Abstract
Objective
To study bacterial 16S RNA in archival prostate samples from 352 patients with benign prostate hyperplasia (BPH) and evaluate whether the presence of bacterial DNA was different in those who later developed prostate cancer (n = 171) and in the matched controls that did not progress to cancer (n = 181).
Methods
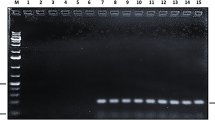
16S DNA PCR followed by cloning and sequencing the positive samples.
Results
In 96/352 (27%) of the prostate tissue specimens 16S RNA were detected. Sequence analysis revealed Propionibacterium acnes as the predominant microorganism (23% of 16S RNA positive patients). The second most frequent isolate—Escherichia coli was found in 12 (12%) patients. The other isolates included Pseudomonas sp. (3 patients), Actinomyces sp. (2), Streptococcus mutans (1), Corynebacterium sp. (2), Nocardioides sp. (1), Rhodococcus sp. (1) Veillonella sp. (2). In P. acnes positive samples 62% exhibited severe histological inflammation versus 50% in the bacteria-negative group (p = 0.602). The presence of P. acnes in the prostate was associated with prostate cancer development (OR 2.17, 95% CI 0.77–6.95).
Conclusions
This study has revealed P. acnes as the most common bacteria in the prostate in BPH. Further studies are needed to clarify its role in contributing to the development of prostatic inflammation and prostate cancer.
Similar content being viewed by others
References
Coussens LM, Werb Z (2002) Inflammation and cancer. Nature 420(6917):860–867
Moss SF, Blaser MJ (2005) Mechanisms of disease: inflammation and the origins of cancer. Nat Clin Pract Oncol 2(2):90–97; quiz 1 p following 113
Lax AJ, Thomas W (2002) How bacteria could cause cancer: one step at a time. Trends Microbiol 10(6):293–299
De Marzo AM, DeWeese TL, Platz EA, et al (2004) Pathological and molecular mechanisms of prostate carcinogenesis: implications for diagnosis, detection, prevention, and treatment. J Cell Biochem 91(3):459–477
Dennis LK, Lynch CF, Torner JC (2002) Epidemiologic association between prostatitis and prostate cancer. Urology 60(1):78–83
Gronberg H (2003) Prostate cancer epidemiology. Lancet 361(9360):859–864
Nelson WG, De Marzo AM, DeWeese TL, Isaacs WB (2004) The role of inflammation in the pathogenesis of prostate cancer. J Urol 172(5 Pt 2):S6–S11; discussion S-2
Platz EA, De Marzo AM (2004) Epidemiology of inflammation and prostate cancer. J Urol 171(2 Pt 2):S36–S40
Wang W, Bergh A, Damber JE (2005) Cyclooxygenase-2 expression correlates with local chronic inflammation and tumor neovascularization in human prostate cancer. Clin Cancer Res 11(9):3250–3256
Cohen RJ, Shannon BA, McNeal JE, Shannon T, Garrett KL (2005) Propionibacterium acnes associated with inflammation in radical prostatectomy specimens: a possible link to cancer evolution? J Urol 173(6):1969–1974
Hochreiter WW, Duncan JL, Schaeffer AJ (2000) Evaluation of the bacterial flora of the prostate using a 16S rRNA gene based polymerase chain reaction. J Urol 163(1):127–130
Krieger JN, Riley DE, Vesella RL, Miner DC, Ross SO, Lange PH (2000) Bacterial dna sequences in prostate tissue from patients with prostate cancer and chronic prostatitis. J Urol 164(4):221–228
Greer CE, Wheeler CM, Manos MM (1994) Sample preparation and PCR amplification from paraffin-embedded tissues. PCR Methods Appl 3(6):S113–S122
Harris KA, Hartley JC (2003) Development of broad-range 16S rDNA PCR for use in the routine diagnostic clinical microbiology service. J Med Microbiol 52(Pt 8):685–691
Berger RE, Krieger JN, Rothman I, Muller CH, Hillier SL (1997) Bacteria in the prostate tissue of men with idiopathic prostatic inflammation. J Urol 157(3):863–865
Gorelick JI, Senterfit LB, Vaughan ED Jr (1988) Quantitative bacterial tissue cultures from 209 prostatectomy specimens: findings and implications. J Urol 139(1):57–60
Keay S, Zhang CO, Baldwin BR, Alexander RB (1999) Polymerase chain reaction amplification of bacterial 16s rRNA genes in prostate biopsies from men without chronic prostatitis. Urology 53(3):487–491
Krieger JN, Riley DE (2002) Prostatitis: what is the role of infection. Int J Antimicrob Agents 19(6):475–479
