Abstract
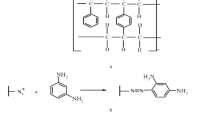
The present study deals with the preconcentration and determination of toxic metal ions using p-tetranitrocalix[4]arene (3) appended silica-based new HPLC column. The synthesized material was characterized using Fourier transform infrared and scanning electron microscopy techniques. The sorption characteristics of the HPLC column were investigated for three toxic metals (Cd2+, Hg2+ and Pb2+) in column agreement. The experiments were performed in five steps that were monitored using a UV–visible diode-array detector. However, all the HPLC experimental results were reconfirmed by using atomic absorption spectrophotometer. The effect of concentration on the sorption efficiency of the column was evaluated for all the three metals and the data obtained were investigated using Langmuir, Freundlich and Dubinin–Redushkevich (D–R) sorption isotherms. The value of coefficient of determination (R 2), i.e. 0.99, suggested that the Freundlich sorption isotherm was found to be the best-fit model for all the three toxic metal ions, whereas, mean free energy was calculated from the D–R isotherm as 5.3, 5.7, and 5.8 kJ/mol for Pb2+, Cd2+, and Hg2+, respectively; suggesting that physical electrostatic force is involved in the sorption process. The developed method was validated for system efficiency, accuracy, and precision.









Similar content being viewed by others
References
Santoyo E, Santoyo-Gutierrez S, Verma SP (2000) Trace analysis of heavy metals in groundwater samples by ion chromatography with post-column reaction and ultraviolet–visible detection. J Chrom A 884:229–241
Kara H, Ayyildiz HF, Topkafa M (2008) Use of aminoprophyl silica-immobilized humic acid for Cu(II) ions removal from aqueous solution by using a continuously monitored solid phase extraction technique in a column arrangement. Coll Surf A 312:62–72
Miretzkya P, Cirelli AF (2009) Hg(II) removal from water by chitosan and chitosan derivatives: a review. J Hazard Mater 167:10–23
Sheng PX, Ting YP, Chen JP, Hong L (2004) Sorption of lead, copper, cadmium, zinc, and nickel by marine algal biomass: characterization of biosorptive capacity and investigation of mechanisms. J Colloid Interf Sci 275:131–141
Vonderheide PA, Meija J, Tepperman K, Puga A, Pinhas AR, States JC, Caruso JA (2004) Retention of Cr(III) by high-performance chelation ion chromatography interfaced to inductively-coupled plasma mass spectrometric detection with collision cell. J Chrom A 1024:129–137
Dermentzis K, Christoforidis A, Valsamidou E, Lazaridou A, Kokkinos N (2011) Removal of hexavalent chromium from electroplating wastewater by electrocoagulation with iron electrodes. Glob Nest J 13:412–418
Barbooti MM, Ageena NA, Tooma MA (2008) Removal of chromium from electroplating wastewater by simple chemical treatment and ion exchange. Eng Tech 26:1–9
Ganeshjeevan R, Chandrasekar R, Yuvaraj S, Radhakrishnan G (2003) Determination of hexavalent chromium by On-line dialysis ion chromatography in a matrix of strong colourants and trivalent chromium. J Chrom A 988:157–165
Ersoz M (2007) Transport of mercury through liquid membranes containing calixarene carriers. Adv Colloid Interface Sci 134:96–104
Durmaz F, Kara H (2010) α-Novel determination method of Cu(II) ions on the immobilized humic acid by SPE-FIA hyphenated technique. Rev Desalin 256:1–8
Gezici O, Tabakci M, Kara H, Yilmaz M (2006) Synthesis of p-tert-butylcalix[4]arene dinitrile bonded aminopropyl silica and investigating its usability as a stationary phase in HPLC. J Macro Sci Part A 43:221–231
Erdemir S, Yilmaz M (2010) Preparation of a new 1,3-alternate-calix[4]arene-bonded HPLC stationary phase for the separation of phenols, aromatic amines and drugs. Talanta 82(4):1240–1246
Solangi IB, Bhatti AA, Qazi MA, Memon S, Bhanger MI (2012) Selective cation recognition by p-tetranitrocalix[4]arene. Pak J Anal Environ Chem 13:129–136
Gutsche CD, Iqbal M, Stewart D (1986) Calixarenes. 18. Synthesis procedures for p-tert-Butylcalix[4]arene. J Org Chem 51:742
Gutsche CD, Lin LG (1986) Calixarenes. 12. The synthesis of functionalized calixarenes. Tetrahedron 42:1633–1644
Verboom W, Durie A, Egberink RJM, Asfari Z, Reinhoud DN (1992) Ipso nitration of p-tert -butylcalix[4]arenes. J Org Chem 57:1313–1316
Foo KY, Hameed BH (2010) Insights into the modeling of adsorption isotherm systems. Chem Eng J 156:2–10
Febrianto J, Kosasiha AN, Sunarso J, Ju YH, Indraswati N, Ismadji S (2009) Equilibrium and kinetic studies in adsorption of heavy metals using biosorbent. A summary of recent studies. J Hazard Mat 162:616–645
Acknowledgments
We would like to thank the National Centre of Excellence in Analytical Chemistry, University of Sindh, Jamshoro/Pakistan, and the Higher Education Commission (YÖK) of Turkey for the financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Durmaz, F., Memon, F.N., Memon, N.A. et al. Preconcentration and Determination of Toxic Metals by A New Calix[4]arene Bonded HPLC Column. Chromatographia 76, 909–919 (2013). https://doi.org/10.1007/s10337-013-2501-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-013-2501-z