Abstract
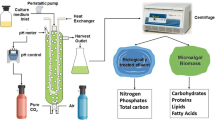
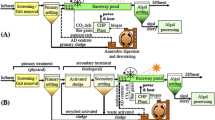
Northern regions are generally viewed as unsuitable for microalgal biofuel production due to unfavorable climate and solar insolation levels. However, these conditions can potentially be mitigated by coupling microalgal cultivation to industrial processes such as wastewater treatment. In this study, we have examined the biomass and lipid productivity characteristics of 14 microalgae isolates (Chlorophyta) from the Canadian province of Saskatchewan. Under both photoautotrophic and mixotrophic cultivation, a distinct linear trend was observed between biomass and lipid productivities in the 14 SK isolates. The most productive strain under cultivation in TAP media was Scenedesmus sp.-AMDD which displayed rates of biomass and fatty acid productivities of 80 and 30.7 mg L−1 day−1, respectively. The most productive strain in B3NV media was Chlamydomonas debaryana-AMLs1b which displayed rates of biomass and fatty acid productivities of 51.7 and 5.9 mg L−1 day−1, respectively. In 11 of the isolates tested, secondary municipal wastewater (MCWW) supported rates of biomass productivity between 21 and 33 mg L−1 day−1 with Scenedesmus sp.-AMDD being the most productive. Three strains, Chlamydomonas debaryana-AMB1, Chlorella sorokiniana-RBD8 and Micractinium sp.-RB1b, showed large increases in biomass productivity when cultivated mixotrophically in MCWW supplemented with glycerol. High relative oleic acid content was detected in 10 of the 14 isolates when grown mixotrophically in media supplemented with acetate. There was no detectable effect on the fatty acid profiles in cells cultivated mixotrophically in glycerol-supplemented MCWW. These data indicate that biomass and lipid productivities are boosted by mixotrophic cultivation. Exploiting this response in municipal wastewater is a promising strategy for the production of environmentally sustainable biofuels.




Similar content being viewed by others
References
Anderson RA (ed) (2005) Algal culturing techniques. Elsevier Academic Press, London, 578 pp
Apt KE, Behrens PW (1999) Commercial developments in microalgal biotechnology. Review. J Phycol 35:215–216
Benemann JR (2003) Biofixation of CO2 and greenhouse gas abatement with microalgae-technology roadmap. Prepared for the U.S. Department of Energy National Energy Technology Laboratory, No. 7010000926
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25:294–306
Christie WW (1989) Gas chromatography and lipids: a practical guide. The Oily Press, Ayr
Christie WW (2003) Lipid analysis: isolation, separation, identification and structural analysis of lipids. The Oily Press, Bridgewater
Clarens AF, Resurreccion EP, White MA, Colosi LM (2010) Environmental life cycle comparison of algae to other bioenergy feedstocks. Environ Sci Technol 44:1813–1819
Ehimen EA, Sun ZF, Carrington CG (2010) Variables affecting the in situ transesterification of microalgae lipids. Fuel 89:677–684
González-López CV, Garcia MCC, Fernandez FGA, Bustos CS, Chisti Y, Sevilla JMF (2010) Protein measurements of microalgal and cyanobacterial biomass. Biores Technol 101:7587–7591
Gorman DS, Levine RP (1965) Cytochrome f and plastocyanin: their sequence in the photosynthetic electron transport chain of Chlamydomonas reinhardii. Proc Natl Acad Sci USA 54:1665–1669
Griffiths MJ, Harrison STL (2009) Lipid productivity as a key characteristic for choosing algal species for biodiesel production. J Appl Phycol 21:493–507
Griffiths MJ, Hille RP, Harrison STL (2010) Selection of direct transesterification as the preferred method for assay of fatty acid content of microalgae. Lipids 45:1053–1060
Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M, Darzins A (2008) Microalgal triacyglycerol as feedstock for biofuel production: perspectives and advances. Plant J 54:621–639
Knothe G (2006) Analyzing biodiesel: standards and other methods. J Am Oil Chem Soc 83:823–833
