Abstract
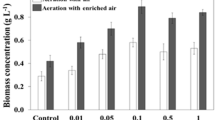
The aim of the present study was to survey the growth and astaxanthin production of E17, an astaxanthin-rich mutant of Chlorella zofingiensis, through feeding the low-cost carbon source cane molasses. In heterotrophic batch cultivation, E17 fed with pretreated molasses achieved biomass (1.79 g L−1 day−1) and astaxanthin (1.99 mg L−1 day−1) productivities comparable to those with glucose, which were about 2- and 2.8-fold of those fed with untreated molasses, respectively. Molasses-induced astaxanthin accumulation may be attributed to the elicited expression of carotenogenic genes, in particular the genes specifically responsible for the ketolation and hydroxylation of β-carotene to form astaxanthin. A two-stage fed-batch strategy was employed to grow E17 and induce astaxathin accumulation, resulting in 45.6 g L−1 biomass and 56.1 mg L−1 astaxanthin, the highest volumetric astaxanthin yield ever reported for this alga. In addition, the astaxanthin production by E17 was tested with a semi-continuous culture method, where the directly diluted raw molasses (giving 5 g L−1 sugar) was used as the carbon source. Little growth inhibition of E17 was observed in the semi-continuous culture with a biomass productivity of 1.33 g L−1 day−1 and an astaxanthin productivity of 0.83 mg L−1 day−1. The mixotrophic semi-continuous cultures enhanced the biomass and astaxanthin productivities by 29.3 % and 42.2 %, respectively. This study highlights the potential of using the industrially cheap cane molasses towards large-scale cost-saving production of the high-value ketocarotenoid astaxanthin.





Similar content being viewed by others
References
Antia NJ, Harrison PJ, Oliveira L (1991) The role of dissolved organic nitrogen in phytoplankton nutrition, cell biology and ecology. Phycologia 30:1–89
Boussiba S, Bing W, Yuan J-P, Zarka A, Chen F (1999) Changes in pigments profile in the green alga Haeamtococcus pluvialis exposed to environmental stresses. Biotechnol Lett 21:601–604
Fawcett JK, Scott JE (1960) A rapid and precise method for the determination of urea. J Clin Pathol 13:156–159
Feng P, Deng Z, Hu Z, Fan L (2011) Lipid accumulation and growth of Chlorella zofingiensis in flat plate photobioreactors outdoors. Bioresour Technol 102:10577–10584
Fraser PD, Bramley PM (2004) The biosynthesis and nutritional uses of carotenoids. Prog Lipid Res 43:228–265
Guerin M, Huntley ME, Olaizola M (2003) Haematococcus astaxanthin: applications for human health and nutrition. Trends Biotechnol 21:210–216
Higuera-Ciapara I, Felix-Valenzuela L, Goycoolea FM (2006) Astaxanthin: a review of its chemistry and applications. Crit Rev Food Sci Nutr 46:185–196
Ip PF, Chen F (2005) Production of astaxanthin by the green microalga Chlorella zofingiensis in the dark. Process Biochem 40:733–738
Ip PF, Wong KH, Chen F (2004) Enhanced production of astaxanthin by the green microalga Chlorella zofingiensis in mixotrophic culture. Process Biochem 39:1761–1766
Jiang L, Wang J, Liang S, Wang X, Cen P, Xu Z (2009) Butyric acid fermentation in a fibrous bed bioreactor with immobilized Clostridium tyrobutyricum from cane molasses. Bioresour Technol 100:3403–3409
Li Y, Huang J, Sandmann G, Chen F (2008) Glucose sensing and the mitochondrial alternative pathway are involved in the regulation of astaxanthin biosynthesis in the dark-grown Chlorella zofingiensis (Chlorophyceae). Planta 228:735–743
Liu Y-P, Zheng P, Sun Z-H, Ni Y, Dong J-J, Zhu L-L (2008) Economical succinic acid production from cane molasses by Actinobacillus succinogenes. Bioresour Technol 99:1736–1742
Liu C-Z, Wang F, Ou-Yang F (2009) Ethanol fermentation in a magnetically fluidized bed reactor with immobilized Saccharomyces cerevisiae in magnetic particles. Bioresour Technol 100:878–882
Liu J, Zhong Y, Sun Z, Huang J, Sandmann G, Chen F (2010a) One amino acid substitution in phytoene desaturase makes Chlorella zofingiensis resistant to norflurazon and enhances the biosynthesis of astaxanthin. Planta 232:61–67
