Abstract
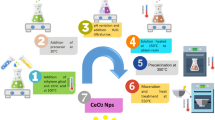
Uniform CeO2 nanoparticles were synthesized via a facile sonochemical reaction between ceric ammonium nitrate and ammonia. Nanoparticles were synthesized via a surfactant free reaction at room temperature in solvent of water. Products were characterized using X-ray diffraction, scanning electron microscopy, photoluminescence (PL) spectroscopy, and energy dispersive X-ray analysis. The effect of different parameters such as precursor, power of pulsation, surfactant and reaction time on the morphology of the products was investigated. It was found that the as-obtained CeO2 nanoparticles exhibit a strong PL peak at 381 nm at room temperature that can be ascribed to the high level transition in the CeO2 semiconductor. The photocatalytic behavior of CeO2 nanoparticles was evaluated using the degradation of a methyl orange aqueous solution under ultraviolet light irradiation. The results show that CeO2 nanoparticles are promising materials with excellent performance in photocatalytic applications.










Similar content being viewed by others
References
A. Saadat-Monfared, M. Mohseni, and M. Hashemi Tabatabaei (2012). Colloid. Surf. A 408, 64.
C. Hu, Z. Zhang, H. Liu, P. Gao, and Z. Lin Wang (2006). Nanotechnology 17, 5983.
C. Sun, H. Li, H. Zhang, Z. Wang, and L. Chen (2005). Nanotechnology 16, 1454.
P. Dutta, S. Pal, and M. S. Seehra (2006). Chem. Mater. 18, 5144.
F. Zhang, P. Wang, J. Koberstein, S. Khalid, and Siu-Wai. Chan (2004). Surf. Sci. 563, 74.
T. Masui, H. Hirai, N. Imanaka, G. Adachi, T. Sakata, and H. Mori (2002). J. Mater. Sci. Lett 21, 489.
D. Zhang, H. Fu, L. Shi, C. Pan, Q. Li, Y. Chu, and W. Yu (2007). Inorg. Chem. 46, 2446.
A. I. