Abstract
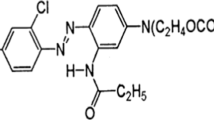
This paper presents results on anaerobic degradation of the azo dye blue HFRL in a bench scale Upflow anaerobic sludge blanket (UASB) reactor operated at ambient temperature. The results show that the addition of yeast extract (500 mg/L) increased color removal (P < 0.05) from 62 to 93% despite the low chemical oxygen demand (COD) removal (~35%) which happened due to volatile fatty acids (VFA) accumulation. There were no differences in color removal (~91%) when yeast extract (500 mg/L) was used in the presence or absence of glucose, suggesting that yeast extract acted as source of redox mediator (riboflavin) and carbon. The specific rate of dye removal increased along the operational phases and depended on the presence of yeast extract, suggesting progressive biomass acclimatization. Analysis of bacterial diversity by Polymerase Chain Reaction-Denaturing Gradient Gel Electrophoresis (PCR–DGGE) method showed there was biomass selection along the bioreactor operation and no evidence of azo dye degrading bacteria predominance. This strengthens the hypothesis that color removal happens extracellularly by the reduction of azo bond by reduced redox mediators, such as riboflavin, which is present in high amount in the yeast extract.
Similar content being viewed by others
Introduction
The textile industry is an important Brazilian economic activity that has been increasing with the economic growth observed in the last years. As a result there has been an increase in the generation of textile wastewater, which contains a variety of chemicals such as dyes, surfactants, and starch, which confers high color and organic matter to the effluent. Although the Brazilian environmental legislation does not estipulate limits for color removal or for effluent discharge, it is clear on the fact that effluent discharges cannot alter the color of the water body which receives the pollution load (CONAMA 2005).
Textile effluents can be treated by a variety of processes that includes biological and physical–chemical schemes. In Brazil, most textile industries employ, for treatment of their wastewater, the well-known biological process of activated sludge. Such process is normally efficient in removing the organic load from wastewater, but is not so effective in decolorizing it. The use of activated carbon or coagulants might be used as complement, but normally generates a high amount of sludge that needs to be properly handled (Dos Santos et al. 2007).
One technology that has the potential of being used for color removal of textile effluents is the anaerobic digestion (Georgiou et al. 2004). In anaerobic conditions, textile pollutants such as the azo dyes (which represent around 70% of the dyes produced worldwide) can be used as electron acceptors, leading to the reduction of the azo bond (–N=N–) and production of amines, mostly aromatic. Indeed, different research groups have demonstrated that removal efficiencies varying from 60 to 80% can be obtained during the anaerobic treatment of azo dye solutions (Mendez-Paz et al. 2005; Dos Santos et al. 2006).
Upflow anaerobic sludge blanket (UASB) reactor has been widely applied in Brazil for the anaerobic treatment of different industrial wastewater (Expired dairy, slaughterhouse, and food processing) due to its inherent advantages, such as the ability of allowing a high sludge age without the need of sludge granulation, as well as the low degree of mechanization and energy requirement. In spite of this, there are a handful of papers on the use of UASB reactor for treating textile effluents. For instance, Somasiri et al. (2008) used an UASB reactor [Hydraulic retention time (HRT) = 24 h] for treating a textile effluent [Chemical oxygen demand (COD) = 2000 mg/L] and showed that a significant color removal (92%) could be achieved. The authors also observed that toxic byproducts accumulated in the reactor, contributing to reduce overall organic matter removal efficiency.
The low growth of anaerobic microorganisms is a major drawback for the treatment of recalcitrant and toxic effluents, which might be worsened by electron transfer limitation between different species of the anaerobic consortia. According to Dos Santos (2005) one strategy to avert this and increase the efficiency of color removal during anaerobic treatment is to use redox mediators. Such compounds are reduced by the reducing equivalents produced by microbial cells (e.g. NADH, FADH) and then re-oxidized by the azo dye, thereby enhancing the decolorization kinetics. Some authors have demonstrated that compounds such as riboflavin (vitamin B2) and sulfonated antraquinones (AQS) can act as redox mediators (Rau et al. 2002; Field and Brady 2003; Cervantes et al. 2006). Dos Santos et al. (2006, 2003) showed that decolorization rates were increased by 2.7 and 6.1 times in the presence of AQS and vitamin B2, respectively, during anaerobic thermophilic degradation of azo dye reactive red 2 (RR2) in an expanded granular sludge blanket (EGSB) reactor. Such study was carried out at controlled temperature and employing a purified form of vitamin B2; and it is important, from the practical engineering point of view, to investigate anaerobic degradation of azo dyes at ambient temperature and using commercial sources of redox mediators.
Therefore, the main objective of this paper is to present results on anaerobic degradation of a synthetic textile effluent containing the azo dye reactive blue HFRL, commonly used in the textile industry, in a bench scale UASB reactor operated at ambient temperature. In addition, it was evaluated the role of yeast extract, source of the redox mediator riboflavin, in enhancing color removal during the anaerobic treatment.
Experimental
Apparatus and operational conditions
The bench scale UASB reactor was built using polyvinyl chloride (PVC) pipes and joints and had a total working volume of 8 L. The upper settler was 250 mm height and had 150 mm of diameter, making up 2 L of the working volume, whereas the digestion chamber had 100 mm of diameter and 800 mm of height, making up 6 L of the working volume. Five sampling ports were installed along the height of the digestion chamber, thus allowing the monitoring of biomass growth, which was estimated by quantifying volatile suspended solids (VSS).
The reactor project followed the parameters recommended by Chernicharo (2007) and resulted in average hydraulic volumetric load and upflow velocity in the reactor of, respectively, 1.3 m3/m3 day (calculated considering the reactor useful volume) and 0.0165 m/h (at the digestion chamber). Such low upflow velocity contributed for nearly complete retention of solids in the reactor.
The bench scale reactor was incubated with anaerobic sludge from a demo scale UASB reactor installed at the Centre for research and training on sanitation (CePTS) UFMG/COPASA, located at the Arrudas WWTP, in Belo Horizonte–Brazil. The bench scale UASB was operated at ambient temperature and under different conditions (Table 1). In all phases the HRT was kept at 19 h and the reactor was run without any discharge of biomass, except during sampling for physico-chemical analyses.
The feed solution was comprised of glucose (except in phase 6), azo dye (except in phase 1), yeast extract (except phases 1, 2, and 5) and macronutrient solution, which composition is described elsewhere (Aquino et al. 2007). In all phases, the minimum COD:N:P proportion of 350:5:1 was observed, according to Chernicharo (2007). The feeding recipient (20 L) was filled up twice a day with freshly prepared influent solution to minimize the growth of microorganisms and consequent removal of COD and color in the feeding line. The feed was pumped into the reactor by means of a peristaltic pump (Dosa Mini 400, HD Hidraulis) at constant flowrate, calculated to maintain a HRT of 19 h.
Analytical procedures and statistical analyses
During reactor operation the following parameters were assessed: VSS in samples collected along the UASB digestion chamber; COD, color, pH, volatile fatty acids (VFA) and glucose in influent and effluent samples. The analyses of pH, COD, and VSS followed the procedures described in the Standard methods for the examination of water and wastewater (APHA 2005). Since the COD was measured in the supernatant produced by centrifuging the samples at 5000 rpm for 15 min (Fanem Excelsa 11, Model 206 BL), all COD values reported in this paper refer to a soluble fraction which contains both dissolved and colloidal species.
The efficiency of color removal and of blue HRFL azo dye degradation were assessed by following the absorbance (λmáx = 654 nm) of the centrifuged solution in a spectrophotometer (HP 8453 UV–Vis, system). VFA analyses were possible using a high performance liquid chromatography (HPLC) in a Hewlett Packard Series 1050 chromatographic equipment with a Biorad Aminex HPX-87H ion exclusion column maintained at 55°C.
For the chromatographic analysis, the injection volume was 40 μL and the mobile phase was 0.01 mol/L H2SO4 at a constant flowrate of 0.6 mL/min. VFA were detected with ultraviolet (UV) light at 210 nm. The VFA methodology was properly validated, achieving the criteria of linearity, precision, selectiveness, and accuracy within the concentration range of 25–1000 mg/L. The limit of detection (LD) was determined as 3 mg/L for all organic acids (C1–C5) except valeric acid which showed a higher LD (12.5 mg/L).
