Abstract
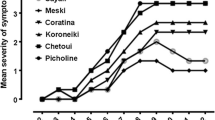
Natural recovery from wilt disease symptoms was evaluated in young olive trees root dip inoculated with Verticillium dahliae in a growth chamber over a 12 week period and, later on, when the trees were transplanted in a V. dahliae-free soil in a lathhouse during a period of 127 weeks. Recovery in an individual tree was considered when a plant showed symptom remission after having reached a maximum value of symptom severity. Recovery accounted for 53% of 464 trees that showed wilt symptoms during observations in the two environments. The remaining trees died. Recurrent wilt symptoms were not observed in recovered trees, and recovery was usually accompanied by the production of new green tissues. Recovery was clearly higher in trees inoculated with a non-defoliating (ND) isolate (86.4%) of the pathogen than in those inoculated with a defoliating (D) isolate (23.9%). The percentage of recovery and the level of resistance were significantly correlated. Recovery accounted for 92.1% of the cases in resistant and moderately susceptible cultivars, reaching 100% in plants inoculated with the ND isolate (Table 2); meanwhile it was three times lower (30.1% of the plants) in susceptible and extremely susceptible diseased trees. In the lathhouse, periodical tissue isolations for monitoring the progress of infections over a period of 127 weeks in recovered trees, showed that the pathogen could only be isolated from trees 19 weeks after inoculation. Pathogen isolation was significantly higher from susceptible and extremely susceptible cultivars (84.6%) than from resistant and moderately susceptible ones (33.3%). Results showed that if a tree overcomes infection by pathogen from a single inoculation, and it is able to begin a recovery process, it will not express wilt symptoms again in a pathogen-free environment. The pathogen remained inactive or dead over time in recovered trees. Thus, new infections from rootlets would be necessary for new symptom expression. Recovery from Verticillium wilt is an important natural mechanism that occurs in a high percentage of infected olive trees, and can complement the resistance of the cultivar, particularly in conditions of low inoculum densities of low virulence isolates of the pathogen in the soil.
Similar content being viewed by others
References
Blanco-López MA, Rodríguez-Jurado D and Jiménez-Díaz RM (1990) Incidence and seasonal variation of Verticillium wilt in olive orchards. In: Proceedings of the 5th International Verticillium Symposium (p. 5) Leningrad, USSR.
Ciccarese F, Frisullo S and Cirulli M (1990) Natural disease recovery from Verticillium wilt in peach. In: Proceedings of the 5th International Verticillium Symposium (p. 6) Leningrad, USSR.
Cirulli M, Amenduni M and Paplomatas EJ (1998) Verticillium wilts of major tree hosts; Stone fruits. In: Compendium of Verticillium Wilt in Tree Species (pp. 17–20) Ponsen & Looijen, Wageningen, The Netherlands.
AM Emechebe CLA Leakely WB Banage (1974) ArticleTitleVerticillium wilt of cacao in Uganda: Wilt induction by mechanical vessel blockage and mode of recovery of diseased plants East African Agricultural Forestry Journal 39 337–343
Hiemstra JA (1995a) Verticillium wilt of Fraxinus excelsior. Ph.D. Thesis, Wageningen Agricultural University, The Netherlands.
JA Hiemstra (1995b) ArticleTitleRecovery of verticillium-infected ash trees Phytoparasitica 23 64–65
Hiemstra JA (1998) Some general features of Verticillium wilt in tress. In: Compendium of Verticillium Wilt in Tree Species (pp. 5–110) Ponsen & Looijen, Wageningen, The Netherlands.
