Abstract
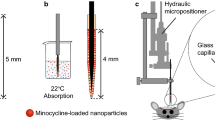
We describe the development of polymer implants that were designed to solidify once injected into rat brains. These implants comprised of glycofurol and copolymers of d,l-lactide (LA), ε-caprolactone and poly(ethylene glycol) (PLECs). Scanning electron microscopy (SEM) and gel permeation chromatography (GPC) showed that the extent of implant degradation was increased with LA content in copolymers. SEM analysis revealed the formation of porosity on implant surface as the degradation proceeds. PLEC with 19.3% mole of LA was chosen to inject in rat brains at the volume of 10, 25 and 40 μl. Body weights, hematological and histopathological data of rats treated with implants were evaluated on day 3, 6, 14, 30 and 45 after the injection. Polymer solution at the injection volume of 10 μl were tolerated relatively well compared to those of 25 and 40 μl as confirmed by higher body weight and healing action (fibrosis tissue) 30 days after treatment. The results from this study suggest a possible application as drug delivery systems that can bypass the blood brain barrier.





Similar content being viewed by others
References
Gabathuler R. Blood–brain barrier transport of drugs for the treatment of brain diseases. CNS Neurol Disord Drug Targ. 2009;8(3):195–204.
Huynh GH, Deen DF, Szoka FC. Barriers to carrier mediated drug and gene delivery to brain tumors. J Control Release. 2006;110(2):236–59.
Gutmann R, Leunig M, Feyh J, Goetz AE, Messmer K, Kastenbauer E, et al. Interstitial hypertension in head and neck tumors in patients: correlation with tumor size. Cancer Res. 1992;52(7):1993–5.
Jain RK. The next frontier of molecular medicine: delivery of therapeutics. Nat Med. 1998;4(6):655–7.
Leunig M, Goetz AE, Dellian M, Zetterer G, Gamarra F, Jain RK, et al. Interstitial fluid pressure in solid tumors following hyperthermia: possible correlation with therapeutic response. Cancer Res. 1992;52(2):487–90.
Leunig M, Yuan F, Menger MD, Boucher Y, Goetz AE, Messmer K, et al. Angiogenesis, microvascular architecture, microhemodynamics, and interstitial fluid pressure during early growth of human adenocarcinoma LS174T in SCID mice. Cancer Res. 1992;52(23):6553–60.
Abbott NJ. Inflammatory mediators and modulation of blood–brain barrier permeability. Cell Mol Neurobiol. 2000;20(2):131–47.
Bart J, Groen HJ, Hendrikse NH, van der Graaf WTA, Vaalburg W, de Vries EGE. The blood–brain barrier and oncology: new insights into function and modulation. Cancer Treat Rev. 2000;26(6):449–62.
Steiniger S, Kreuter J, Khalansky AS, Skidan IN, Bobruskin AI, Smirnova ZS, et al. Chemotherapy of glioblastoma in rats using doxorubicin-loaded nanoparticles. Int J Cancer. 2004;109(5):759–67.
Wu D, Pardridge WM. Blood–brain barrier transport of reduced folic acid. Pharm Res. 1999;16(3):415–9.
Laske DW, Youle RJ, Oldfield EH. Tumor regression with regional distribution of the targeted toxin TF-CRM107 in patients with malignant brain tumors. Nat Med. 1997;3(12):1362–8.
Guerin C, Olivi A, Weingart JD, Lawson HC, Brem H. Recent advances in brain tumor therapy: local intracerebral drug delivery by polymers. Investig New Drugs. 2004;22(1):27–37.
Brem H, Langer R. Polymer-based drug delivery to the brain. Sci Med. 1996;3:9.
Gallego JM, Barcia JA, Barcia-Marino C. Fatal outcome related to carmustine implants in glioblastoma multiforme. Acta Neurochir. 2007;149(3):261–5. discussion 5.
Boongird A, Nasongkla N, Hongeng S, Sukdawong N, Sa-Nguanruang W, Larbcharoensub N. Biocompatibility study of glycofurol in rat brains. Exp Biol Med. 2011;236(1):77–83.
