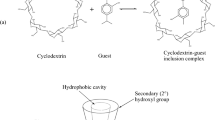
Abstract
Steroids are abundant in living organisms and are responsible for various biological functions. They are good candidates for inclusion complexes formation with cyclodextrins; therefore, plenty of literature describes these complexes and their application in various fields. There are, however, hardly any reviews summarizing this tremendous amount of information. Here, we review steroid drugs in relation to their complexation with cyclodextrins, highlighting the effect of cyclodextrin–steroid interaction on the solubility, stability and bioavailability of the entrapped steroid drugs. We have collected data on the changes in the pharmacokinetics of cyclodextrin-formulated steroid drugs designed for various routes of administration. We show some cyclodextrin-enabled steroid formulations introduced to the market. Sugammadex, the first cyclodextrin derivative approved as active pharmaceutical ingredient, which revolutionized anesthesia, is also discussed. Several examples demonstrate the catalysis and inhibition of various biotransformation reactions of steroids by cyclodextrin complexation used in biotechnology. The laboratory- and pilot-scale experiments for environmental application of cyclodextrin–steroid interaction are based on cyclodextrin-containing sorbents for capturing residual steroid drugs, such as contraceptives from purified wastewater.



Similar content being viewed by others
References
Ahmed SM (1997) Effect of cyclodextrins on the chemical stability of ST1435, a contraceptive steroid progestin, in aqueous solution. J Incl Phenom Mol Recognit Chem 27(1):85–96. https://doi.org/10.1023/A:1007946525783
Ahmed SM (1998) Improvement of solubility and dissolution of 19-norprogesterone via inclusion complexation. J Incl Phenom Mol Recognit Chem 30(2):111–125. https://doi.org/10.1023/A:1007979430842
Albers E, Müller BW (1992) Complexation of steroid hormones with cyclodextrin derivatives: substituent effects of the guest molecule on solubility and stability in aqueous solution. J Pharm Sci 81(8):756–761. https://doi.org/10.1002/jps.2600810808
Alberth M, Wu WM, Winwood D, Bodor N (1991) Lipophilicity, solubility and permeability of loteprednol etabonate: a novel, soft anti-inflammatory corticosteroid. J Biopharm Sci 2(2):115–125
Alekhina TM, Ryzhkova VM, Gusarova TI, Kurakov VV, Klabnichkina GA (1993) Microbiological transformation of steroid-β-cyclodextrin inclusion compounds. Khim Farm Zh 27(4):59–62
Andersen FM, Bundgaard H (1983) Influence of β-cyclodextrin on the stability of hydrocortisone in aqueous solution. Arch Pharm Chem Sci Ed 11(2):61–66. https://doi.org/10.1007/BF02974095
Andersen FM, Bundgaard H (1984) The influence of cyclodextrin complexation on the stability of betamethasone 17-valerate. Int J Pharm 20(1–2):155–162. https://doi.org/10.1016/0378-5173(84)90226-6
Anderson WR, Simpkins JW, Brewster ME, Bodor NS (1989) Evidence for prolonged suppression of stress-induced release of adrenocorticotropic hormone and corticosterone with a brain-enhanced dexamethasone-redox delivery system. Neuroendocrin 50(1):9–16. https://doi.org/10.1159/000125196
Arimori K, Uekama K (1987) Effects of beta- and gamma-cyclodextrins on the pharmacokinetic behavior of prednisolone after intravenous and intramuscular administrations to rabbits. J Pharmacobiodyn 10:390–395. https://doi.org/10.1248/bpb1978.10.390
Arimori K, Sakai A, Otagiri M, Uekama K (1984) Improvement in biopharmaceutics of prednisolone by β- and γ- cyclodextrins. J Incl Phenom 1(4):387–394. https://doi.org/10.1007/BF00665481
Arriagas N, Cabral-Marques H (2013) Obtention and characterization of a ciclesonide/methyl-beta cyclodextrin complex. In: Abstract book of 3rd European conference on cyclodextrins, Antalya, Turkey, 2–4 October 2013, p 30
Badawy FSI, Marshall AL, Ghorab MM, Adeyeye MC (1996) A study of the complexation between danazol and hydrophilic cyclodextrin derivatives. Drug Dev Ind Pharm 22(9 and 10):959–966. https://doi.org/10.3109/03639049609065926
Bary AR, Tucker IG, Davies NM (2000) Considerations in the use of hydroxypropyl-beta-cyclodextrin in the formulation of aqueous ophthalmic solutions of hydrocortisone. Eur J Pharm Biopharm 50:237–244. https://doi.org/10.1016/s0939-6411(00)00108-9
