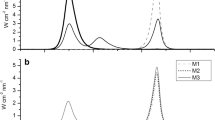
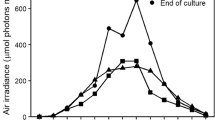
Abstract
The microalga Haematococcus pluvialis Flotow has been the subject of a number of studies concerned with maximizing astaxanthin production for use in animal feeds and for human consumption. Several of these studies have specifically attempted to ascertain the optimal temperature and irradiance combination for growth of H. pluvialis, but there has been a great deal of disagreement between laboratories. “Ideal” levels of temperature and irradiance have been reported to range from 14 to 28°C and 30 to 200 μmol photons m−2 s−1. The objective of the present study was to simultaneously explore temperature and irradiance effects for a single strain of H. pluvialis (UTEX 2505) across an experimental region that encompassed the reported “optimal” combinations of these factors for multiple strains. To this end, a two-dimensional experimental design based on response surface methodology (RSM) was created. Maximum growth rates for UTEX 2505 were achieved at 27°C and 260 μmol photons m−2 s−1, while maximum quantum yield for stable charge separation at PSII (Fv/Fm) was achieved at 27°C and 80 μmol photons m−2 s−1. Maximum pigment concentrations correlated closely with maximum Fv/Fm. Numeric optimization of growth rate and Fv/Fm produced an optimal combination of 27°C and 250 μmol photons m−2 s−1. Polynomial models of the various response surfaces were validated with multiple points and were found to be very useful for predicting several H. pluvialis UTEX 2505 responses across the entire two-dimensional experimental design space.






Similar content being viewed by others
References
Allen DM (1971) Mean square error of prediction as a criterion for selecting variables. Technometrics 13:469–475
Anderson MJ, Whitcomb PJ (2005) RSM simplified: optimizing processes using response surface methods for design of experiments. Productivity Press, New York
Belsley DA, Kuh E, Welsch RE (1980) Regression diagnostics: identifying influential data and sources of collinearity. Wiley, New York
Borowitzka MA, Huisman JM, Osborn A (1991) Culture of the astaxanthin-producing green alga Haematococcus pluvialis. I. Effects of nutrients on growth and cell type. J Appl Phycol 3:295–304
Boussiba S (2000) Carotenogenesis in the green alga Haematococcus pluvialis: cellular physiology and stress response. Physiol Plant 108:111–117
Boussiba S, Vonshak A (1991) Astaxanthin accumulation in the green alga Haematococcus pluvialis. Plant Cell Physiol 32:1077–1082
Boussiba S, Bing W, Yuan J-P, Zarka A, Chen F (1999) Changes in pigments profile in the green alga Haematococcus pluvialis exposed to environmental stresses. Biotechnol Lett 21:601–604
Box GEP (1979) Robustness in the strategy of scientific model building. In: Launer RL, Wilkinson GN (eds) Robustness in statistics. Academic, New York, pp 201–236
Box GEP, Cox DR (1964) An analysis of transformations (with discussion). J Royal Statistical Soc Ser B 26:211–246
Box GEP, Draper NR (1971) Factorial designs, the X’X criterion, and some related matters. Technometrics 13:731–741
Butler WL (1978) Energy distribution of the photochemical apparatus of photosynthesis. Annu Rev Plant Physiol 29:345–378
Chaumont D, Thèpenier C (1995) Carotenoid content in growing cells of Haematococcus pluvialis during a sunlight cycle. J Appl Phycol 7:529–537
Cifuentes AS, González MA, Vargas S, Hoeneisen M, González N (2003) Optimization of biomass, total carotenoids and astaxanthin production in Haematococcus pluvialis flotow strain steptoe (Nevada, USA) under laboratory conditions. Biol Res 36:343–357
Cook RD, Weisberg S (1982) Residuals and influence in regression. Chapman Hall, New York
Del Rio E, Acién G, García-Malea MC, Rivas J, Molina-Grima E, Guerrero MG (2005) Efficient one-step production of astaxanthin by the microalga Haematococcus pluvialis in continuous culture. Biotechnol Bioeng 91:808–815
Derringer G, Suich R (1980) Simultaneous optimization of several response variables. J Qual Technol 12:214–219
Di Mascio P, Murphy ME, Sies H (1991) Antioxidant defense systems: the role of carotenoids, tocopherols, and thiols. Am J Clin Nutr 53:194S–200S