Quantification of VFA was used to estimate the COD caused by such intermediate products according to Eq. 1, suggested by Aquino and Stuckey (2003).
In order to verify any significant differences amongst the parameters, the software Statistica® was used. As the number of samples was reduced, it was not possible to verify their normality. For this reason the more conservative non-parametric tests of Kruskal–Wallis ANOVA and Mann–Whitney were applied. The evaluation has been made from the P-value, which should be lower than 0.05 so that the hypothesis of equal samples is rejected with a 95% confidence interval.
Microbial analysis
Microbial diversity analysis was performed using the Polymerase Chain Reaction-Denaturing Gradient Gel Electrophoresis (PCR–DGGE) approach (Muyzer et al. 1993). For this, 50 mL of sludge samples collected at the end of each operational phase was centrifuged at 5000 rpm for 10 min to remove interstitial water. Then the pellet was further washed with PBS buffer (NaCl 130 mM, Na2HPO4 0.7 mM, NaH2PO4 0.3 mM, pH 7.2) in order to remove extracellular nucleic acids.
DNA extractions from sludge samples were performed in triplicates using a phenol–chloroform protocol, as described in Griffiths et al. (2000). 16S-rRNA genes from the purified DNA were amplified using a primer set specific for Bacteria domain (357F–GC e 907R). Each PCR reaction was performed in PCR buffer (1×, Fermentas), MgCl2 (2.0 mM, Fermentas), dNTP (0.8 mM), primer (500 nM each, Bioneer), BSA (0.2 mg/L), Taq polimerase (1.5 μm/μL), and 1 μL of DNA template for a final volume of 50 μL. The PCR program was performed in a Biocycler thermociclador with initial denaturation at 3 min at 94°C; 30 cycles at 94°C for 1 min; 55°C for 1 min; 72°C for 1 min, and a final extension at 72°C for 7 min.
DGGE of the PCR-amplified 16S-rDNA was performed according to Muyzer et al. (1993). The polyacrylamide concentration used in the DGGE analysis was 6% and the denaturing gradient was from 30 to 70%. The denaturing gradient gel was then submitted to electrophoresis at 100 V at 60°C for 16 h. Finally, the resulting gels were stained with ethidium bromide and photographed under UV transillumination.
Results and discussions
Effect of yeast extract on color and COD removal
The results presented in Fig. 1 show that the median COD removal was very low during the first phase, and this probably happened due to biomass adaptation. During the second phase biomass was more adapted and the median efficiencies of COD and color removal were, respectively, 49 and 65%. During the third phase, the organic load did not change significantly (0.718 kg/m3 day) despite the addition of 100 mg/L of yeast extract, but the median efficiency of COD removal was lower than that observed in the previous phase and the data were inexplicably highly variable.
The presence of 100 mg/L of yeast extract in the feed, led to a median efficiency of color removal of 60%, which was not statistically different (P > 0.05) from the value observed in phase 2. Likewise, the COD values and removal efficiencies obtained in phases 2 and 3 were not statistically different, mainly because of the high COD variability observed in phase 3. These data show that the use of yeast extract at lower concentration (100 mg/L) did not enhance color removal nor affected COD removal, which remained low due to accumulation of byproducts, as discussed below. It is also possible that during phase 3 biomass was not fully adapted to use yeast extract as source of redox mediator (riboflavin), and some data that will be further presented indicates there was a progressive biomass adaptation in the UASB reactor.
During the fourth phase, when yeast extract was supplemented at a higher concentration (500 mg/L), it was observed an increase in the organic load (from 0.718 to 1.181 kg/m3 day) since glucose was maintained as carbon source (350 mg/L) in the feed solution. In this condition the color removal significantly increased (P < 0.05) from 62 to 93% despite the low COD removal (~35%). When the results of the fourth phase are compared to those of the sixth phase (reactor fed with 500 mg/L of yeast extract without glucose), data analysis show that there is statistical difference only for COD removal, since the median removal efficiency increased from 35 to 56% in the sixth phase. This probably happened due to the reduction of organic load (from 1.181 to 0.689 kg/m3 day) which led to a lower accumulation of VFA as shown in Fig. 2.