Basu K, Nair A, Williamson PA, Mukhopadhyay S, Lipworth BJ (2009) Airway and systemic effects of soluble and suspension formulations of nebulized budesonide in asthmatic children. Ann Allergy Asthma Immunol 103(5):436–441. https://doi.org/10.1016/s1081-1206(10)60365-1
Becirevic-Lacan M, Filipovic-Grcic J (2000) Effect of hydroxypropyl-β-cyclodextrin on hydrocortisone dissolution from films intended for ocular drug delivery. Pharmazie 55(7):518–520
Belikov VG, Kompantseva EV, Gavrilin MV, Umnova EF (1991) Potential use of β-cyclodextrin to improve the procedure for obtaining prednisolone. Khim-Farm Zh 25(2):48–49
Bhattarai B, Manickavachagam M, Suri R (2012) Development of novel cyclodextrin adsorbents for the removal of emerging contaminants from water. In: Abstracts of papers, 244th ACS national meeting and exposition, Philadelphia, 19–23 August 2012, ENVR-5
Bilensoy E, Sen M, Hincal AA (2006) Effect of drug physicochemical properties on in vitro characteristics of amphiphilic cyclodextrin nanospheres and nanocapsules. J Microencapsul 23(1):59–68. https://doi.org/10.1080/02652040500286227
Blode H, Schuermann R, Benda N (2008) Novel ethinyl estradiol-beta-cyclodextrin clathrate formulation does not influence the relative bioavailability of ethinyl estradiol or coadministered drospirenone. Contracept 77(3):171–176. https://doi.org/10.1016/j.contraception.2007.10.009
Bodor N, Murakami T, Wu WM (1992) Soft drugs 18. Oral and rectal delivery of loteprednol etabonate, a novel soft corticosteroid, in rats-for safer treatment of gastrointestinal inflammation. Pharm Res 12(6):674–869
Bodor N, Drustrup J, Wu W (2000) Effect of cyclodextrins on the solubility and stability of a novel soft corticosteroid, loteprednol etabonate. Pharmazie 55(3):206–209
Bom A, Bradley M, Cameron K, Clark JK, Van Egmond J, Feilden H, MacLean EJ, Muir AW, Palin R, Rees DC, Zhang MQ (2002) A novel concept of reversing neuromuscular block: chemical encapsulation of rocuronium bromide by a cyclodextrin-based synthetic host. Angew Chem Int Ed Engl 41:266–270. https://doi.org/10.1002/1521-3757(20020118)114:2%3c275:AID-ANGE275%3e3.0.CO;2-A
Brewster ME, Estes KS, Bodor N (1989) Development of a non-surfactant formulation for alfaxalone through the use of chemically-modified cyclodextrins. J Parenter Sci Technol 43(6):262–265
Brewster ME, Anderson WR, Loftsson T, Huang MJ, Bodor N, Pop E (1995) Preparation, characterization and anesthetic properties of 2-hydroxypropyl-β-cyclodextrin complexes of pregnanolone and pregnenolone in rat and mouse. J Pharm Sci 84(10):1154–1159. https://doi.org/10.1002/jps.2600841004
Brown GA, Martini ER, Roberts BS, Vukovich MD, King DS (2002) Acute hormonal response to sublingual androstenediol intake in young men. J Appl Physiol 92:142–146
Cabral-Marques H, Almeida R (2009) Optimisation of spray-drying process variables for dry powder inhalation (DPI) formulations of corticosteroid/cyclodextrin inclusion complexes. Eur J Pharm Biopharm 73(1):121–129. https://doi.org/10.1016/j.ejpb.2009.05.002
Cai W, Sun T, Liu P, Chipot C, Shao X (2009) Inclusion mechanism of steroid drugs into β-cyclodextrins. Insights from free energy calculations. J Phys Chem B 113(22):7836–7843. https://doi.org/10.1021/jp901825w
Caira MR, Bourne SA, Samsodien H, Smith VJ (2015) Inclusion complexes of 2-methoxyestradiol with dimethylated and permethylated β-cyclodextrins: models for cyclodextrin–steroid interaction. Beilstein J Org Chem 11:2616–2630
Cameron KS, Fletcher D, Fielding L (2002) An NMR study of cyclodextrin complexes of the steroidal neuromuscular blocker drug rocuronium bromide. Magn Reson Chem 40:251–260. https://doi.org/10.3762/bjoc.11.281
Carron M, Zarantonello F, Tellaroli P, Ori C (2016) Efficacy and safety of sugammadex compared to neostigmine for reversal of neuromuscular blockade: a meta-analysis of randomized controlled trials. J Clin Anesth 35:1–12. https://doi.org/10.1016/j.jclinane.2016.06.018
Cavalli R, Peira E, Caputo O, Gasco MR (1999) Solid lipid nanoparticles as carriers of hydrocortisone and progesterone complexes with.β-cyclodextrins. Int J Pharm 182(1):59–69. https://doi.org/10.1016/s0378-5173(99)00066-6
Ceschel GC, Berlati F, Ronchi C, Porziotta E (1993) Determination of the release of corticosteroids in topical formulations. Boll Chim Farm 132(2):49–50
Chang SL, Banga AK (1998) Transdermal iontophoretic delivery of hydrocortisone from cyclodextrin solutions. J Pharm Pharmacol 50(6):635–640. https://doi.org/10.1111/j.2042-7158.1998.tb06897.x
