim K, Kim J, Liu M, Cho J (2011) Germanium nanotubes prepared by using the Kirkendall effect as anodes for high-rate lithium batteries. Angew Chem Int Ed 50:9647–9650
Zeng HC (2006) Synthetic architecture of interior space for inorganic nanostructures. J Mater Chem 16:649–662
Yu JG, Guo HT, Davis SA, Mann S (2006) Fabrication of hollow inorganic microspheres by chemically induced self-transformation. Adv Funct Mater 16:2035–2041
Caruso F, Caruso RA, Mӧhwald H (1998) Nanoengineering of inorganic and hybrid hollow spheres by colloidal templating. Science 282:1111–1114
Velikov KP, van Blaaderen A (2001) Synthesis and characterization of monodisperse core-shell colloidal spheres of zinc sulfide and silica. Langmuir 17:4779–4786
Gao XY, Zhang JS, Zhang LD (2002) Hollow sphere selenium nanoparticles: their in-vitro Anti hydroxyl radical effect. Adv Mater 14:290–293
Qi LM, Li J, Ma JM (2002) Biomimetic morphogenesis of calcium carbonate in mixed solutions of surfactants and double-hydrophilic block copolymers. Adv Mater 14:300–303
Railsback JG, Johnston-Peck Aaron C, Wang JW, Tracy JB (2010) Size-dependent nanoscale Kirkendall effect during the oxidation of nickel nanoparticles. ACS Nano 4:1913–1920
Zhou SH, Varughese B, Eichhorn B, Jackson G, McIlwrath K (2005) Pt-Cu core-shell and alloy nanoparticles for heterogeneous NO x reduction: anomalous stability and reactivity of a core-shell nanostructure. Angew Chem Int Ed 44:4539–4543
Ke FS, Huang L, Cai JS, Sun SG (2007) Electroplating synthesis and electrochemical properties of macroporous Sn-Cu alloy electrode for lithium-ion batteries. Electrochim Acta 52:6741–6747
Li QY, Hu SJ, Wang HQ, Wang FP, Zhong XX, Wang XY (2009) Study of copper foam-supported Sn thin film as a high-capacity anode for lithium-ion batteries. Electrochim Acta 54:5884–5888
Kim MG, Sim S, Cho J (2010) Novel core-shell Sn-Cu anodes for lithium rechargeable batteries prepared by a redox-transmethalation reaction. Adv Mater 22:5154–5158
Naille S, Dedryvère R, Martinez H, Leroy S, Lippens PE, Jumas JC, Gonbeau D (2007) XPS study of electrode/electrolyte interfaces of η-Cu6Sn5 electrodes in Li-ion batteries. J Power Sources 174:1086–1090
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 51143009 and 51273145).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fan, X., Tang, X., Ma, D. et al. Novel hollow Sn–Cu composite nanoparticles anodes for Li-ion batteries prepared by galvanic replacement reaction. J Solid State Electrochem 18, 1137–1145 (2014). https://doi.org/10.1007/s10008-013-2370-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-013-2370-2