Kong QX, Li L, Martinez B, Chen P, Ruan R (2010) Culture of microalgae Chlamydomonas reinhardtii in wastewater for biomass feedstock production. Appl Biochem Biotechnol 160:9–18
Lee YK (2001) Microalgal mass culture systems and methods: their limitation and potential. J Appl Phycol 13:307–315
McGinn PJ, Dickinson KE, Bhatti S, Frigon JC, Guiot SR, O’Leary SJB (2011) Integration of microalgae cultivation with industrial waste remediation for biofuel and bioenergy production: opportunities and limitations. Photosyn Res 109:231–247
Ogawa T, Aiba S (1981) Bioenergetic analysis of mixotrophic growth in Chlorella vulgaris and Scenedesmus acutus. Biotechnol Bioeng 23:1121–1132
Ogbonna JC, Tanaka H (1998) Cyclic autotrophic/heterotrophic cultivation of photosynthetic cells: a method of achieving continuous cell growth under light/dark cycles. Biores Technol 65:65–72
Oswald WJ (2003) My sixty years in applied algology. J Appl Phycol 15:99–106
Oswald WJ, Golueke C (1960) Biological transformation of solar energy. Adv Appl Microbiol 2:223–262
Park JBK, Craggs RJ (2010) Wastewater treatment and algal production in high rate algal ponds with carbon dioxide addition. Water Sci Technol 61:633–639
Park JBK, Craggs RJ, Shilton AN (2011) Wastewater treatment high rate algal ponds for biofuel production. Biores Technol 102:35–42
Perez-Garcia O, Bashan Y, Puente ME (2001) Organic carbon supplementation of sterilized municipal wastewater is essential for heterotrophic growth and removing ammonium by the microalga Chlorella vulgaris. J Phycol 47:190–199
Powell N, Shilton A, Pratt S, Chisti Y (2011) Luxury uptake of phosphorous by microalgae in full-scale waste stabilization ponds. Water Sci Technol 63:704–709
Sheehan J, Dunahay T, Benemann J, Roessler P (1998) A look back at the U.S. Department of Energy’s Aquatic Species Program—biodiesel from algae. National Renewable Energy Program. NREL/TP-580-24190
Sukenik A, Zmora O, Carmeli Y (1993) Biochemical quality of marine unicellular algae with special emphasis on lipid composition. II. Nannochloropsis sp. Aquacult 117:313–326
Talbot P, de la Noüe J (1993) Tertiary treatment of wastewater with Phormidium bohneri (Schmidle) under various light and temperature conditions. Water Res 27:153–159
Tran HL, Hong SJ, Lee CG (2009) Evaluation of extraction methods for recovery of fatty acids from Botryococcus braunii LB 572 and Synechocystis sp. PCC 6803. Biotechnol Bioprocess Eng 14:187–192
Wan M, Liu P, Xia J, Rosenberg JN, Oyler GA, Betenbaugh MJ, Nie Z, Qiu G (2011) The effect of mixotrophy on microalgal growth, lipid content, and expression levels of three pathway genes in Chlorella sorokiniana. Appl Microbiol Biotechnol 91:835–844
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press Inc, New York, pp 315–322
Yang C, Hua Q, Shimizu K (2000) Energentics and carbon metabolism during growth of microalgal cells under photoautotrophic, mixotrophic and cyclic light-autotrophic/dark-heterotrophic conditions. Biochem Eng J 6:87–102
Zhu CJ, Lee YK (1997) Determination of biomass dry weight of marine microalgae. J Appl Phycol 9:189–194
Acknowledgements
We would like to thank staff members Dereck Avery, Shawn Taylor and Nigel Crouse of Mill Cove Wastewater Treatment Plant, Bedford, Nova Scotia, for their kind support and access to secondary effluent. We would also like to thank Shabana Bhatti for the helpful comments which improved the manuscript. This work was supported by the AAFC-NRCan-NRC’s National Bioproducts Program. The work performed in Kenneth Wilson’s laboratory was supported by the University of Saskatchewan and the Canadian Foundation for Innovation. This is NRC publication no. 54076.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, K.C., Whitney, C., McNichol, J.C. et al. Mixotrophic and photoautotrophic cultivation of 14 microalgae isolates from Saskatchewan, Canada: potential applications for wastewater remediation for biofuel production. J Appl Phycol 24, 339–348 (2012). https://doi.org/10.1007/s10811-011-9772-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-011-9772-2