Chun IK, Kim BY (1992) Influence of various cyclodextrins on the stability of hydrocortisone 17-butyrate in aqueous solution. Arch Pharm Res 15(2):176–183. https://doi.org/10.1007/bf02974095
Cooper A, Nutley M, MacLean EJ, Cameron K, Fielding L, Mestres J, Palin R (2005) Mutual induced fit in cyclodextrin-rocuronium complexes. Org Biomol Chem 3(10):1863–1871. https://doi.org/10.1039/B415903A
Cramer F, Henglein FM (1957) Regularities of formation of cyclodextrin inclusion complexes. Chem Ber 90:2561–2571
Crini G (2014) Review: a History of cyclodextrins. Chem Rev 114(21):10940–10975. https://doi.org/10.1021/cr500081p
daSilveira AM, Duchene D, Ponchel G (2000) Influence of solubility and partition coefficient on the loading of combined poly(isobutylcyanoacrylate) and hydroxypropyl-beta-cyclodextrin nanoparticles by steroids. STP Pharma Sci 10(4):309–314
de Boer HD, van Egmond J, Driessen JJ, Booij LHD (2007) Update on the management of neuromuscular block: focus on sugammadex. Neuropsychiatr Dis Treat 3(5):539–544
de Carvalho LB, Burusco KK, Jaime C, Venancio T, Carvalho AFS, Murgas LDS, Pinto LDMA (2018) Complexes between methyltestosterone and β-cyclodextrin for application in aquaculture production. Carbohydr Polym 179:386–393. https://doi.org/10.1016/j.carbpol.2017.09.023
De Hassonville SH, Perly B, Piel G, Van Hees T, Barillaro V, Bertholet P, Delattre L, Evrard B (2003) Inclusion complexes of cyproterone acetate with cyclodextrins in aqueous solution. J Incl Phenom Macrocycl Chem 44(1–4):289–292
Dietzel K, Estes KS, Brewster ME, Bodor NS, Derendorf H (1990) The use of 2-hydroxypropyl-β-cyclodextrin as a vehicle for intravenous administration of dexamethasone in dogs. Int J Pharm 59(3):225–230. https://doi.org/10.1016/0378-5173(90)90113-i
Estes KS, Brewster ME, Webb AI, Bodor N (1990) A non-surfactant formulation for alfaxalone based on an amorphous cyclodextrin: activity studies in rats and dogs. Int J Pharm 65(1–2):101–107. https://doi.org/10.1016/0378-5173(90)90014-U
Fenyvesi E, Szente L (2016) Cyclodextrin-enabled novel waste water treatment technologies: from the test tube to the waste water treatment plant. Magyar Kémiai Folyóirat 122(2–4):83–89. http://www.mkf.mke.org.hu/images/stories/docs/2016_234a/MKF_2016_83.pdf. Accessed 20 Mar 2017)
Fenyvesi É, Vikmon MA, Szente L (2016) Cyclodextrins in food technology and human nutrition: benefits and limitations. Crit Rev Food Sci Nutr 56:1981–2004. https://doi.org/10.1080/10408398.2013.809513
Fenyvesi É, Puskás I, Szente L (2018) Cyclodextrin-steroid interactions and applications to pharmaceuticals, food, biotechnology and environment. In: Lichtfouse E, Fourmentin S, Crini G (eds) Environmental chemistry for a sustainable world, vol 7. Springer, Berlin, pp 19–57
Filipovic-Grcic J, Voinovich D, Moneghini M, Becirevic-Lacan M, Magarotto L, Jalsenjak I (2000) Chitosan microspheres with hydrocortisone and hydrocortisone-hydroxypropyl-β-cyclodextrin inclusion complex. Eur J Pharm Sci 9(4):373–379. https://doi.org/10.1016/S0928-0987(99)00078-0
Flood KG, Reynolds ER, Snow NH (2000) Characterization of inclusion complexes of betamethasone-related steroids with cyclodextrins using high-performance liquid chromatography. J Chromatogr A 903(1–2):49–65. https://doi.org/10.1016/S0021-9673(00)00867-0
Ford MM, Nickel JD, Finn DA (2005) Treatment with and withdrawal from finasteride alter ethanol intake patterns in male C57BL/6 J mice: potential role of endogenous neurosteroids. Alcohol (New York) 37(1):23–33. https://doi.org/10.1016/j.alcohol.2005.11.002
Forgo P, Gondos Gy (2002) A study of β-cyclodextrin inclusion complexes with progesterone and hydrocortisone using rotating frame Overhauser spectroscopy. Monat Chem 133(1):101–106. https://doi.org/10.1007/s007060270011
Fulop Z, Nielsen TT, Larsen KL, Loftsson T (2013) Dextran-based cyclodextrin polymers: their solubilizing effect and self-association. Carbohydr Polym 97(2):635–642. https://doi.org/10.1016/j.carbpol.2013.05.053
Gruiz K, Molnár M, Fenyvesi E, Hajdu C, Atkári Á, Barkács K (2011) Cyclodextrins in innovative engineering tools for risk-based environmental management. J Incl Phenom Macrocycl Chem 70(3–4):299–306. https://doi.org/10.1007/s10847-010-9909-y
Habon I, Stadler-Szoke A, Szejtli J (1985) Cyclodextrin inclusion complexes of steroids. Magy Kem Lapja 40(5):231–236 (in Hungarian)