These results indicate that the anaerobic microorganisms were more adapted to the toxic aromatic amines produced during the azo dye reduction since the amount of azo dye degradation during phase 6 was not statistically different (P > 0.05) from phase 4. In order to investigate the dynamics of bacterial diversity along the reactor operation DGGE analysis was carried out and the results are shown in Fig. 3. By comparing the band profile among samples collected from phases 2, 3, 4 and 5 it can be observed that there is a trend of decreasing the numbers of DGGE bands as time went by. Considering that the band profile represents predominant bacterial diversity, this result suggests that there was a progressive biomass adaptation, with the selection of those more adapted to degrade the azo dye and cope with the aromatic amines.
In addition, assuming that a carbon source is needed to produce the compounds that reduce either the redox mediators or the azo dye in solution, the results shown in Fig. 1 also indicate that yeast extract (500 mg/L) acted as carbon source. In other words, the use of 500 mg/L of yeast extract (source of redox mediator) in the presence (phase 4) or absence (phase 6) of glucose resulted in a similar dye removal efficiency (91%), indicating that glucose is not necessary as co-substrate when yeast extract (500 mg/L) is supplemented, which suggests that the process of the descolorization is relatively non–specific with respect to its electron donor (Van der Zee and Villaverde 2005). This is important since the simultaneous addition of glucose and yeast extract, as seen in phase 4, significantly increased the applied organic load and led to a reduction in COD removal efficiency (when compared to phase 6) due to a higher accumulation of VFA (Fig. 2).
The comparison of color removal efficiencies of phase 5, when the reactor was fed with glucose and without yeast extract, with phase 4 (with glucose and yeast extract) clearly shows that the absence of yeast extract led to a decrease (from 92 to 62%) in color removal, proving that the use of yeast extract (source of the redox mediator riboflavin) enhanced degradation of the azo dye blue HFRL. Others authors such as Nigam et al. (1995) also demonstrated the effect of yeast extract (0.5% w/v) in improving the reduction of azo dyes by bacterial culture isolated from samples of wastewater containing dyes. However, such authors used yeast extract as a culture medium complement and did not discuss the role of yeast extract as source of redox mediator. Acoording to Van der Zee and Cervantes (2009) redox mediators have the ability of accelerating the azo dye reduction, and this is highly relevant for the applicability of biological techniques to remove color from textile effluents in full scale wastewater treatment plants.
The average color removal efficiency (92%) obtained in a UASB reactor supplemented with 500 mg/L of yeast extract (phase 6) can be considered high since a comparable efficiency (97%) of azo dye removal was obtained by Ortiz (2008), only when an anaerobic effluent was firstly treated in an aerobic reactor and then by adsorption onto activated carbon. In addition to a high color removal, the conditions used in phase 6 allowed the degradation of half of the influent COD. The COD removal efficiency was not higher due to accumulation of VFA, which represented ~40% of the soluble COD leaving the reactor (Fig. 2). As it will be discussed below, the accumulation of VFA reflects instability of the anaerobic system, which happened in all operational phases (except phase 1) probably due to the accumulation of aromatic amines. Such compounds, which are toxic to microorganisms and formed during anaerobic degradation of azo dyes, normally accumulate in anaerobic reactors. For instance Isik and Sponza (2008) showed that only 28% of the aromatic amines generated by the degradation of the azo dyes studied were removed in an UASB reactor.
VFA accumulation and residual COD
Figure 2 shows that accumulation of VFA was significant during all operational phases. VFA accumulation contributed from 29 to 65% of the soluble COD, with the highest value being observed during phase 4. Since VFA are intermediate products of the anaerobic digestion, their accumulation reflects an imbalance between the acidogenic and methanogenic microorganisms, which have quite distinct growth rates. Such imbalance might happen due to any factor that hampers the reproduction of the slow growing methanogens, such as pH outside the 6.5–7.5 range, low temperature, lack of nutrients and/or presence of toxic compounds.
