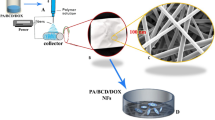
Abstract
In this study, zein nanofibers based siRNA delivery system has been attempted for the first time. Here, the amphiphilic property of zein and the size advantage of nanofibers have been brought together in developing an ideal delivery system for siRNA. The morphological analysis of the GAPDH-siRNA loaded zein nanofibers revealed the proper encapsulation of the siRNA in the polymeric matrix. The loading efficiency of this delivery system was found to be 58.57 ± 2.4 % (w/w). The agarose gel analysis revealed that the zein nanofibers preserved the integrity of siRNA for a longer period even at the room temperature. The in vitro release studies not only depicted the sustaining potential of the zein nanofibers but also ensured the release of sufficient quantity of siRNA required to induce the gene silencing effect. The amphiphilic property of zein supported the cell attachment and thereby facilitated the transfection of siRNA into the cells. qRT-PCR analysis confirmed the potential of the developed system in inducing the desired gene silencing effect. Thus, electrospun zein nanofibers have been successfully employed for the delivery of siRNA which has a great therapeutic potential.







Similar content being viewed by others
References
Shin H, Jo S, Mikos AG. Biomimetic materials for tissue engineering. Biomaterials. 2003;24:4353–64.
Richardson TP, Peters MC, Ennett AB, Mooney DJ. Polymeric system for dual growth factor delivery. Nat Biotechnol. 2001;19:1029–34.
Luu YK, Kim K, Hsiao BS, Chu B, Hadjiargyrou M. Development of a nanostructured DNA delivery scaffold via electrospinning of PLGA and PLA–PEGblock copolymers. J Control Release. 2003;89:341–53.
Laabs TL, Wang H, Katagiri Y, McCann T, Fawcett JW, Geller HM. Inhibiting glycosaminoglycan chain polymerization decreases the inhibitory activity of astrocyte-derived chondroitin sulfate proteoglycans. J Neurosci. 2007;27:14494–501.
Kamath RS, Fraser AG, Dong Y, Poulin G, Durbin R, Gotta M, Kanapin A, LeBot N, Moreno S, Sohrmann M, Welchman DP, Zipperien P, Ahringer J. Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature. 2003;421:231–7.
Berns K, Hijmans EM, Mullenders J, Brummelkamp TR, Velds A, Heimerikx M, Kerkhoven RM, Madiredjo M, Nijkamp W, Weigelt B, Agami R, Ge W, Cavet G, Linsley PS, Beijersbergen RL, Bernards R. A large-scale RNAi screen in human cells identifies new components of the p53 pathway. Nature. 2004;428:431–7.
Lum L, Yao S, Mozer B, Rovescalli A, Von Kessler D, Nirenberg M, Beachy PA. Identification of Hedgehog pathway components by RNAi in Drosophila cultured cells. Science. 2003;299:2039–45.
Butz K, Ristriani T, Hengstermann A, Denk C, Scheffner M, Hoppe-Seyler F. siRNA targeting of the viral E6 oncogene efficiently kills human papilloma virus positive cancer cells. Oncogene. 2003;22:5938–45.
Lieskovan SH, Heidel JD, Bartlett DW, Davis ME, Triche TJ. Sequence specificknockdown of EWS-FLI1 by targeted, non-viral delivery of small interfering RNA inhibits tumor growth in a murine model of metastatic Ewing’s sarcoma. Cancer Res. 2005;65:8984–92.
Miller VM, Xia H, Marrs GL, Gouvion CM, Lee G, Davidson BL, Paulson HL. Allele-specific silencing of dominant disease genes. Proc Natl Acad Sci. 2003;100:7195–200.
Singer O, Marr RA, Rockenstein E, Crews L, Coufal NG, Gage FH, Verma IM, Masliah E. Targeting BACE1 with siRNAs ameliorates Alzheimer disease neuropathologyin a transgenic model. Nat Neurosci. 2005;8:1343–9.
Shi JJ, Xiao Z, Votruba AR, Vilos C, Farokhzad OC. Differentially charged hollow core/shell lipid–polymer–lipid hybrid nanoparticles for small interfering RNA delivery. Angew Chem. 2011;123:7165–9.
Chen JL, Wang H, Gao JQ, Chen HL, Liang WQ. Liposomes modified with polycation used for gene delivery: preparation, characterization and transfection in vitro. Int J Pharm. 2007;343:255–61.
Colonna C, Conti B, Genta I, Alpar OH. Non-viral dried powders for respiratory gene delivery prepared by cationic and chitosan loaded liposomes. Int J Pharm. 2008;364:108–18.
Sioud M, Sorensen DR. Cationic liposome-mediated delivery of siRNAs in adultMice. Biochem Biophys Res Commun. 2003;312:1220–5.
Howard KA, Rahbek UL, Liu XD, Damgaard CK, Glud SZ, Andersen MO, Hovgaard MB, Schmitz A, Nyengaard JR, Besenbacher F, Kjems J. RNA interference in vitro and in vivo using a chitosan/siRNA nanoparticles system. Mol Ther. 2006;14:476–84.
Trabulo S, Resina S, Simoes S, Lebleu B, de Lima MCP. A non-covalent strategycombining cationic lipids and CPPs to enhance the delivery of splice correctingOligonucleotides. J Control Release. 2010;145:149–58.