Liu J, Huang J, Fan KW, Jiang Y, Zhong Y, Sun Z, Chen F (2010b) Production potential of Chlorella zofingienesis as a feedstock for biodiesel. Bioresour Technol 101:8658–8663
Liu J, Sun Z, Zhong Y, Huang J, Hu Q, Chen F (2012a) Stearoyl-acyl carrier protein desaturase gene from the oleaginous microalga Chlorella zofingiensis: cloning, characterization and transcriptional analysis. Planta 236:1665–1676
Liu J, Huang J, Jiang Y, Chen F (2012b) Molasses-based growth and production of oil and astaxanthin by Chlorella zofingiensis. Bioresour Technol 107:393–398
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 25:402–408
Lorenz RT, Cysewski GR (2000) Commercial potential for Haematococcus microalgae as a natural source of astaxanthin. Trends Biotechnol 18:160–167
Masojidek J, Sergejevova M, Rottnerova K, Jirka V, Korecko J, Kopecky J, Zatkova I, Torzillo G, Stys D (2009) A two-stage solar photobioreactor for cultivation of microalgae based on solar concentrators. J Appl Phycol 21:55–63
Meijer EA, Wijffels RH (1998) Development of a fast, reproducible and effective method for the extraction and quantification of proteins of micro-algae. Biotechnol Tech 12:353–358
Orosa M, Valero JF, Herrero C, Abalde J (2001) Comparison of the accumulation of astaxanthin in Haematococcus pluvialis and other green microalgae under N-starvation and high light conditions. Biotechnol Lett 23:1079–1085
Orosa M, Franqueira D, Cid A, Abalde J (2005) Analysis and enhancement of astaxanthin accumulation in Haematococcus pluvialis. Bioresour Technol 96:373–378
Renaud SM, Thinh L-V, Parry DL (1999) The gross chemical composition and fatty acid composition of 18 species of tropical Australian microalgae for possible use in mariculture. Aquaculture 170:147–159
Sharma A, Vivekanand V, Singh RP (2008) Solid-state fermentation for gluconic acid production from sugarcane molasses by Aspergillus niger ARNU-4 employing tea waste as the novel solid support. Bioresour Technol 99:3444–3450
Shi X-M, Zhang X-W, Chen F (2000) Heterotrophic production of biomass and lutein by Chlorella protothecoides on various nitrogen sources. Enzyme Microb Technol 27:312–318
Steinbrenner J, Sandmann G (2006) Transformation of the green alga Haematococcus pluvialis with a phytoene desaturase for accelerated astaxanthin biosynthesis. Appl Environ Microb 72:7477–7484
Sun N, Wang Y, Li Y-T, Huang J-C, Chen F (2008) Sugar-based growth, astaxanthin accumulation and carotenogenic transcription of heterotrophic Chlorella zofingiensis (Chlorophyta). Process Biochem 43:1288–1292
Xiong W, Gao C, Yan D, Wu C, Wu Q (2010) Double CO2 fixation in photosynthesis–fermentation model enhances algal lipid synthesis for biodiesel production. Bioresour Technol 101:2287–2293
Yan D, Lu Y, Chen Y-F, Wu Q (2011) Waste molasses alone displaces glucose-based medium for microalgal fermentation towards cost-saving biodiesel production. Bioresour Technol 102:6487–6493
Zhang BY, Geng YH, Li ZK, Hu HJ, Li YG (2009) Production of astaxanthin from Haematococcus in open pond by two-stage growth one-step process. Aquaculture 295:275–281
Zhong Y, Huang J, Liu J, Li Y, Jiang Y, Xu Z, Sandmann G, Chen F (2011) Functional characterization of various algal carotenoid ketolases reveals that ketolating zeaxanthin efficiently is essential for high production of astaxanthin in transgenic Arabidopsis. J Exp Bot 62:3659–3669
Acknowledgments
This study was partially supported by a grant from the 985 Project of Peking University and by the State Oceanic Administration of China.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Liu, J., Sun, Z., Zhong, Y. et al. Utilization of cane molasses towards cost-saving astaxanthin production by a Chlorella zofingiensis mutant. J Appl Phycol 25, 1447–1456 (2013). https://doi.org/10.1007/s10811-013-9974-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-013-9974-x