Figure 4 shows that median pH values varied from 7.13 to 7.19, with 6.5 being the minimum value observed only during one operational phase. It is noteworthy that when the pH dropped below 6.8 alkalinity (NaHCO3) was added straightway inside the reactor to keep it around neutrality, and this explains the high pH amplitude observed in some operational phases (e.g. phases 4 and 5). As far as the reactor temperature is concerned, Fig. 4 shows that the minimum value was 20°C and that the median temperature varied from 23 to 26.5°C. These results suggest that pH and temperature were not responsible for the increased VFA accumulation observed in some phases. Since the feed solution contained essential vitamins and minerals, nutrient deficiency could not be blamed either for VFA accumulation. This opens up the possibility for microbial toxicity, which is consistent with the fact that biological degradation of most azo dyes lead to the formation of toxic aromatic amines (Isik and Sponza 2003; Mendez-Paz et al. 2005; Dos Santos et al. 2006, 2003). In addition, it is also possible that the dye itself and/or some of its impurities might also be toxic to anaerobic microorganisms.
In particular, the high VFA accumulation observed in phase 4 might be explained by the fact that during this phase the organic load was increased from 0.718 to 1.181 Kg COD/m3 day due to the simultaneous addition of glucose (350 mg/L) and yeast extract (500 mg/L). This condition of abundance of easily degradable substrate might have led to an increase in the population of the fast growing acidogenic bacteria which promptly produced VFA, mainly acetate. According to the DGGE results (Fig. 3), an important difference between phase 4 and the other phases is the presence of a band that seems to be exclusive of phase 4. This suggests that there is at least one difference in the bacterial community, maybe among fermentative bacteria, which could be a bacterial population involved in VFA production in this particular phase.
Facultative anaerobic bacteria, such as species of Pseudomonas and Escherichia coli have been reported to play a role on decolorization process under anaerobic conditions (Bhatt et al. 2005; Kalyani et al. 2008; Isik and Sponza 2003). For instance a bacterial strain Pseudomonas aeruginosa NBAR12 isolated from textile dye wastewater was capable of decolorizing diazo reactive blue 172 when glucose and yeast extract was supplied in the medium (Bhatt et al. 2005). According to Isik and Sponza (2003) during the degradation of azo dyes under anaerobic conditions by E. coli and Pseudomonas sp. strains, benzidine and VFA were detected as degradation intermediates of dyes and glucose, demonstrating that aromatic amines were not further mineralized and accumulated at the end of the anaerobic incubation period.
According to the results reported in the present study, the fact that mainly acetate accumulated in solution indicates that the slow growing methanogens did not keep up with the acidogenic bacteria, and this might have happened due to toxicity caused by aromatic amines that likely accumulated in solution. It is important to notice that the VFA accumulation was similar during phases 3 and 5, since the CODVFA/CODtotal ratio were, respectively, 49 and 48%. This was somehow expected since the operational conditions were the same in these two phases, and indicates that the reactor performance was reproducible. The fact that less CODVFA and less residual COD was observed during phase 5 indicates an increase in biomass adaptation from phase 3 to 5. In addition, it was observed a reduction in VFA accumulation from phase 4 onwards; suggesting a progressive biomass adaptation.
Figure 5 shows that the specific rate of dye removal, estimated by converting the samples absorbance at 654 nm into azo dye concentration and by using data of VSS, increased along the operational phases, except during phase 5. In this phase there was a reduction in the specific rate of dye removal due to the absence of yeast extract in the feed solution. This is a further indication that yeast extract might have acted as source of redox mediator and that there was progressive biomass acclimatization along the operational phases. Biomass acclimatization was favored because there was virtually 100% of solids retention due to the low upflow velocity in the UASB and no sludge discharge.
VFA accumulation is undesirable since it decreases COD removal efficiency. However, since such intermediate compounds are biodegradable, their removal can be attained by improving the conditions for the growth of VFA consumers (acetogenic and methanogenic bacteria). In the absence of stressful conditions the acidogenic, acetogenic, and methanogenic steps occur at a regular pace so that the rate of VFA production equals that of VFA degradation. The fact that the CODVFA was the lowest during phase 6 indicates that the presence of yeast extract (500 mg/L) allied to the higher biomass acclimatization somehow enhanced VFA degradation. However, the COD caused by other compounds (CODnot VFA) was 1.5 times the COD caused by VFA (CODVFA). Such unknown organics might be aromatic amines, other azo dye byproducts as well as soluble microbial products (SMP).
