Abstract
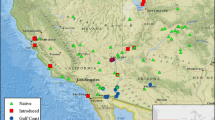
While numerous studies have documented patterns of invasion by non-indigenous plant species, few have considered the invasive properties of non-native genotypes of native species. Characteristics associated with specific genotypes, such as tolerance to disturbance, may mistakenly be applied to an entire species in the absence of genetic information, which consequently may affect management decisions. We report here on the incidence and growth of an introduced lineage of Phragmites australis in the Gulf of Mexico coastal zone of Louisiana. P. australis was collected from nine separate locations for inclusion in a series of growth experiments. Chloroplast DNA analysis indicated that specimens collected from four locations in the Mississippi River Delta represented the introduced Eurasian haplotype; the remainder represented the gulf coast haplotype. Three distinct genotypes, or clones, were identified within each haplotype via analysis using amplified fragment length polymorphisms, which also revealed reduced genetic diversity of the gulf coast clones compared to the Eurasian clones. Clones of each haplotype were planted along with three other native macrophytes at similar densities in a restored brackish marsh and monitored for growth. After 14 months, the Eurasian haplotype had spread vegetatively to cover about 82% of the experimental plots, more than four times the coverage (18%) of the gulf coast haplotype. Thus, the use of P. australis plantings for wetland restoration should consider the genetic lineage of plants used since our results indicate the potential of the Eurasian haplotype to grow rapidly at newly restored sites. This rapid growth may limit the establishment of more slowly growing native species.




Similar content being viewed by others
References
Angradi TR, Hagan SM, Able KW (2001) Vegetation type and the intertidal macroinvertebrate fauna of a brackish marsh: Phragmites vs. Spartina. Wetlands 21:75–92
Bart D, Hartman JM (2002) Environmental constraints on early establishment of Phragmites australis in salt marshes. Wetlands 22:201–213
Bart D, Hartman JM (2003) The role of large rhizome dispersal and low salinity windows in the establishment of common reed, Phragmites australis, in salt marshes: new links to human activities. Estuaries 26:436–443
Benoit LK, Askins RA (1999) Impact of the spread of Phragmites on the distribution of birds in Connecticut tidal marshes. Wetlands 19:194–208
Buchsbaum RN, Catena J, Hutchins E, James-Pirri M-J (2006) Changes in marsh vegetation, Phragmites australis, and nekton in response to increased tidal flushing in a New England salt marsh. Wetlands 26:544–557
Burdick DM, Buchsbaum R, Holt E (2001) Variation in soil salinity associated with expansion of Phragmites australis in salt marshes. Environ Exp Bot 46:247–261
Burdick DM, Konisky RA (2003) Determinants of expansion for Phragmites australis, common reed, in natural and impacted coastal marshes. Estuaries 26:407–416
Chabreck RH, Palmisano AW (1973) The effects of Hurricane Camille on the marshes of the Mississippi River Delta. Ecology 54:1118–1123
Chambers RM, Meyerson LA, Saltonstall K (1999) Expansion of Phragmites australis in tidal wetlands of North America. Aquat Bot 64:261–273
Chambers RM, Osgood DT, Bart DJ, Montalto F (2003) Phragmites australis invasion and expansion in tidal wetlands: interactions among salinity, sulfide, and hydrology. Estuaries 26:398–406
Clevering OA, Lissner J (1999) Taxonomy, chromosome numbers, clonal diversity and population dynamics of Phragmites australis. Aquat Bot 64:185–208
D’Amato F (1997) Role of somatic mutations in the evolution of higher plants. Caryologia 59:1–15
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Ellstrand NC, Schierenbeck KA (2000) Hybridization as a stimulus for the evolution of invasiveness in plants? Proc Nat Acad Sci USA 97:7043–7050
Farnsworth EJ, Meyerson LA (2003) Comparative ecophysiology of four wetland plant species along a continuum of invasiveness. Wetlands 23:750–762
Findlay S, Groffman P, Dye S (2003) Effects of Phragmites removal on marsh nutrient cycling. Wetlands Ecol Manag 11:157–165
Hauber DP, White DA, Powers SP, DeFrancesch FR (1991) Isozyme variation and correspondence with unusual reflectance patterns in Phragmites australis. Plant Syst Evol 178:1–8
Howard RJ, Rafferty PS (2006) Clonal variation in response to salinity and flooding stress in four marsh macrophytes of the northern Gulf of Mexico, USA. Environ Exp Bot 56:301–313
