Abstract
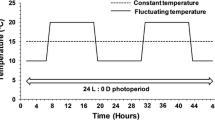
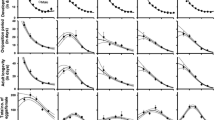
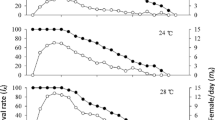
We focused on the influence of different temperature amplitudes on development and reproduction of the two-spotted spider mite, Tetranychus urticae Koch, at a 16:8 (L:D) h photoperiod and 60–95 % RH. The temperature amplitudes varied from 0 to 24 °C in steps of 6 °C; i.e. 22 ± 0, 22 ± 3, 22 ± 6, 22 ± 9 and 22 ± 12 °C. Temperature changed every 24 h between a low and an upper value, but without changing the average temperature (22 °C). The number of eggs laid by five females for 24 h was slightly lower at 22 ± 12 °C than at constant temperature (22 ± 0 °C), and egg hatchability differed among the five temperature regimes. Developmental time at 22 ± 0 °C was shorter than that at 22 ± 3 and 22 ± 6 °C, but longer than that at 22 ± 9 and 22 ± 12 °C. The oviposition period, total fecundity per female and adult longevity gradually decreased with increasing amplitudes. Sex ratio was similar at all five temperature regimes. The intrinsic rate of natural increase (r m) was affected by temperature amplitude and the r m-values at all amplitudes except 22 ± 12 °C were higher than that at constant temperature. Thus, this study showed that variable temperature regimes influence population growth rates of T. urticae and that large amplitude regimes are stressful for this species.





Similar content being viewed by others
References
Amarasekare P, Sifuentes R (2012) Elucidating the temperature response of survivorship in insects. Funct Ecol 26:959–968
Beck S (1983) Thermal and thermoperiodic effects on larval development and diapause in the European corn borer, Ostrinia nubilalis. J Insect Physiol 29:107–112
Birch LC (1948) The intrinsic rate of natural increase of an insect population. J Anim Ecol 17:15–26
Bolland HR, Gutierrez J, Flechtmann CHW (1998) World catalogue of the spider mite family (Acari: Tetranychidae). Leiden, Brill, p 392
Boosma JJ, Nachman G (2002) Analysis of sex ratios in social insects. In: Hardy ICW (ed) Sex ratios: concepts and research methods. Cambridge University Press, Cambridge, pp 93–111
Bounfour M, Tanigoshi L (2001) Effect of temperature on development and demographic parameters of Tetranychus urticae and Eotetranychus carpini borealis (Acari: Tetranychidae). Ann Entomol Soc Am 94:400–404
Bryant S, Bale J, Thomas C (1999) Comparison of development and growth of nettle-feeding larvae of Nymphalidae (Lepidoptera) under constant and alternating temperature regimes. Eur J Entomol 96:143–148
Caswell H (2001) Matrix population models: construction, analysis, and interpretation, 2nd edn. Sinauer Associates, Massachusetts, p 722
Chen C-Y, Chiu M-C, Kuo M-H (2013) Effect of warming with temperature oscillations on a low-latitude aphid, Aphis craccivora. Bull Entomol Res 103:406–413
Dang CK, Schindler M, Chauvet E, Gessner MO (2009) Temperature oscillation coupled with fungal community shifts can modulate warming effects on litter decomposition. Ecology 90:122–131
Dell AI, Pawar S, Savage VM (2011) Systematic variation in the temperature dependence of physiological and ecological traits. PNAS 108:10591–10596
Fantinou AA, Perdikis DCh, Chatzoglou CS (2003) Development of immature stages of Sesamia nonagrioides (Lepidoptera: Noctuidae) under alternating and constant temperatures. Environ Entomol 32:1337–1342
Gabre RM, Adham FK, Chi H (2005) Life table of Chrysomya megacephala (Fabricius) (Diptera: Calliphoridae). Act Oecol 27:179–183
Gotoh T (1986) Life-history parameters of the hawthorn spider mite, Tetranychus viennensis Zacher (Acarina: Tetranychidae), on deciduous oak. Appl Entomol Zool 21:389–393
Gotoh T, Sugimoto N, Pallini A, Knapp M, Hernandez-Suarez E, Ferragut F, Ho CC, Migeon A, Navajas M, Nachman G (2010) Reproductive performance of seven strains of the tomato red spider mite Tetranychus evansi (Acari: Tetranychidae) at five temperatures. Exp Appl Acarol 52:239–259