During phase 6 color removal reached 91% (median value) and the removal of soluble COD was just 55% because of the presence of VFA and unknown compounds in the ratio of approximately 1:3. Since little is known about the degradation of SMP and azo dye intermediates, the formation of such compounds somehow dictates the maximum COD removal that can be obtained in anaerobic systems treating azo dyes. For the sake of comparison, Dos Santos (2005) observed rates of COD removal higher than 90% utilizing glucose as co-substrate, azo dye (RR2) and anthraquinone-2,6-disulphonate (AQDS) as source of mediator redox; however, the anaerobic conditions were more favorable for microbial growth since the reactors were operated at either mesophilic (35°C) or thermophilic (55°C) range of temperature.
Microbial ecology
The fact that higher color removal rates were observed in phases 4 and 6 (Fig. 1) despite the low time of operation (Table 1) may be indicative that azo dye degradation was accomplished in the absence of specialized microorganisms (which have azoreductase enzymes, for instance). This hypothesis is also supported by the DGGE profiles that showed similar results for samples with and without yeast extract (phases 4 and 5, respectively). Therefore it can be inferred that the biomass selection observed during the bioreactor operation is probably due to the presence of aromatic amines or the dye itself rather than the specific use of yeast extract during the azo dye degradation.
The results presented before may also suggest that biological color removal happened by the reduction of the azo bond by riboflavin present in yeast extract. Such redox mediator might be reduced by electron carriers (e.g. NADH) at the cell surface and then be re-oxidized by the azo dye. According to this hypothesis, color removal would actually take place outside the cell, i.e. in the bulk solution. If this is true, color removal would rely on microorganisms only to produce reducing equivalents via their oxidative process, and this could be carried out by the fast growing acidogenic bacteria. In this scenario color removal would not depend on the development of a specific microorganism, making the kinetics of color removal much quicker, such as observed in Figs. 1 and 4.
Finally, the fact that the median color removal efficiency decreased from 92% (phase 4) to 62% (phase 5) when yeast extract was dropped out from the feed solution, and then went back up again to 91% (phase 6) when it was reintroduced in the reactor, indicates the importance of yeast extract as source of redox mediators, strengthening the hypothesis that color removal did not depend on a specialized microorganism.
Conclusions
This paper showed the addition of yeast extract (500 mg/L), in presence or absence of glucose, significantly increased color removal from 62 to 92% despite low COD removal (55% at the best scenario), which was due to VFA accumulation. VFA accumulation contributed from 29 to 65% of the soluble COD and might have resulted from toxicity due to the dye and/or aromatic amines. Biomass acclimatization increased along operation, resulting in specific dye removal rate of 19.6 g/Kg SSV day and lower VFA accumulation at the last operational phase. The results demonstrated the potential application of UASB reactor for azo dye degradation at ambient temperature (~25°C), especially when the feed was supplemented with yeast extract, a cheap source of the redox mediator riboflavin.
References
APHA (2005) Standard methods for the examination of water and wastewater, 21st edn. EUA. American Public Health Association, Washington, D.C
Aquino SF, Stuckey DC (2003) Soluble microbial products (SMP) formation in anaerobic chemostats in the presence of toxic compounds. Water Res 38:255–266
Aquino SF, Chernicharo CAL, Foresti E, Santos MLF dos, Monteggia LO (2007) Metodologias para determinação da atividade metanogênica específica (AME) em lodos anaeróbios. Eng Sanit Amb 12:192–201
Bhatt N, Patel KC, Keharia H, Madamwar D (2005) Decolorization of diazo dye reactive blue 172 by Pseudomonas aeruginosa. J Basic Microbiol 45:407–418