ML Lacy CE Horner (1966) ArticleTitleBehaviour of Verticillium dahliae in the rizosphere and on roots of plants susceptible, resistant, and immune to wilt Phytopathology 56 427–430
BA Latorre PT Allende (1983) ArticleTitleOccurrence and incidence of Verticillium wilt on Chilean avocado groves Plant Disease 67 445–447
AG Levin S Lavee L Tsror (Lahkim) (2003) ArticleTitleEpidemiology and effects of Verticillium wilt on yield of olive trees (cvs. Barnea and Souri) irrigated with saline water in Israel Phytoparasitica 31 333–343
FJ López-Escudero MA Blanco-López (2001) ArticleTitleEffect of a single or double soil solarization to control Verticillium wilt in established olive orchards in Spain Plant Disease 85 489–496
FJ López-Escudero C delRío JM Caballero MA Blanco-López (2004) ArticleTitleEvaluation of olive cultivars for resistance to Verticillium dahliae European Journal of Plant Pathology 110 79–85
Martos-Moreno C, Caballero JM, del Río C and Blanco-López MA (2001) Epidemiological behaviour of olive cultivars in orchards infested with mixtures of defoliating and non-defoliating isolates of Verticillium dahliae. In: Proceedings of the 8th International Verticillium Symposium (p. 67) Cordoba, Spain.
Paplomatas EJ and Elena K (1998) Verticillium wilts of major tree hosts; Pistachio. In: Compendium of Verticillium wilt in Tree Species (pp. 31–32) Ponsen & Looijen, Wageningen, The Netherlands.
MLV Resende J Flood RM Cooper (1995) ArticleTitleEffect of method of inoculation, inoculum density and seedling age at inoculation on the expression of resistance of cocoa (Theobroma cacao L.) to Verticillium dahliae Kleb Plant Patholology 43 104–111
Rodríguez-Jurado D (1993) Interacciones huésped-parásito en la marchitez del olivo (Olea europaea L.) inducida por Verticillium dahliae Kleb. Ph.D. Thesis, Universidad de Córdoba, Spain.
Schnathorst WC (1981) Life cycle and epidemiology of Verticillium. In: Fungal Wilt Diseases of Plants (pp. 81–111) Academic Press, New York.
AL Shigo (1984) ArticleTitleCompartmentalization: A conceptual framework for understanding how tree grow and defend themselves Annual Review of Phytopathology 22 189–214 Occurrence Handle10.1146/annurev.py.22.090184.001201
WA Sinclair KL Smith AO Larsen (1981) ArticleTitleVerticillium wilt of maples: Symptoms related to the movement of the pathogens in stems Phytopathology 71 340–345
Talboys PW (1968) Water deficits in vascular disease. In: Water Deficits and Plant Growth. Vol II: Plant Water Consumption and Response, (pp. 255–311) Academic Press, New York and London.
JB Taylor NT Flentje (1968) ArticleTitleInfection, recovery from infection and resistance of apricot trees to Verticillium albo-atrum New Zealand Journal of Botany 61 417–426
JT Tippett AL Shigo (1981) ArticleTitleBarrier zone formation: A mechanism of tree defense against vascular pathogens IAWA bulletin n.s. 2 163–168
EC Tjamos DA Biris EJ Paplomatas (1991) ArticleTitleRecovery of olive trees with Verticillium wilt after individual application of soil solarization in established olive orchards Plant Disease 75 557–562
A Vigouroux (1975) ArticleTitleVerticillium dahliae, agent d'un déperissement de l'olivier en France Annals Phytopatholgie 7 37–44
A Vigouroux C Castelain (1969) ArticleTitleLa verticilliose de l’abricotier. I. Premières observations sur les symptômes et l’èvolution de la maladie. Hypothèses sur quelques facteurs de variation Annals Phytopathologie 1 427–448
S Wilhelm J Taylor Bruce (1965) ArticleTitleControl of Verticillium wilt in olive trough natural recovery and resistance Phytopathology 55 310–316
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
López-Escudero, F.J., Blanco-López, M.A. Recovery of Young Olive Trees from Verticillium dahliae. Eur J Plant Pathol 113, 367–375 (2005). https://doi.org/10.1007/s10658-005-3145-0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10658-005-3145-0