Hashem FM, Halim A, Ghanem E, El-Leithy S, Shaker DS (2003) Effect of cyclodextrins on the solubility and in vitro permeation of corticosterone through hairless mouse skin. Bull Fac Pharm (Cairo Univ) 41(2):117–129
Helm H, Andersen A, Müller BW, Waaler T (1994) Stabilization of digitalis glycosides through 2-hydroxypropyl-β-cyclodextrin complexation in aqueous solution at pH 7.3. Pharmazie 49(7):494–496
Hermens WAJJ, Deurloo MJM, Romeyn SG, Verhoef JC, Merkus FWHM (1990) Nasal absorption enhancement of 17β-estradiol by dimethyl-β-cyclodextrin in rabbits and rats. Pharm Res 7:500–503
Hesselink PGM, van Vliet S, De Vries H, Witholt B (1989) Optimization of steroid side chain cleavage by mycobacterium sp. in the presence of cyclodextrins. Enzyme Microb Technol 11:398–404
Higuchi T, Connors KA (1965) Phase-solubility techniques. Adv Anal Chem Instrum 4:117–212
Holbrook WP, Kristmundsdottir T, Loftsson T (1998) Aqueous hydrocortisone mouthwash solution: clinical evaluation. Acta Odontol Scand 56(3):157–160
Hunter JM, Flockton E (2006) The doughnut and the hole: a new pharmacological concept for anaesthetists. Br J Anaesth 97:123–126. https://doi.org/10.1093/bja/ael158
Jadhav GS, Vavia PR (2008) Physicochemical, in silico and in vivo evaluation of a danazol-beta-cyclodextrin complex. Int J Pharm 352(1–2):5–16. https://doi.org/10.1016/j.ijpharm.2007.10.005
Jadhav GS, Vavia PR, Nandedkar TD (2007) Danazol-beta-cyclodextrin binary system: a potential application in emergency contraception by the oral route. AAPS Pharm Sci Tech 8(2):35. https://doi.org/10.1208/pt0802035
Jadoun J, Bar R (1993) Microbial transformations in a cyclodextrin medium. Part 3. Cholesterol oxidation by Rhodococcus Erythropolis. Appl Microbiol Biotechnol 40(2):230–240. https://doi.org/10.1007/BF00170372
Jansook P, Loftsson T (2009) CDs as solubilizers: effects of excipients and competing drugs. Int J Pharm 379(1):32–40. https://doi.org/10.1016/j.ijpharm.2009.06.005
Jansook P, Moya-Ortega MD, Loftsson T (2010) Effect of self-aggregation of gamma-cyclodextrin on drug solubilization. J Incl Phenom Macrocycl Chem 68(1–2):229–236. https://doi.org/10.1007/s10847-010-9779-3
Jiang L, Liu Y, Liu S, Li M, Hu X, Zeng G, Hu X, Liu S, Liu S, Huang B, Li M (2016) Fabrication of β-cyclodextrin/poly (l-glutamic acid) supported magnetic graphene oxide and its adsorption behavior for 17β-estradiol. Chem Eng J 308:597–605. https://doi.org/10.1016/j.cej.2016.09.067
Jover A, Budal RM, Al-Soufi W, Meijide F, Vazquez Tato J, Yunes RA (2003) Spectra and structure of complexes formed by sodium fusidate and potassium helvolate with beta- and gamma-cyclodextrin. Steroids 68(1):55–64. https://doi.org/10.1016/s0039-128x(02)00115-0
Kata M, Haragh L (1981) Spray embedding of spironolactone with β-cyclodextrin. Pharmazie 36(11):784–785
Kear CL, Yang J, Godwin DA, Felton LA (2008) Investigation into the mechanism by which cyclodextrins influence transdermal drug delivery. Drug Dev Ind Pharm 34(7):692–697. https://doi.org/10.1080/03639040701842428
Keating GM (2016) Sugammadex: a review of neuromuscular blockade reversal. Drugs 76(10):1041–1052. https://doi.org/10.1007/s4026510016-0604-1
Khomutov SM, Donova MV (2011) Nanodimer cyclodextrin ligands with high affinity to steroids. J Incl Phenom Macrocycl Chem 70(3):353–357. https://doi.org/10.1007/s10847-010-9896-z
Khomutov SM, Sukhodolskaya GV, Donova MV (2007) The inhibitory effect of cyclodextrin on the degradation of 9alpha-hydroxyandrost-4-ene-3, 17-dione by Mycobacterium sp. VKM Ac-1817D. Biocatal Biotransform 25(5):386–392. https://doi.org/10.1080/10242420701510510
Koizumi K, Okada Y, Kubota Y, Utamura T (1987) Inclusion complexes of poorly water-soluble drugs with glucosyl-cyclodextrins. Chem Pharm Bull 35:3413–3418. https://doi.org/10.1248/cpb.35.3413
Kompantseva EV, Gavrilin MV, Botezat-Belyi YuK, Umnova EF, Andronik IY (1990) Investigation of β-cyclodextrin-cortexolone interaction. Khim Farm Zh 24(10):81–82
Kralova K, Mitterhauszerova L (1989) Interactions of β-cyclodextrin with steroid compounds in aqueous solutions. Pharmazie 44(9):623–625
Kristinsson JK, Fridriksdottir H, Thorisdottir S, Sigurdardottir AM, Stefansson E, Loftsson T (1996) Dexamethasone-cyclodextrin-polymer co-complexes in aqueous eye drops. Invest Ophthalmol Vis Sci 37:1199–1203
Kristmundsdottir T, Loftsson T, Holbrook WP (1996) Formulation and clinical evaluation of a hydrocortisone solution for the treatment of oral disease. Int J Pharm 139(1,2):63–68. https://doi.org/10.1016/0378-5173(96)04565-6