Hunter KL, Fox DA, Brown LM, Able KW (2006) Responses of resident marsh fishes to stages of Phragmites australis invasion in three mid Atlantic estuaries. Estuaries Coasts 29:487–498
Kimura M (1983) The neutral theory of molecular evolution. Cambridge University Press, Cambridge, UK
Kiviat K, Hamilton E (2001) Phragmites use by native North Americans. Aquat Bot 69:341–357
Klekowski EJ Jr (1997) Somatic mutation theory of clonality. In: de Kroon H, van Groenendael J (eds) The ecology and evolution of clonal plants. Backhuys Publishers, Leiden, The Netherlands, pp 227–241
Konisky RA, Burdick DM (2004) Effects of stressors on invasive and halophytic plants of New England salt marshes: a framework for predicting response to tidal restoration. Wetlands 24:434–447
Koppitz H (1999) Analysis of genetic diversity among selected populations of Phragmites australis world-wide. Aquat Bot 64:209–221
Kuhl H, Koppitz H, Rollentschek H, Kohl J-G (1999) Clone specific differences in a Phragmites australis stand I. Morphology, genetics and site description. Aquat Bot 64:235–246
Lambertini C, Gustafsson MHG, Frydenberg J, Lissner J, Speranza M, Brix H (2006) A phylogeographic study of the cosmopolitan genus Phragmites (Poaceae) based on AFLPs. Plant Syst Evol 258:161–182
League MT, Colbert EP, Seliskar DM, Gallagher JL (2006) Rhizome growth dynamics of native and exotic haplotypes of Phragmites australis (common reed). Estuaries Coasts 29:269–276
Leonard LA, Wren PA, Beavers RL (2002) Flow dynamics and sedimentation in Spartina alterniflora and Phragmites australis marshes of the Chesapeake Bay. Wetlands 22:414–424
Levine JM, Vilá M, D’Antonio CM, Dukes JS, Grigulis K, Lavorel S (2003) Mechanisms underlying the impacts of exotic plant invasions. Proc R Soc Lond 270:775–781
Ludwig DF, Iannuzzi TJ, Esposito AN (2003) Phragmites and environmental management: a question of values. Estuaries 26:624–630
Lynch EA, Saltonstall K (2002) Paleoecological and genetic analyses provide evidence for recent colonization of native Phragmites australis populations in a Lake Superior wetland. Wetlands 22:637–646
Lynch M (1990) The similarity index and DNA fingerprinting. Mol Biol Evol 7:478–484
Metzler K, Rozas R (1987) Additional notes on the tidal wetlands of the Connecticut River. Newsl Conn Bot Soc 15:1–6
Minchinton TE (2002) Disturbance by wrack facilitates spread of Phragmites australis in a coastal marsh. J Exp Mar Biol Ecol 281:89–107
Minchinton TE, Bertness M (2003) Disturbance-mediated competition and the spread of Phragmites australis in a coastal marsh. Ecol Appl 13:1400–1416
Moore HH, Neiring WA, Marsicano LJ, Dowdell M (1999) Vegetation change in created emergent wetlands (1988–1996) in Connecticut (USA). Wetlands Ecol Manag 7:177–191
Osgood DT, Yozzo DJ, Chambers RM, Jacobson D, Hoffman T, Wnek J (2003) Tidal hydrology and habitat utilization by resident nekton in Phragmites and non-Phragmites marshes. Estuaries 26:522–533
O’Neil T (1949) The muskrat in the Louisiana coastal marshes. Louisiana Department of Wildlife and Fisheries, New Orleans, LA
Orson RA (1999) A paleoecological assessment of Phragmites australis in New England tidal marshes: changes in plant community structure during the last few millennia. Biol Invasions 1:149–158
Orson RA, Warren RS, Niering WA (1987) Development of a tidal marsh in a New England river valley. Estuaries 10:20–27
Pellegrin D, Hauber DP (1999) Isozyme variation among populations of the clonal species, Phragmites australis (Cav.) Trin. ex Steudel. Aquat Bot 63:241–259
Penfound WT, Hathaway ES (1938) Plant communities in the marshlands of southeastern Louisiana. Ecol Monogr 8:1–56
Raichel DL, Able KW, Hartman JM (2003) The influence of Phragmites (common reed) on the distribution, abundance, and potential prey of a resident marsh fish in the Hackensack Meadowlands, NJ. Estuaries 26:511–521
Rice D, Rooth J, Stevenson JC (2000) Colonization and expansion of Phragmites australis in upper Chesapeake Bay tidal marshes. Wetlands 20:280–299
Rogers SO, Bendich AJ (1985) Extraction of DNA from milligram amounts of fresh, herbarium, and mummified plant tissue. Plant Mol Biol 5:69–76
Rooth JE, Stevenson JC (2000) Sediment deposition patterns in Phragmites australis communities: implications for coastal areas threatened by rising sea-level. Wetlands Ecol Manag 8:173–183
Rooth JE, Windham L (2000) Phragmites on death row: is biocontrol really necessary? Wetland J 12:29–37