Hagstrum DW, Milliken GA (1991) Modeling differences in insect developmental times between constant and fluctuating temperatures. Ann Entomol Soc Am 84:369–379
Khanamani M, Fathipour Y, Hajiqanbar H (2013) Population growth response of Tetranychus urticae to eggplant quality: application of female age-specific and age-stage, two-sex life tables. Int J Acarol 39:638–648
Lawo JP, Lawo NC (2011) Misconceptions about the comparison of intrinsic rates of natural increase. J Appl Entomol 135:715–725
Manly BFJ (1990) Stage-structured populations: sampling analysis and simulation. Chapman and Hall, London, p 187
Margolies DC, Wrensch DL (1996) Temperature-induced changes in spider mite fitness: offsetting effects of development time, fecundity, and sex ratio. Entomol Exp Appl 78:111–118
McCullagh P, Nelder JA (1989) Generalized linear models. Monographs on statistics and applied probability, vol 37, 2nd edn. Chapman and Hall, Boca Raton, p 511
Migeon A, Dorkeld F (2006–2013) Spider mites web: a comprehensive database for the Tetranychidae. http://www.montpellier.inra.fr/CBGP/spmweb. Accessed 28 Feb 2014
Mironidis GK, Savopoulou-Soultani M (2008) Development, survivorship, and reproduction of Helicoverpa armigera (Lepidoptera: Noctuidae) under constant and alternating temperatures. Environ Entomol 37:16–28
Peto R, Pike MC (1973) Conservatism of the approximation Σ (O − E)2/E in the logrank test for survival data or tumor incidence data. Biometrics 29:579–584
Renault D, Nedvéd O, Hervant F, Vernon P (2004) The importance of fluctuating thermal regimes for repairing chill injuries in the tropical beetle Alphitobius diaperinus (Coleoptera: Tenebrionidae) during exposure to low temperature. Physiol Entomol 29:139–145
Sabelis MW (1985) Reproductive strategies. In: Helle W, Sabelis MW (eds) Spider mites: their biology, natural enemies and control. Elsevier, Amsterdam, pp 265–278
SAS (2006) SAS Enterprise Guide 4.1. SAS Institute Inc. SAS Campus Drive, Cary NC 27513
Siddiqui W, Barlow C, Randolph P (1973) Effects of some constant and alternating temperatures on population growth of the pea aphid, Acyrthosiphon pisum (Homoptera: Aphididae). Can Entomol 105:145–156
Snell TW (1978) Fecundity, developmental time, and population growth rate. Oecologia 32:119–125
Sokal RR, Rohlf FJ (1995) Biometry, 3rd edn. WH Freeman, New York, p 887
Southwood TRE, Henderson PA (2000) Ecological methods. Wiley, New York, p 592
SPSS (2009) Manual of PASW® Statistic Base 18. SPSS Japan Inc., Tokyo, p 331 (in Japanese)
Uchida M (1982) Ecological studies on the abundance and diapauses of spider mites and the damage caused by the spider mites in Japanese pear orchards. Spec Bull Tottori Tree Exp Stn 2:1–63 (in Japanese)
Valicente FH, O’Neil RJ (1995) Effects of host plants and feeding regimes on selected life history characteristics of Podisus maculiventris (Say) (Heteroptera: Pentatomidae). Biol Control 5:449–461
Vangansbeke D, Schrijver LD, Spranghers T, Audenaert J, Verhoeven R, Nguyen DT, Gobin B, Tirry L, Clereq PD (2013) Alternating temperatures affect life table parameters of Phytoseiulus persimilis, Neoseiulus californicus (Acari: Phytoseiidae) and their prey Tetranychus urticae (Acari: Tetranychidae). Exp Appl Acarol 61:285–298
Wrensch DL (1985) Reproductive parameters. In: Helle W, Sabelis MW (eds) Spider mites: their biology, natural enemies and control. Elsevier, Amsterdam, pp 165–170
Wyatt IJ, White PF (1977) Simple estimation of intrinsic increase rates for aphids and tetranychid mites. J Appl Ecol 14:757–766
Acknowledgments
We are grateful to Dr. Y. Kitashima, Dr. T. Matsuda and Mr. R. Sugawara, Ibaraki University, for their kind help in this research.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gotoh, T., Saito, M., Suzuki, A. et al. Effects of constant and variable temperatures on development and reproduction of the two-spotted spider mite Tetranychus urticae (Acari: Tetranychidae). Exp Appl Acarol 64, 465–478 (2014). https://doi.org/10.1007/s10493-014-9841-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-014-9841-8