Lahiani-Skiba M, Barbot C, Bounoure F, Joudieh S, Skiba M (2006) Solubility and dissolution rate of progesterone-cyclodextrin-polymer systems. Drug Dev Ind Pharm 32(9):1043–1058. https://doi.org/10.1080/03639040600897093
Landy D, Mallard I, Ponchel A, Monflier E, Fourmentin S (2012) Remediation technologies using cyclodextrins: an overview. Environ Chem Lett 10:225–237. https://doi.org/10.1007/s10311-011-0351-1
Larsen KL, Aachmann FL, Wimmer R, Stella VJ, Kjolner UM (2005) Phase solubility and structure of the inclusion complexes of prednisolone and 6alpha-methyl prednisolone with various cyclodextrins. J Pharm Sci 94(3):507–515
Larsen KL, Andersen SB, Morbak AL, Wimmer R (2007) Inclusion complexes of fusidic acid and three structurally related compounds with cyclodextrins. J Incl Phenom Macrocycl Chem 57:185–190. https://doi.org/10.1007/s10847-006-9198-7
Lee JC, Seo MS, Kim SR (2012) A beta-cyclodextrin, polyethyleneimine and silk fibroin hydrogel containing Centella asiatica extract and hydrocortisone acetate: releasing properties and in vivo efficacy for healing of pressure sores. Clin Exp Dermatol 37(7):762–771. https://doi.org/10.1111/j.1365-2230.2011.04331.x
Lee LL, Athanassoglou V, Pandit JJ (2016) Neuromuscular blockade in the elderly patient. J Pain Res 9:437–444. https://doi.org/10.2147/JPR.S85183
Leigh HM, Meister PD, Weintraub A, Reineke LM, Eppstein SH, Murray HC, Peterson DH (1952) Microbiological transformations of steroids. I. Introduction of oxygen at carbon-11 of progesterone. J Am Chem Soc 73(23):5933–5936. https://doi.org/10.1021/ja01143a033
Liu FY, Kildsig DO, Mitra AK (1990) Beta-cyclodextrin/steroid complexation: effect of steroid structure on association equilibria. Pharm Res 7(8):869–873
Liu FY, Kildsig DO, Mitra AK (1992) Complexation of 6-acyl-O-β-cyclodextrin derivatives with steroids-effects of chain length and substitution degree. Drug Dev Ind Pharm 18(15):1599–1612. https://doi.org/10.3109/03639049209040889
Liu J, Wang L, Shen Y, Liqiu S, Wang M (2018) Effect of β-cyclodextrins derivatives on steroids biotransformation by arthrobacter simplex. Appl Biochem Biotechnol 185(4):1004–1013. https://doi.org/10.1007/s12010-018-2704-8
Loftsson T (2014) Self-assembled cyclodextrin nanoparticles and drug delivery. J Incl Phenom Macrocycl Chem 80:1–7. https://doi.org/10.1007/s10847-013-0375-1
Loftsson T, Siguroardottir AM (1994) The effect of poly(vinylpyrrolidone) and hydroxypropyl methyl cellulose on HPβCD complexation of hydrocortisone and its permeability through hairless mouse skin. Eur J Pharm Sci 2(4):30–297. https://doi.org/10.1016/0928-0987(94)90013-2
Loftsson T, Stefansson E (2002) Cyclodextrins in eye drop formulations: enhanced topical delivery of corticosteroids to the eye. Acta Ophthalmol Scand 80(2):144–150. https://doi.org/10.1034/j.1600-0420.2002.800205.x
Loftsson T, Olafsdottir BJ, Bodor N (1991) The effects of cyclodextrins on transdermal delivery of drugs. Eur J Pharm Biopharm 37:30–33. https://doi.org/10.1002/jps.20992
Loftsson T, Frioriksdottir H, Ingvarsdottir G, Jonsdottir B, Siguroardottir AM (1994a) The influence of 2-hydroxypropyl-β-cyclodextrin on diffusion rates and transdermal delivery of hydrocortisone. Drug Dev Ind Pharm 20(9):1699–1708. https://doi.org/10.3109/03639049409050210
Loftsson T, Fridriksdottir H, Thorisdottir S, Stefansson E (1994b) The effect of hydroxypropyl methylcellulose on release of dexamethazone from aqueous 2-hydroxypropyl-b-cyclodextrin formulations. Int J Pharm 104:181–184
Loftsson T, Masson M, Sigurdsson HH (2002) Cyclodextrins and drug permeability through semi-permeable cellophane membranes. Int J Pharm 232(1–2):35–43. https://doi.org/10.1016/S0378-5173(01)00895-X
Lohade AA, Singh DJ, Parmar JJ, Hegde DD, Menon MD, Soni PS, Samad A, Gaikwad RV (2007) Albumin microspheres of fluticasone propionate inclusion complexes for pulmonary delivery. Indian J Pharm Sci 69(5):707–709. https://doi.org/10.4103/0250-474X.38486
Lopata A, Darvas F, Stadler-Szoke A, Szejtli J (1985) Quantitative structure-stability relationships among inclusion complexes of cyclodextrins. Part 2. Steroid hormones. In: Seydel JK (ed) QSAR strategies Des Bioact Compd, proceedings European symposium quantitative structure-activity relationship, 5th, Meeting Date 1984, VCH, Weinheim, pp 353–6
