Abstract
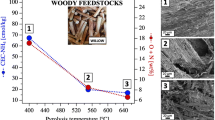
A wide range of waste biomass/waste wood feedstocks abundantly available at mine sites provide the opportunity to produce biochars for cost-effective improvement of mine tailings and contaminated land at metal mines. In the present study, soft- and hardwood biochars derived from pine and jarrah woods at high temperature (700 °C) were characterized for their physiochemical properties including chemical components, electrical conductivity, pH, zeta potential, cation-exchange capacity (CEC), alkalinity, BET surface area and surface morphology. Evaluating and comparing these characteristics with available data from the literature have affirmed the strong dictation of precursor type on the physiochemical properties of the biochars. The pine and jarrah wood feedstocks are mainly different in their proportions of cellulose, hemicellulose and lignin, resulting in biochars with heterogeneous physiochemical properties. The hardwood jarrah biochar exhibits much higher microporosity, alkalinity and electrostatic capacity than the softwood pine. Correlation analysis and principal component analysis also show a good correlation between CEC–BET–alkalinity, and alkalinity–ash content. These comprehensive characterization and analysis results on biochars’ properties from feedstocks of hardwood (from forest land clearance at mine construction) and waste pine wood (from mining operations) will provide a good guide for tailoring biochar functionalities for remediating metal mine tailings. The relatively inert high-temperature biochars can be stored for a long term at mine closure after decades of operations.







Similar content being viewed by others
References
Ahmad, M., Lee, S. S., Dou, X., Mohan, D., Sung, J. K., Yang, J. E., et al. (2012a). Effects of pyrolysis temperature on soybean stover- and peanut shell-derived biochar properties and TCE adsorption in water. Bioresource technology, 118, 536–544.
Ahmad, M., Lee, S. S., Yang, J. E., Ro, H. M., Han Lee, Y., & Sik Ok, Y. (2012b). Effects of soil dilution and amendments (mussel shell, cow bone, and biochar) on Pb availability and phytotoxicity in military shooting range soil. Ecotoxicology and Environmental Safety, 79, 225–231.
Ahmad, M., Lee, S. S., Rajapaksha, A. U., Vithanage, M., Zhang, M., Cho, J. S., et al. (2013a). Trichloroethylene adsorption by pine needle biochars produced at various pyrolysis temperatures. Bioresource Technology, 143, 615–622.
Ahmad, M., Moon, D. H., Vithanage, M., Koutsospyros, A., Lee, S. S., Yang, J. E., et al. (2014). Production and use of biochar from buffalo-weed (ambrosia trifida L.) for trichloroethylene removal from water. Journal of Chemical Technology and Biotechnology, 89(1), 150–157.
Ahmad, M., Rajapaksha, A. U., Lim, J. E., Zhang, M., Bolan, N., Mohan, D., et al. (2013b). Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere, 99, 19–33.
Barrett, E. P., Joyner, L. G., & Halenda, P. P. (1951). The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. Journal of the American Chemical Society, 73(1), 373–380.
Břendová, K., Tlustoš, P., Száková, J., & Habart, J. (2012). Biochar properties from different materials of plant origin. European Chemical Bulletin, 1(12), 535–539.
Brown, R. (2009). Biochar production technology. In J. Lehmann & S. Joseph (Eds.), Biochar for environmental management (pp. 127–146). London: Earthscan.
Cao, X., Pignatello, J. J., Li, Y., Lattao, C., Chappell, M. A., Chen, N., et al. (2012). Characterization of wood chars produced at different temperatures using advanced solid-state 13C NMR spectroscopic techniques. Energy and Fuels, 26(9), 5983–5991.
Chen, B., Zhou, D., & Zhu, L. (2008). Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environmental Science and Technology, 42(14), 5137–5143.
Chun, Y., Sheng, G., Chiou, C. T., & Xing, B. (2004). Compositions and sorptive properties of crop residue-derived chars. Environmental Science and Technology, 38(17), 4649–4655.
Delgado, A. V., González-Caballero, F., Hunter, R. J., Koopal, L. K., & Lyklema, J. (2007). Measurement and interpretation of electrokinetic phenomena. Journal of Colloid and Interface Science, 309(2), 194–224.
Dias, B. O., Silva, C. A., Higashikawa, F. S., Roig, A., & Sánchez-Monedero, M. A. (2010). Use of biochar as bulking agent for the composting of poultry manure: effect on organic matter degradation and humification. Bioresource Technology, 101(4), 1239–1246.
Downie, A., Crosky, A., & Munroe, P. (2009). Physical properties of biochar. In J. Lehmann & S. Joseph (Eds.), Biochar for environmental management: science and technology (pp. 13–32). London: Earthscan.
Enders, A., Hanley, K., Whitman, T., Joseph, S., & Lehmann, J. (2012). Characterization of biochars to evaluate recalcitrance and agronomic performance. Bioresource Technology, 114, 644–653.