Weinberg BD, Patel RB, Wu H, Blanco E, Barnett CC, Exner AA, et al. Model simulation and experimental validation of intratumoral chemotherapy using multiple polymer implants. Med Bio Eng Comput. 2008;46(10):1039–49.
Ivaturi VD, Riss JR, Kriel RL, Siegel RA, Cloyd JC. Bioavailability and tolerability of intranasal diazepam in healthy adult volunteers. Epilepsy Res. 2009;84(2–3):120–6.
Eliaz RE, Szoka FC. Robust and prolonged gene expression from injectable polymeric implants. Gene Ther. 2002;9(18):1230–7.
Bagger MA, Nielsen HW, Bechgaard E. Nasal bioavailability of peptide T in rabbits: absorption enhancement by sodium glycocholate and glycofurol. Eur J Pharm Sci. 2001;14(1):69–74.
Hu Y, Jiang X, Ding Y, Zhang L, Yang C, Zhang J, et al. Preparation and drug release behaviors of nimodipine-loaded poly(caprolactone)–poly(ethylene oxide)–polylactide amphiphilic copolymer nanoparticles. Biomaterials. 2003;24(13):2395–404.
Tsai CY, Chow NH, Ho TS, Lei HY. Intracerebral injection of myelin basic protein (MBP) induces inflammation in brain and causes paraplegia in MBP-sensitized B6 mice. Clin Exp Immunol. 1997;109(1):127–33.
Paxinos G, Watson C. The rat brain in stereotaxic coordinates. 5th ed. Burlington: Elsevier Academic Press; 2005.
Siparsky GL, Voorhees KJ, Miao F. Hydrolysis of polylactic acid (PLA) and polycaprolactone (PCL) in aqueous acetonitrile solutions: autocatalysis. J Environ Polym Degrad. 1998;6(1):31–41.
Gallego JM, Barcia JA, Barcia-Mariño C. Fatal outcome related to carmustine implants in glioblastoma multiforme. Acta Neurochir (Wien). 2007;149(3):261–5.
Patel RB, Solorio L, Wu H, Krupka TM, Exner AA. Effect of injection site on in situ implant formation and drug release in vivo. J Control Release. 2010;147:350–8.
Krupka TM, Weinberg BD, Ziats NP, Haaga JR, Exner AA. Injectable polymer depot combined with radiofrequency ablation for treatment of experimental carcinoma in rat. Investiga Radiol. 2006;41(12):890–7.
Bjugstad KB, Lampe K, Kern DS, Mahoney M. Biocompatibility of poly(ethylene glycol)-based hydrogels in the brain: an analysis of the glial response across space and time. J Biomed Mater Res Part A. 2010;95A(1):79–91.
Vertenten G, Vlaminck L, Gorski T, Schreurs E, Van Den Broeck W, Duchateau L, et al. Evaluation of an injectable, photopolymerizable three-dimensional scaffold based on d:l: -lactide and epsilon-caprolactone in a tibial goat model. J Mater Sci. 2008;19(7):2761–9.
Murakami H, Kobayashi M, Takeuchi H, Kawashima Y. Utilization of poly(dl-lactide-co-glycolide) nanoparticles for preparation of mini-depot tablets by direct compression. J Control Release. 2000;67(1):29–36.
Ekholm M, Hietanen J, Tulamo RM, Muhonen J, Lindqvist C, Kellomaki M, et al. Tissue reactions of subcutaneously implanted mixture of epsilon-caprolactone–lactide copolymer and tricalcium phosphate. An electron microscopic evaluation in sheep. J Mater Sci. 2003;14(10):913–8.
Acknowledgments
This research project is supported by Mahidol University, Thailand. We thank the National Institute of Health, Department of Medical Sciences, Ministry of Public Health for the help in animal care and handling.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nasongkla, N., Boongird, A., Hongeng, S. et al. Preparation and biocompatibility study of in situ forming polymer implants in rat brains. J Mater Sci: Mater Med 23, 497–505 (2012). https://doi.org/10.1007/s10856-011-4520-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-011-4520-3