Luthra U, Khadpekar S, Trivedi A, Shetty A, Kumar H (2015) Biotransformation of 4-androstene-3, 17-dione to androst-1, 4-diene-3, 17-dione by Nocardioides Simplex. WJPPS 4(11):1935–1943
Ma YH, Wang M, Fan Z, Shen YB, Zhang LT (2009) The influence of host-guest inclusion complex formation on the biotransformation of cortisone acetate 1-dehydrogenation. J Steroid Biochem Mol Biol 117(4–5):146–151. https://doi.org/10.1016/j.jsbmb.2009.08.007
Masson M, Loftsson T, Masson G, Stefansson E (1999) Cyclodextrins as permeation enhancers: some theoretical evaluations and in vitro testing. J Contr Release 59(1):107–118. https://doi.org/10.1016/S0168-3659(98)00182-5
Miro A, d’Angelo I, Nappi A, La Manna P, Biondi M, Mayol L, Musto P, Russo R, Rotonda MIL, Ungaro F, Quaglia F (2013) Engineering poly(ethylene oxide) buccal films with cyclodextrin: a novel role for an old excipient? Int J Pharm 452:283–291. https://doi.org/10.1016/j.ijpharm.2013.05.030
Moellgaard AF, Bundgaard H (1983) Inclusion complexation of spironolactone with cyclodextrins. Arch Pharm Chem Sci Ed 11(1):7–14
Müller BW, Brauns U (1986) Hydroxypropyl-β-cyclodextrin derivatives: influence of average degree on substitution on complexing ability and surface activity. J Pharm Sci 75(6):571–572. https://doi.org/10.1002/jps.2600750609
Nagy ZM, Molnár M, Fekete-Kertész I, Molnár-Perl I, Fenyvesi É, Gruiz K (2014) Removal of emerging micropollutants from water using cyclodextrin. Sci Total Environ 485–486:9–711. https://doi.org/10.1016/j.scitotenv.04.003
Oishi K, Moriuchi A (2010) Removal of dissolved estrogen in sewage effluents by beta-cyclodextrin polymer. Sci Total Environ 409(1):112–115. https://doi.org/10.1016/j.scitotenv.2010.09.031
Okada Y, Koizumi K (1998) Some properties and the inclusion behavior of three positional isomers of 61,6nDi-O-α-D-glucosyl-cyclomaltoheptaoses (β-cyclodextrins). Chem Pharm Bull 46(2):319–323
Pike MC, Spicer DV, Daniels A, Daniels JR (2004) Nasal spray steroid formulation and method. US. Patent Application Publications US 2004022738
Pitha J, Pitha J (1985) Amorphous water-soluble derivatives of cyclodextrins: nontoxic dissolution enhancing excipients. J Pharm Sci 74(9):987–990. https://doi.org/10.1002/jps.2600740916
Pitha J, Harman SM, Michel ME (1986) Hydrophilic cyclodextrin derivatives enable effective oral administration of steroidal hormones. J Pharm Sci 75(2):165–167. https://doi.org/10.1002/jps.2600750213
Pitha J, Anaissie EJ, Uekama K (1987) Gamma-cyclodextrin:testosterone complex suitable for sublingual administration. J Pharm Sci 76:788–790. https://doi.org/10.1002/jps.2600761007
Pitha J, Hoshino T, Torres-Labandeira J, Irie T (1992) Preparation of drug-hydroxypropyl cyclodextrin complexes by a method using ethanol or aqueous ammonium hydroxide as cosolubilizers. Int J Pharm 80(2–3):253–258. https://doi.org/10.1016/0378-5173(92)90282-7
Podhaisky HP, Bracht S (2007) Pharmaceutical films for oral use. PCT Int. Appl. WO 2007085498
Preiss A, Mehnert W, Froemming KH (1994) Complexation of hydrocortisone with β-cyclodextrin and hydroxypropyl-β-cyclodextrin. Arch Pharm (Weinh Ger) 327(11):729–734
Radu CD, Salariu M, Avadanei M, Ghiciuc C, Foia L, Lupusoru EC, Ferri A, Ulea E, Lipsa F (2013) Cotton-made cellulose support for anti-allergic pajamas. Carbohydr Polym 95:479–486. https://doi.org/10.1016/j.carbpol.2013.03.037
Rodriguez-Tenreiro C, Alvarez-Lorenzo C, Rodriguez-Perez A, Concheiro A, Torres-Labandeira JJ (2007) Estradiol sustained release from high affinity cyclodextrin hydrogels. Eur J Pharm Biopharm 66(1):55–62. https://doi.org/10.1016/j.ejpb.2006.09.003
Roglič U, Žnidaršič-Plazl P, Plazl I (2005) The Influence of β-cyclodextrin on the kinetics of progesterone transformation by Rhizopus Nigricans. Biocatal Biotransform 23(5):299–305. https://doi.org/10.1080/10242420500175929
Salapatek AM, Lee J, Patel D, D’Angelo P, Liu JH, Zimmerer RO, Pipkin JD (2011) Solubilized nasal steroid (CDX-947) when combined in the same solution nasal spray with an antihistamine (CDX-313) provides improved, fast-acting symptom relief in patients with allergic rhinitis. Allergy Asthma Proc 32(3):221–229. https://doi.org/10.2500/aap.2011.32.3444
Salehian B, Wang C, Alexander G, Davidson T, McDonald V, Berman N, Dudley RE, Ziel F, Swerdloff RS (1995) Pharmacokinetics, bioefficacy, and safety of sublingual testosterone cyclodextrin in hypogonadal men: comparison to testosterone enanthate–a clinical research center study. J Clin Endocrinol Metab 80(12):3567–3575. https://doi.org/10.1210/jcem.80.12.8530600