Glaser, B., Lehmann, J., & Zech, W. (2002). Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—A review. Biology and Fertility of Soils, 35(4), 219–230.
Han, Y., Boateng, A. A., Qi, P. X., Lima, I. M., & Chang, J. (2013). Heavy metal and phenol adsorptive properties of biochars from pyrolyzed switchgrass and woody biomass in correlation with surface properties. Journal of Environmental Management, 118, 196–204.
Hossain, M. K., Strezov, V., Yin Chan, K., & Nelson, P. F. (2010). Agronomic properties of wastewater sludge biochar and bioavailability of metals in production of cherry tomato (Lycopersicon esculentum). Chemosphere, 78(9), 1167–1171.
Huang, L., Baumgartl, T., & Mulligan, D. (2012). Is rhizosphere remediation sufficient for sustainable revegetation of mine tailings? Annals of Botany, 110, 223–238.
Huang, L., Baumgartl, T., Zhou, L., & Mulligan, R. D. (2014). The new paradigm for phytostabilising mine wastes—Ecologically engineered pedogenesis and functional root zones, Life-of-Mine 2014 Conference. Brisbane: AUSIMM.
Hunter, R. J. (1981). Zeta potential in colloid science: Principles and applications. London: Academic Press.
Jackson, Michael, Eberhardt, Thomas, Boateng, Akwasi, Mullen, Charles, & Groom, Les. (2013). Evaluation of biochars by temperature programmed oxidation/mass spectrometry. BioResources., 8(4), 5461–5474.
Jiang, S., Huang, L., Nguyen, T. A. H., Ok, Y. S., Rudolph, V., Yang, H., et al. (2016). Copper and zinc adsorption by softwood and hardwood biochars under elevated sulphate-induced salinity and acidic pH conditions. Chemosphere, 142, 67–71.
Jiang, J., & Xu, R. (2013). Application of crop straw derived biochars to Cu(II) contaminated Ultisol: Evaluating role of alkali and organic functional groups in Cu(II) immobilization. Bioresource Technology, 133, 537–545.
Karaosmanoǧlu, F., Işıḡıgür-Ergüdenler, A., & Sever, A. (2000). Biochar from the straw-stalk of rapeseed plant. Energy and Fuels, 14(2), 336–339.
Keiluweit, M., Nico, P. S., Johnson, M. G., & Kleber, M. (2010). Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environmental Science and Technology, 44(4), 1247–1253.
Kim, P., Johnson, A. M., Essington, M. E., Radosevich, M., Kwon, W.-T., Lee, S.-H., et al. (2013). Effect of pH on surface characteristics of switchgrass-derived biochars produced by fast pyrolysis. Chemosphere, 90(10), 2623–2630.
Kuhlbusch, T. A. J. (1995). Method for determining black carbon in residues of vegetation fires. Environmental Science and Technology, 29(10), 2695–2702.
Lee, J. W., Kidder, M., Evans, B. R., Paik, S., Buchanan Iii, A. C., Garten, C. T., et al. (2010). Characterization of biochars produced from cornstovers for soil amendment. Environmental Science and Technology, 44(20), 7970–7974.
Li, X., Bond, P. L., Van Nostrand, J. D., Zhou, J., & Huang, L. (2015). From lithotroph- to organotroph-dominant: directional shift of microbial community in sulphidic tailings during phytostabilization. Scientific Reports, 5, 12978.
Li, J., Li, Y., Wu, Y., & Zheng, M. (2014). A comparison of biochars from lignin, cellulose and wood as the sorbent to an aromatic pollutant. Journal of Hazardous Materials, 280, 450–457.
Li, X., You, F., Huang, L., Strounina, E., & Edraki, M. (2013). Dynamics in leachate chemistry of Cu-Au tailings in response to biochar and woodchip amendments: A column leaching study. Environmental Sciences Europe, 25, 32.
Liang, B., Lehmann, J., Solomon, D., Kinyangi, J., Grossman, J., O’Neill, B., et al. (2006). Black carbon increases cation exchange capacity in soils. Soil Science Society of America Journal, 70(5), 1719–1730.
Link, S., Arvelakis, S., Spliethoff, H., De Waard, P., & Samoson, A. (2008). Investigation of biomasses and chars obtained from pyrolysis of different biomasses with solid-state 13C and 23Na nuclear magnetic resonance spectroscopy. Energy and Fuels, 22(5), 3523–3530.
López-Ramón, V., Moreno-Castilla, C., Rivera-Utrilla, J., & Radovic, L. R. (2003). Ionic strength effects in aqueous phase adsorption of metal ions on activated carbons. Carbon, 41(10), 2020–2022.
Lottermoser, B. G. (2010). Tailings, Mine Wastes (pp. 205–241). Berlin, Heidelberg: Springer.
Mathers, N. J., & Xu, Z. (2003). Solid-state 13C NMR spectroscopy: Characterization of soil organic matter under two contrasting residue management regimes in a 2-year-old pine plantation of subtropical Australia. Geoderma, 114(1–2), 19–31.