Zhang SB, Zhao B, Jiang HM, Wang B, Ma BC. Cationic lipids and polymers mediated vectors for delivery of siRNA. J Control Release. 2007;123:1–10.
Crombez L, Herrada GA, Konate K, Nguyen QN, McMaster GK, Brasseur R, Heitz F, Divita G. A new potent secondary amphipathic cell-penetrating peptide for siRNA delivery into mammalian cells. Mol Ther. 2009;17:95–103.
Davidson TJ, Harel S, Arboleda VA, Prunell GF, Shelanski ML, Greene LA, Troy CM. Highly efficient small interfering RNA delivery to primary mammalian neurons induces microRNA-like effects before mRNA degradation. J Neurosci. 2004;24:10040–6.
Lundberg P, El-Andaloussi S, Sutlu T, Johansson H, Langel U. Delivery of short interfering RNA using endosome lytic cell-penetrating peptides. Faseb J. 2007;21:2664–71.
Veldhoen S, Laufer SD, Trampe A, Restle T. Cellular delivery of small interfering RNA by a non-covalently attached cell-penetrating peptide: quantitative analysis of uptake and biological effect. Nucl Acids Res. 2006;34:6561–73.
Cao HQ, Jiang X, Chai C, Chew SY. RNA interference by nanofiber-based siRNA delivery system. J Control Release. 2010;144:203–12.
Rujitanaroj P, Wang YC, Wang J, Chew SY. Nanofiber-mediated controlled release of siRNA complexes for long term gene-silencing applications. Biomaterials. 2011;32:5915–23.
Chew SY, Wen J, Yim EKF, Leong KW. Sustained Release of Proteins from Electrospun Biodegradable Fibers. Biomacromolecules. 2005;6:2017–24.
Gandhi M, Srikar R, Yarin AL, Megaridis CM, Gemeinhart RA. Mechanistic examination of protein release from polymer nanofibers. Mol Pharm. 2009;6:641–7.
Srikar R, Yarin AL, Megaridis CM, Bazilevsky AV, Kelley E. Desorption-limited mechanism of release from polymer nanofibers. Langmuir. 2008;24:965–74.
Kim HS, Yoo HS. Matrix metalloproteinase-inspired suicidal treatments of diabetic ulcers with siRNA-decorated nanofibrous meshes. Gene Ther. 2013;20:378–85.
Nitta SK, Numata K. Biopolymer-based nanoparticles for drug/gene delivery and tissue engineering. Int J Mol Sci. 2013;14:1629–54.
Elzoghby AO, Wael MS, Nazik AE. Protein-based nanocarriers as promising drug and gene delivery systems. J Control Release. 2012;161:38–49.
Regier MC, Taylor JD, Borcyk T, Yang Y, Pannier AK. Fabrication and characterization of DNA-loaded zein nanospheres. J Nanobiotechnol. 2012;10:1–13.
Karthikeyan K, Guhathakarta S, Rajaram R, Korrapati PS. Electrospun zein/eudragit nanofibers based dual drug delivery system for the simultaneous delivery of aceclofenac and pantoprazole. Int J Pharm. 2012;438:117–22.
Normand J, Karasek MA. c. In Vitro Cell Dev Biol Anim. 1995;31:447–55.
Mosmann T. Rapid calorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65:55–63.
Zeng J, Xu X, Chen X, Liang Q, Bian X, yang L, Jing X. Biodegradable electrospun fibers for drug delivery. J Control Release. 2003;92:227–31.
Jiang Q, Reddy N, Yang Y. Cytocompatible cross-linking of electrospun zein fibers for the development of water-stable tissue engineering scaffolds. Acta Biomater. 2010;6:4042–51.
Dhandayuthapani B, Poulose AC, Nagaoka Y, Hasumura T, Yoshida Y, Maekawa T, Kumar DS. Biomimetic smart nanocomposite: in vitro biological evaluation of zein electrospun fluorescent nanofiber encapsulated CdS quantum dots. Biofabrication. 2012;4:025008.
Forbes K, Desforges M, Garside R, Aplin JD, Westwood M. Methods for siRNA-mediated reduction of mRNA and protein expression in human placental explants, isolated primary cells and cell lines. Placenta. 2009;30:124–9.
Acknowledgments
The authors K. Karthikeyan and Venkat Raghavan Krishnaswamy are extremely grateful to the Council of Scientific and Industrial Research, New Delhi, for the award of senior research fellowship. We are grateful to Dr. A. B. Mandal, Director, CSIR-Central Leather Research Institute for providing the encouragement and facilities to carry out this work. This work was supported by Council of Scientific and Industrial Research (CSIR), New Delhi under the Project “Advance Drug Delivery (ADD—CSC 0302).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10856_2015_5439_MOESM1_ESM.tif
Fig. S1. Phase contrast microscopic images depicting the attachment and spreading of cells on zein nanofibers at lower (a) and higher magnification (b). (TIFF 363 kb)
Rights and permissions
About this article
Cite this article
Karthikeyan, K., Krishnaswamy, V.R., Lakra, R. et al. Fabrication of electrospun zein nanofibers for the sustained delivery of siRNA. J Mater Sci: Mater Med 26, 101 (2015). https://doi.org/10.1007/s10856-015-5439-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10856-015-5439-x