Domínguez-Bocanegra AR, Legarreta IG, Jeronimo FM, Campocosio AT (2004) Influence of environmental and nutritional factors in the production of astaxanthin from Haematococcus pluvialis. Bioresource Technol 92:209–214
Elliot AM (1931) Morphology and life history of Haematococcus pluvialis. Arch Protistenk 82:250–272
Fábregas J, Domínguez A, Regueiro M, Maseda A, Otero A (2000) Optimization of culture medium for the continuous cultivation of the microalga Haematococcus pluvialis. Appl Microbiol Biotechnol 53:530–535
Fábregas J, Domínguez A, Maseda A, Otero A (2003) Interactions between irradiance and nutrient availability during astaxanthin accumulation and degradation in Haematococcus pluvialis. Appl Microbiol Biotechnol 61:545–551
Fan L, Vonshak A, Boussiba S (1994) Effect of temperature and irradiance on growth of Haematococcus pluvialis (Chlorophyceae). J Phycol 30:829–833
Foss P, Storebakken T, Schiedt K, Liaaen-Jensen S, Austreng E, Streiff K (1984) Carotenoids in diets for salmonids. I. Pigmentation of rainbow trout with the individual optical isomers of astaxanthin in comparison with canthaxanthin. Aquaculture 41:213–226
Genty B, Briantais J-M, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Gong X, Chen F (1997) Optimization of culture medium for growth of Haematococcus pluvialis. J Appl Phycol 9:437–444
Goodwin TW, Jamikorn M (1954) Studies in carotogenesis: 11. Carotenoid synthesis in the alga Haematococcus pluvialis. Biochem J 57:376–381
Grünewald K, Hagen C, Braune W (1997) Secondary carotenoid accumulation in flagellates of the green alga Haematococcus lacustris. Eur J Phycol 32:387–392
Guerin M, Huntley ME, Olaizola M (2003) Haematococcus astaxanthin: applications for human health and nutrition. Trends Biotechnol 21:210–216
Hagen C, Grünewald K, Xyländer M, Rothe E (2001) Effect of cultivation parameters on growth and pigment biosynthesis in flagellated cells of Haematococcus pluvialis. J Appl Phycol 13:79–87
Hahn GJ, Meeker WQ (1991) Statistical intervals: a guide for practitioners. Wiley & Sons, New York
Harker M, Tsavalos AJ, Young AJ (1995) Use of response surface methodology to optimize carotenogenesis in the microalga, Haematococcus pluvialis. J Appl Phycol 7:399–406
Harker M, Tsavalos AJ, Young AJ (1996) Factors responsible for astaxanthin formation in the chlorophyte Haematococcus pluvialis. Bioresource Technol 55:207–214
Havaux M, Strasser RJ, Greppin H (1991) A theoretical and experimental analysis of the qP and qN coefficients of chlorophyll fluorescence quenching and their relation to photochemical and nonphotochemical events. Photosynth Res 27:41–55
Inborr J (1998) Haematococcus, the poultry pigmentor. Feed Mix 6:31–34
Jeon Y-C, Cho C-W, Yun Y-S (2006) Combined effects of light intensity and acetate concentration on the growth of unicellular microalga Haematococcus pluvialis. Enzyme Microbial Technol 39:490–495
Kobayashi M, Kakizono T, Nagai S (1991) Astaxanthin production by a green alga, Haematococcus pluvialis accompanied with morphological changes in acetate media. J Ferment Bioeng 71:335–339
Kobayashi M, Kakizono T, Nagai S (1993) Enhanced carotenoid biosynthesis by oxidative stress in acetate-induced cyst cells of a green unicellular alga, Haematococcus pluvialis. Appl Environ Microbiol 59:867–873
Kobayashi M, Kurimura Y, Kakizono T, Nishio N, Tsuji Y (1997) Morphological changes in the life cycle of the green alga Haematococcus pluvialis. J Ferment Bioeng 84:94–97
Kvíderová J, Lukavský J (2001) A new unit of crossed gradients of temperature and light. In: Elster J, Seckbach J, Vincent WF, Lhotsky O (eds) Algae and extreme environments. Nova Hedwigia 123:539–548
Lee YK, Ding SY (1994) Cell cycle and accumulation of astaxanthin in Haematococcus lacustris (Chlorophyta). J Phycol 30:445–449
Lee YK, Soh CW (1991) Accumulation of astaxanthin in Haematococcus lacustris (Chlorophyta). J Phycol 27:575–577
Lorenz RT, Cysewski GR (2000) Commercial potential for Haematococcus microalgae as a natural source of astaxanthin. Trends Biotechnol 18:160–167
MacIntyre HL, Cullen JJ (2005) Using cultures to investigate the physiological ecology of microalgae. In: Andersen RA (ed) Algal culturing techniques. Elsevier, London, pp 287–326