Moreno-Castilla, C., & Rivera-Utrilla, J. (2001). Carbon materials as adsorbents for the removal of pollutants from the aqueous phase. Materials Research Society Bulletin, 26(11), 890–894.
Mukherjee, A., Zimmerman, A. R., & Harris, W. (2011). Surface chemistry variations among a series of laboratory-produced biochars. Geoderma, 163(3–4), 247–255.
Mukome, F. N. D., Zhang, X., Silva, L. C. R., Six, J., & Parikh, S. J. (2013). Use of chemical and physical characteristics to investigate trends in biochar feedstocks. Journal of Agricultural and Food Chemistry, 61(9), 2196–2204.
Nguyen, B. T., Lehmann, J., Hockaday, W. C., Joseph, S., & Masiello, C. A. (2010). Temperature sensitivity of black carbon decomposition and oxidation. Environmental Science and Technology, 44(9), 3324–3331.
R Development Core Team (2011), R: A Language and Environment for Statistical Computing. Vienna, Austria: the R Foundation for Statistical Computing. ISBN: 3-900051-07-0. Available online at http://www.R-project.org/. Accessed 20 Feb 2016.
Rayment, G. E., & Higginson, F. R. (1992). Australian laboratory handbook of soil and water chemical methods. Melbourne: Inkata Press.
Rehrah, D., Bansode, R. R., Hassan, O., & Ahmedna, M. (2016). Physico-chemical characterization of biochars from solid municipal waste for use in soil amendment. Journal of Analytical and Applied Pyrolysis,. doi:10.1016/j.jaap.2015.12.022.
Ronsse, F., van Hecke, S., Dickinson, D., & Prins, W. (2013). Production and characterization of slow pyrolysis biochar: Influence of feedstock type and pyrolysis conditions. Global Change Biology Bioenergy, 5(2), 104–115.
Shinogi, Y., & Kanri, Y. (2003). Pyrolysis of plant, animal and human waste: Physical and chemical characterization of the pyrolytic products. Bioresource Technology, 90(3), 241–247.
Singh, B. P., Hatton, B. J., Singh, B., Cowie, A. L., & Kathuria, A. (2010a). Influence of biochars on nitrous oxide emission and nitrogen leaching from two contrasting soils. Journal of Environmental Quality, 39(4), 1224–1235.
Singh, B., Singh, B. P., & Cowie, A. L. (2010b). Characterisation and evaluation of biochars for their application as a soil amendment. Soil Research, 48(7), 516–525.
Slattery, W., Ridley, A., & Windsor, S. (1991). Ash alkalinity of animal and plant products. Australian Journal of Experimental Agriculture, 31(3), 321–324.
Song, W., & Guo, M. (2012). Quality variations of poultry litter biochar generated at different pyrolysis temperatures. Journal of Analytical and Applied Pyrolysis, 94, 138–145.
Spokas, K. A. (2010). Review of the stability of biochar in soils: Predictability of O: C molar ratios. Carbon Management, 1(2), 289–303.
Uchimiya, M. (2011). Biochar characteristics and function as a heavy metal sorbent in soil: Role of surface ligands. In Abstracts of Papers of the American Chemical Society, 242.
Uchimiya, M., Lima, I. M., Thomas Klasson, K., Chang, S. C., Wartelle, L. H., & Rodgers, J. E. (2010). Immobilization of heavy metal ions (CuII, CdII, NiII, and PbII) by broiler litter-derived biochars in water and soil. Journal of Agricultural and Food Chemistry, 58(9), 5538–5544.
Van Zwieten, L., Kimber, S., Downie, A., Morris, S., Petty, S., Rust, J., et al. (2010). A glasshouse study on the interaction of low mineral ash biochar with nitrogen in a sandy soil. Soil Research, 48(7), 569–576.
You, F. (2015). Rehabilitation of organic carbon and microbial community structure and functions in Cu-Pb-Zn mine tailings for in situ engineering technosols sustainable minerals institute (p. 79). St Lucia: The University of Queensland.
Yuan, J., & Xu, R. (2011). The amelioration effects of low temperature biochar generated from nine crop residues on an acidic Ultisol. Soil Use and Management, 27(1), 110–115.
Yuan, J., Xu, R., & Zhang, H. (2011). The forms of alkalis in the biochar produced from crop residues at different temperatures. Bioresource Technology, 102(3), 3488–3497. doi:10.1016/j.biortech.2010.11.018.
Zhao, L., Cao, X., Mašek, O., & Zimmerman, A. (2013). Heterogeneity of biochar properties as a function of feedstock sources and production temperatures. Journal of Hazardous Materials, 256–257, 1–9.
Acknowledgments
The present study was partially funded by the UWA-UQ bilateral collaboration grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, S., Nguyen, T.A.H., Rudolph, V. et al. Characterization of hard- and softwood biochars pyrolyzed at high temperature. Environ Geochem Health 39, 403–415 (2017). https://doi.org/10.1007/s10653-016-9873-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-016-9873-6