Schaller SJ, Fink H (2013) Sugammadex as a reversal agent for neuromuscular block: an evidence-based review. Core Evid 8:57–67. https://doi.org/10.2147/CE.S35675
Schipper NGM, Hermens WAJJ, Romeyn SG, Verhoef J, Merkus FWHM (1990) Nasal absorption of 17β-estradiol and progesterone from a dimethyl cyclodextrin inclusion formulation in rats. Int J Pharm 64(1):61–66. https://doi.org/10.1016/0378-5173(90)90179-8
Schwartz DH, Engelke A, Wenz G (2017) Solubilizing steroidal drugs by b-cyclodextrin derivatives. Int J Pharm 531:559–567. https://doi.org/10.1016/j.ijpharm.2017.07.046
Seo H, Uekama K (1989) Enhanced bioavailability of digoxin by gamma-cyclodextrin complexation: evaluation for sublingual an d oral administrations in humans. J Pharm Soc Jpn 109(10):778–782
Shaker DS, Ghanem AH, Li SK, Warner KS, Hashem FM, Higuchi WI (2003) Mechanistic studies of the effect of hydroxypropyl-beta-cyclodextrin on in vitro transdermal permeation of corticosterone through hairless mouse skin. Int J Pharm 253(1–2):1–11. https://doi.org/10.1016/S0378-5173(02)00625-7
Shen Y, Ma B, Zheng Y, Luo J, Zhi Y, Wang M (2010) The mechanism of β-cyclodextrin on the 11β-hydroxylation biotransformation of steroid. In: Proceedings of 3rd international conference biomedical engineering and informatics, BMEI 2010, Yantai, 5, 5639462, pp 1964–1967
Shi JH, Chen K, Xu J (2014) Characterization of the inclusion interaction between prednisolone and di-O-methyl-beta-cyclodextrin: spectroscopic methods and molecular modeling. J Mol Liq 194:172–178. https://doi.org/10.1016/j.molliq.2014.01.023
Shimada K, Oe T, Hirose Y, Komine Y (1989) Retention behavior of cardiac steroids using cyclodextrin in the mobile phase in high-performance liquid chromatography. J Chromatogr 478(2):339–347
Shinoda T, Kagatani S, Maeda A, Konno Y, Hashimoto H, Hara K, Fujita K, Sonobe T (1999) Sugar-branched-cyclodextrins as injectable drug carriers in mice. Drug Dev Ind Pharm 25(11):1185–1192. https://doi.org/10.1081/DDC-100102286
Sigma-Aldrich (2017) http://www.sigmaaldrich.com/catalog/product/sigma/e4389 Accessed 13 Mar 2017
Sigurdardottir AM, Loftsson T (1995) The effect of polyvinylpyrrolidone on cyclodextrin complexation of hydrocortisone and its diffusion through hairless mouse skin. Int J Pharm 126(1,2):73–78. https://doi.org/10.1016/0378-5173(95)04095-1
Singer Y, Shity H, Bar R (1991) Microbial transformations in a cyclodextrin medium. Part 2. Reduction of androstenedione to testosterone by Saccharomyces Cerevisiae. Appl Microbiol Biotechnol 35(6):731–737. https://doi.org/10.1007/BF00169886
Sorgenfrei IF, Norrild K, Larsen PB, Stensballe J, Ostergaard D, Prins ME, Viby-Mogensen J (2006) Reversal of rocuronium-induced neuromuscular block by the selective relaxant binding agent sugammadex: a dose-finding and safety study. Surv Anesthesiol 50(6):286–287. https://doi.org/10.1097/01.sa.0000238953.66744.e7
Stefansson E, Loftsson T (2003) Cyclodextrins in eye drop formulations. J Incl Phenom Macrocycl Chem 44(1–4):23–27. https://doi.org/10.1034/j.1600-0420.2002.800205.x
Stünkel CA, Dudley RE, Yen SS (1991) Sublingual administration of testosterone-hydroxypropyl-beta-cyclodextrin inclusion complex simulates episodic androgen release in hypogonadal men. J Clin Endocrinol Metab 72(5):1054–1059. https://doi.org/10.1210/jcem-72-5-1054
Szejtli J (1982) Cyclodextrins and their inclusion complexes. Akadémiai Kiadó, Budapest
Szejtli J (1986) Cyclodextrins in biotechnology. Starch/Staerke 38:388–390. https://doi.org/10.1002/star.19860381107
Szejtli J (1988) Cyclodextrin technology. Kluwer, Alphen aan den Rijn
Szejtli J (1990) The cyclodextrins and their applications in biotechnology. Carbohydr Polym 12(4):375–392. https://doi.org/10.1016/0144-8617(90)90088-A
Szejtli J (1998) Ciklodextrinek alkalmazása vegyipari termékekben és eljárásokban. (Application of cyclodextrins in chemical industrial products and technologies, in Hungarian). Magy Kém Lapja 53(6):271–282
Szente L (2006) Cyclodextrins in anesthesiology. Cyclodext News 20(10):265–269
Szente L, Fenyvesi É (2017) Cyclodextrin-lipid complexes: cavity size matters. Struct Chem 28(2):479–492. https://doi.org/10.1007/s11224-016-0884-9
Taylor GT, Weiss J, Pitha J (1989) Testosterone in a cyclodextrin-containing formulation: behavioral and physiological effects of episode-like pulses in rats. Pharm Res 6:641–646