Krieger JN, Riley DE (2002) Bacteria in the chronic prostatitis-chronic pelvic pain syndrome: molecular approaches to critical research questions. J Urol 167(6):2574–2583
Krieger JN, Riley DE, Roberts MC, Berger RE (1996) Prokaryotic DNA sequences in patients with chronic idiopathic prostatitis. J Clin Microbiol 34(12):3120–3128
Lee JC, Muller CH, Rothman I, et al (2003) Prostate biopsy culture findings of men with chronic pelvic pain syndrome do not differ from those of healthy controls. J Urol 169(2):584–587; discussion 7–8
Nickel JC, Downey J, Young I, Boag S (1999) Asymptomatic inflammation and/or infection in benign prostatic hyperplasia. BJU Int 84(9):976–981
Riley DE, Berger RE, Miner DC, Krieger JN (1998) Diverse and related 16S rRNA-encoding DNA sequences in prostate tissues of men with chronic prostatitis. J Clin Microbiol 36(6):1646–1652
Shiina H, Himeno Y, Ishibe T (1992) Organisms in the prostate and antibiotics in the treatment of postoperative infections. Urol Int 48(2):187–190
Shoskes DA, Shahed AR (2000) Detection of bacterial signal by 16S rRNA polymerase chain reaction in expressed prostatic secretions predicts response to antibiotic therapy in men with chronic pelvic pain syndrome. Tech Urol 6(3):240–242
Szoke I, Torok L, Dosa E, Nagy E, Scultety S (1998) The possible role of anaerobic bacteria in chronic prostatitis. Int J Androl 21(3):163–168
Tanner MA, Shoskes D, Shahed A, Pace NR (1999) Prevalence of corynebacterial 16S rRNA sequences in patients with bacterial and “nonbacterial” prostatitis. J Clin Microbiol 37(6):1863–1870
Fredricks DN, Fiedler TL, Marrazzo JM (2005) Molecular identification of bacteria associated with bacterial vaginosis. N Engl J Med 353(18):1899–1911
Bruggemann H, Henne A, Hoster F, et al (2004) The complete genome sequence of Propionibacterium acnes, a commensal of human skin. Science 305(5684):671–673
Ullmann AJ, Helmreich-Becker I, Maeurer MJ, et al (2000) Hepatic granuloma due to Propionibacterium acnes in a patient with acute myelogenous leukaemia. Clin Infect Dis 30(1):219–220
Harada K, Tsuneyama K, Sudo Y, Masuda S, Nakanuma Y (2001) Molecular identification of bacterial 16S ribosomal RNA gene in liver tissue of primary biliary cirrhosis: is Propionibacterium acnes involved in granuloma formation? Hepatology 33(3):530–536
Pan SC, Wang JT, Hsueh PR, Chang SC (2005) Endocarditis caused by Propionibacterium acnes: an easily ignored pathogen. J Infect 51(4):e229–e231
Chaudhry IA, Shamsi FA, Al-Rashed W (2005) Bacteriology of chronic dacryocystitis in a tertiary eye care center. Ophthal Plast Reconstr Surg 21(3):207–210
Ishige I, Eishi Y, Takemura T, et al (2005) Propionibacterium acnes is the most common bacterium commensal in peripheral lung tissue and mediastinal lymph nodes from subjects without sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis 22(1):33–42
Lightfoot N, Conlon M, Kreiger N, Sass-Kortsak A, Purdham J, Darlington G (2004) Medical history, sexual, and maturational factors and prostate cancer risk. Ann Epidemiol 14(9):655–662
Patel DA, Bock CH, Schwartz K, Wenzlaff AS, Demers RY, Severson RK (2005) Sexually transmitted diseases and other urogenital conditions as risk factors for prostate cancer: a case-control study in Wayne County, Michigan. Cancer Causes Control 16(3):263–273
Galobardes B, Davey Smith G, Jeffreys M, Kinra S, McCarron P (2005) Acne in adolescence and cause-specific mortality: lower coronary heart disease but higher prostate cancer mortality: the Glasgow Alumni Cohort Study. Am J Epidemiol 161(12):1094–1101
Acknowledgments
This study was financially supported by The Kempe Foundation, The Cancer Research Foundation of Northern Sweden, Cancerfonden, The Percy Falk Foundation for prostate cancer research, Sweden, Maud & Birger Gustavsson Foundation, The Medical Faculty, Umeå University, Sweden.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alexeyev, O., Bergh, J., Marklund, I. et al. Association between the presence of bacterial 16S RNA in prostate specimens taken during transurethral resection of prostate and subsequent risk of prostate cancer (Sweden). Cancer Causes Control 17, 1127–1133 (2006). https://doi.org/10.1007/s10552-006-0054-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10552-006-0054-2