Rouget M, Richardson DM (2003) Inferring process from pattern in plant invasions: a semi mechanistic model incorporating propagule pressure and environmental factors. Am Nat 162:713–724
Saghai-Maroof MA, Soliman DM, Jorgenson RA, Allard RW (1984) Ribosomal DNA spacer-length polymorphisms in barley: Mendelian inheritance, chromosomal location, and population dynamics. Proc Nat Acad Sci USA 81:8014–8018
Saltonstall K (2002) Cryptic invasion by a non-native genotype of the common reed, Phragmites australis, into North America. Proc Nat Acad Sci USA 99:2445–2449
Saltonstall K (2003a) Genetic variation among North American populations of Phragmites australis: implications for management. Estuaries 26:445–452
Saltonstall K (2003b) A rapid method for identifying the origin of North American Phragmites populations using RFLP analysis. Wetlands 23:1043–1047
Saltonstall K, Peterson PM, Soreng RJ (2004) Recognition of Phragmites australis subsp americanus (Poaceae: Arundinoideae) in North America: evidence from morphological and genetic analyses. SIDA 21:683–692
Silliman BR, Bertness MA (2004) Shoreline development drives invasion of Phragmites australis and the loss of plant diversity in New England salt marshes. Conserv Biol 18:1424–1434
Talley TS, Levin LA (2001) Modification of sediments and macrofauna by an invasive marsh plant. Biol Invasions 3:51–68
Thuiller W, Richardson DM, Rouget M, Proches S, Wilson JRU (2006) Interactions between environment, species traits, and human uses describe patterns of plant invasions. Ecol 8:1755–1769
Travis SE, Proffitt CE, Ritland K (2004) Population structure and inbreeding vary with successional stage in created Spartina alterniflora marshes. Ecol Appl 14:1189–1202
Trocki CL, Paton PWC (2006) Assessing habitat selection by foraging egrets in salt marshes at multiple spatial scales. Wetlands 26(2):307–312
Vasquez EA, Glenn EP, Brown JJ, Guntenspergen GR, Nelson SG (2005) Salt tolerance underlies the cryptic invasion of North American salt marshes by an introduced haplotype of the common reed Phragmites australis (Poaceae). Mar Ecol Prog Ser 298:1–8
Warren RS, Fell PE, Grimsby JL, Buck EL, Rilling GC, Fertik RA (2001) Rates, patterns and impacts of Phragmites australis expansion and effects of experimental Phragmites control on vegetation, macroinvertebrates, and fish within tidelands of the lower Connecticut River. Estuaries 24:90–107
Weinstein MP, Balletto JH (1999) Does the common reed, Phragmites australis, affect essential fish habitat? Estuaries 22:793–802
Weis JS, Weis P (2003) Is the invasion of the common reed, Phragmites australis, into tidal marshes of the eastern US an ecological disaster? Mar Pollut Bull 46:816–820
White DA, Hauber DP, Hood CS (2004) Clonal differences in Phragmites australis from the Mississippi River delta. Southeastern Nat 3:531–544
Windham L, Ehrenfeld JG (2003) Net impact of a plant invasion on nitrogen-cycling processes within a brackish tidal marsh. Ecol Appl 13:883–897
Windham L, Lathrop RG (1999) Effects of Phragmites australis (common reed) invasion on aboveground biomass and soil properties in brackish tidal marshes of the Mullica River, New Jersey. Estuaries 22:927–935
Windham L, Weis JS, Weis P (2003) Uptake and distribution of metals in two dominant salt marsh macrophytes, Spartina alterniflora (cordgrass) and Phragmites australis (common reed). Mar Pollut Bull 42:811–816
Zedler JB, Kercher S (2004) Causes and consequences of invasive plants in wetlands. Crit Rev Plant Sci 23:431–452
Acknowledgments
Partial funding for this study was provided by the Louisiana Department of Natural Resources, Coastal Restoration Division, Agreement No. 2503-98-36. The assistance of Russell Walters and Jennifer Kemmerer with plant collection, and Timothy Daigle, Brian Briggs, Natalie Klingensmith, and Troy Olney with field data collection is appreciated. We thank Darren Johnson for statistical consultation and assistance, Kristie Weeks for editorial review, and Christina Boudreaux for assistance with graphics. The manuscript was improved with critical comments from Donald Hauber, Beth Middleton, and two anonymous reviewers. The mention of trade names or commercial products does not constitute endorsement or recommendation for use by US Geological Survey.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Howard, R.J., Travis, S.E. & Sikes, B.A. Rapid growth of a Eurasian haplotype of Phragmites australis in a restored brackish marsh in Louisiana, USA. Biol Invasions 10, 369–379 (2008). https://doi.org/10.1007/s10530-007-9136-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-007-9136-7