Cervantes FJ, Enriquez JE, Mendoz A, Hernandes ZMR, Razo-Flores E, Field JA (2006) The role of sulphate reduction on the reductive decolorization of the azo dye reactive orange 14. Water Sci Technol 54:171–177
Chernicharo CAL (2007) Princípios do tratamento biológico de águas residuárias “Reatores Anaeróbios”. Editora UFMG, Belo Horizonte
CONAMA (2005) National Council of Environment resolution number 357, March 17, Brasil
Dos Santos AB (2005) Reductive decolourisation of dyes by thermophilic anaerobic granular sludge. PhD thesis, Wageningen University
Dos Santos AB, Cervantes FJ, Yaya-Beas RE, Van Lier JB (2003) Effect of redox mediator, AQDS, on the decolourisation of a reactive azo dye containing triazine group in a thermophilic anaerobic EGSB reactor. Ezyme Microb Tech 33:942–951
Dos Santos AB, Cervantes FJ, Van Lier JB (2006) Potentials of high-temperature anaerobic treatment and redox mediators for the reductive decolourisation of azo dyes from textile wastewaters. Water Sci Tecnol 54:151–156
Dos Santos AB, Cervantes FJ, Van Lier JB (2007) Impacto dos mediadores redox na remoção de cor de corantes azo e antraquinônico por lodo granular anaeróbio sob condições mesofílicas e termofílicas. Eng Sanit Amb 12:102–108
Field JA, Brady J (2003) Riboflavin as a redox mediator accelerating the reduction of azo dye mordant yellow 10 by anaerobic granular sludge. Water Sci Technol 48:187–193
Georgiou D, Metallinou C, Aivasidis A, Voudrias E, Gimouhopoulos K (2004) Decolorization of azo-reactive dyes and cotton-textile wastewater using anaerobic digestion and acetate-consuming bacteria. Biochem Eng J 19:75–79
Griffiths RI, Whiteley AS, O′Donnell AG, Bailey MJ (2000) Rapid method for coextraction DNA and RNA from natural environments for analysis of riobosomal DNA- and rRNA-based microbial community composition. Appl Environ Microbiol 66:5488–5491
Isik M, Sponza DT (2003) Effect of oxygen on decolorization of azo dyes by Escherichia coli and Pseudomonas sp. and fate of aromatic amines. Process Biochem 38:1183–1192
Isik M, Sponza DT (2008) Anaerobic/aerobic treatment of a simulated textile wastewater. Sep Purif Technol 60:64–72
Kalyani DC, Patil PS, Jadhav JP, Govindwar SP (2008) Biodegradation of reactive textile dye red BLI by an isolated bacterium Pseudomonas sp. SUK. Bioresour Technol 99:464–4635
Mendez-Paz D, Omil F, Lema JM (2005) Anaerobic treatment of azo dye acid orange 7 under batch conditions. Ezyme Microb Tech 36:264–272
Muyzer G, Wall EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Nigam P, Banat IM, Singh D, Marchant R (1995) Microbial process for the decolorization of textile effluent containing azo, diazo and reactive dyes. Process Biochem 31:435–442
Ortiz ACT (2008) Tratamento biológico com biomassa suspendida anaeróbico/aeróbico de efluente de uma indústria têxtil contendo corante azo. In: XXXI Congresso Interamericano AIDIS, Santiago, Chile
Rau J, Knackmuss HJ, Stolz A (2002) Effects of different quinoid redox mediators on the anaerobic reduction of azo dyes by bacteria. Environ Sci Tech 36:1497–1504
Somasiri W, Li XF, Ruan WQ, Jian C (2008) Evaluation of the efficacy of upflow anaerobic sludge blanket reactor in removal of colour and reduction of COD in real textile wastewater. Bioresource Technol 99:3692–3699
Van der Zee FP, Cervantes J (2009) Impact and application of electron shuttles on the redox (bio) transformation of contaminants: a review. Biotechnol Adv 27:256–277
Van der Zee FP, Villaverde S (2005) Combined anaerobic–aerobic treatment of azo dyes—a short review of bioreactor studies. Water Res 39:1425–1440
Acknowledgments
The authors would like to thank the following institutions for their financial support: Coordenação de Aperfeiçoamento de Pessoal de Nível Superior–CAPES, Conselho Nacional de Desenvolvimento Científico e Tecnológico–CNPq, Fundação de Amparo à Pesquisa do Estado de Minas Gerais–FAPEMIG, Universidade Federal de Ouro Preto-UFOP.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baêta, B.E.L., Aquino, S.F., Silva, S.Q. et al. Anaerobic degradation of azo dye Drimaren blue HFRL in UASB reactor in the presence of yeast extract a source of carbon and redox mediator. Biodegradation 23, 199–208 (2012). https://doi.org/10.1007/s10532-011-9499-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-011-9499-4