Thi TD, Nauwelaerts K, Froeyen M, Baudemprez L, Van Speybroeck M, Augustijns P, Annaert P, Martens J, Van Humbeeck J, Van den Mooter G (2010) Comparison of the complexation between methylprednisolone and different cyclodextrins in solution by 1H-NMR and molecular modeling studies. J Pharm Sci 99(9):3863–3873. https://doi.org/10.1002/jps.22227
Tongiani S, Ozeki T, Stella VJ (2009) Sulfobutyl ether-alkyl ether mixed cyclodextrin derivatives with enhanced inclusion ability. J Pharm Sci 98(12):4769–4780. https://doi.org/10.1002/jps.21791
Udvardy NI, Bartho I, Hantos G, Trinn M, Vida Z, Szejtli J, Stadler I, Habon I, Balázs Á (1981). Process for the intensification of the microbiological transformation of steroid compounds. Hungarian patent application HU19810002785
Uekama K, Fujinaga T, Otagiri M, Seo H, Tsuruoka M (1981) Enhanced bioavailability of digoxin by.gamma-cyclodextrin complexation. J Pharmacobiodyn 4(9):735–737
Uekama K, Fujinaga T, Hirayama F, Otagiri M, Yamasaki M (1982a) Inclusion complexations of steroid hormones with cyclodextrins in water and in solid phase. Int J Pharm 10(1):1–15. https://doi.org/10.1016/0378-5173(82)90057-6
Uekama K, Fujinaga T, Hirayama F, Otagiri M, Kurono Y, Ikeda K (1982b) Effects of cyclodextrins on the acid hydrolysis of digoxin. J Pharm Pharmacol 34(10):627–630. https://doi.org/10.1111/j.2042-7158.1982.tb04690.x
Uekama K, Otagiri M, Uemura Y, Fujinaga T, Arimori K, Matsuo N, Tasaki K, Sugii A (1983) Improvement of oral bioavailability of prednisolone by β-cyclodextrin complexation in humans. J Pharmacobiodyn 6(2):124–127
Uekama K, Otagiri M, Sakai A, Irie T, Matsuo N, Matsuoka Y (1985) Improvement in the percutaneous absorption of beclomethasone dipropionate by γ-cyclodextrin complexation. J Pharm Pharmacol 37(8):532–535. https://doi.org/10.1111/j.2042-7158.1985.tb03061.x
Vardanyan R, Hruby V (2016) Synthesis of best-seller drugs. Academic press, Cambridge
Vianna RFL, Bentley MVLB, Ribeiro G, Carvalho FS, Neto AF, de Oliveira DCR, Collett JH (1998) Formation of cyclodextrin inclusion complexes with corticosteroids: their characterization and stability. Int J Pharm 167(1–2):205–213. https://doi.org/10.1016/S0378-5173(98)00068-4
Volkova DA, Bannikova GE, Lopatin SA, Il’in MM, Gabinskaya KN, Andryushina VA, Gracheva IM, Varlamov VP (1999) Effect of a β-cyclodextrin-epichlorohydrin copolymer on the solubility of cortexolone. Pharm Chem J 33(11):601–603
Werner U, Damge C, Maincent P, Bodmeier R (2004) Properties of in situ gelling nasal inserts containing estradiol/methyl beta-cyclodextrin. J Drug Deliv Sci Technol 14(4):275–284. https://doi.org/10.1016/S1773-2247(04)50048-5
Wintgens V, Amiel C (2010) Water-soluble γ-cyclodextrin polymers with high molecular weight and their complex forming properties. Eur Polym J 46(9):1915–1922. https://doi.org/10.1016/j.eurpolymj.2010.06.014
Won YJ, Lim BG, Lee DK, Kim H, Kong MH, Lee IO (2016) Sugammadex for reversal of rocuronium-induced neuromuscular blockade in pediatric patients: a systematic review and meta-analysis. Medicine (Baltimore) 95(34):e4678. https://doi.org/10.1097/MD.0000000000004678
Wördenbag HJ, Pras N, Frijlink HW, Lerk CF, Malingre TM (1990) Cyclodextrin-facilitated bioconversion of 17 β-estradiol by a phenoloxidase from mucuna pruriens cell cultures. Phytochem 29(5):1551–1554. https://doi.org/10.1016/0031-9422(90)80119-2
Wouessidjewe D, Crassous A, Duchene D, Coleman A, Rysanek N, Tsoucaris G, Perly B, Djedaini F (1989) Inclusion of spironolactone in cyclomaltoheptaose: a guest affected by the hospitality of the host. Carbohydr Res 192:313–321. https://doi.org/10.1016/0008-6215(89)85189-4
Znidarsic-Plazl P, Plazl I (2010) Development of a continuous steroid biotransformation process and product extraction within microchannel system. Catal Today 157(1–4):315–320. https://doi.org/10.1016/j.cattod.2010.01.042
Zwiers A, van den Heuvel M, Smeets J, Rutherford S (2011) Assessment of the potential for displacement interactions with sugammadex.A pharmacokinetic-pharmacodynamic modelling approach. Clin Drug Investig 31(2):101–111. https://doi.org/10.2165/11584730-000000000-00000
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fenyvesi, É., Puskás, I. & Szente, L. Applications of steroid drugs entrapped in cyclodextrins. Environ Chem Lett 17, 375–391 (2019). https://doi.org/10.1007/s10311-018-0807-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-018-0807-7