MacIntyre HL, Kana TM, Anning T, Geider RJ (2002) Photoacclimation of photosynthesis irradiance response curves and photosynthetic pigments in microalgae and cyanobacteria. J Phycol 38:17–38
Margalith PZ (1999) Production of ketocarotenoids by microalgae. Appl Microbiol Biotechnol 51:431–438
Meyers SF, Latscha L (1997) Carotenoids. In: D’Abramo LR, Conklin DE, Akiyama DM (eds) Crustacean nutrition advances in world aquaculture, vol. 6. The World Aquaculture Society, Baton Rouge, LA, USA, pp 164–186
Miller NJ, Sampson J, Candeias LP, Bramley PM, Rice-Evans CA (1996) Antioxidant activities of carotenes and xanthophylls. FEBS Lett 384:240–242
Montgomery DC, Peck EA, Vining GG (2001) Introduction to linear regression analysis, 3rd edn. Wiley, New York
Moya MJ, Sánchez-Guardamino ML, Vilavella A, Barberà E (1997) Growth of Haematococcus lacustris: a contribution to kinetic modeling. J Chem Tech Biotechnol 68:303–309
Myers RH (1990) Classical and modern regression with applications, 2nd edn. PWS-KENT, Boston
Myers RH, Montgomery DC (2002) Response surface methodology: process and product optimization using designed experiments, 2nd edn. Wiley, New York
Nelder JA (1998) The selection of terms in response surface models — how strong is the weak heredity principle? Am Stat 52:315–318
Nelder JA, Mead R (1965) A simplex method for function minimization. Comput J 7:308–313
Niedz RP, Evens TJ (2006) A solution to the problem of ion confounding in experimental biology. Nature Methods 3:417
Olaizola M (2000) Commercial production of astaxanthin from Haematococcus pluvialis using 25,000-liter outdoor photobioreactors. J Appl Phycol 12:499–506
Peixoto J (1990) A property of well-formulated polynomial regression models. Am Stat 44:26–30
Rao AR, Sarada R, Ravishankar GA (2007) Stabilization of astaxanthin in edible oils and its use as antioxidant. J Sci Food Agric 87:957–965
Richardson K, Beardall J, Raven JA (1983) Adaptation of unicellular algae to irradiance: an analysis of strategies. New Phytol 93:157–191
Santos MF, Mesquita JF (1984) Ultrastructural study of Haematococcus lacustris (Girod) Rostafinski (Volvocales). I. Some aspects of carotenogenesis. Cytologia 49:215–228
Schreiber U, Bilger W (1993) Progress in chlorophyll fluorescence research: major developments during the past years in retrospect. Prog Bot 54:151–173
Schiedt K, Leuenberger FJ, Vecchi M, Glinz E (1985) Absorption, retention and metabolic transformations of carotenoids in rainbow trout, salmon and chicken. Pure Appl Chem 57:685–692
Schreiber U, Schliwa U, Bilber B (1986) Continuous recording of photochemical and non-photochemical chlorophyll fluorescence quenching with a new type of modulation fluorometer. Photosynth Res 10:51–62
Torzillo G, Göksan T, Isik O, Gökpinar S (2005) Photon irradiance required to support optimal growth and interrelations between irradiance and pigment composition in the green alga Haematococcus pluvialis. Eur J Phycol 40:233–240
Van Heukelem L, Thomas CS (2001) Computer-assisted high performance liquid chromatography method development with applications to the isolation and analysis of phytoplankton pigments. J Chrom A 910:31–49
Weisberg S (1985) Applied linear regression, 2nd edn. Wiley & Sons, New York
Yuan J-P, Chen F (1997) Identification of astaxanthin isomers in Haematococcus lacustris by HPLC-photodiode array detection. Biotechnol Techniques 11:455–459
Acknowledgements
We would like to thank Mr. Peter D’Aiuto for excellent technical assistance with culturing H. pluvialis and Ms. Rita Bowker for assistance with the HPLC measurements. We would also like to thank the folks at Stat-Ease for the extremely informative discussions on the various statistical aspects of this research. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Evens, T.J., Niedz, R.P. & Kirkpatrick, G.J. Temperature and irradiance impacts on the growth, pigmentation and photosystem II quantum yields of Haematococcus pluvialis (Chlorophyceae). J Appl Phycol 20, 411–422 (2008). https://doi.org/10.1007/s10811-007-9277-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-007-9277-1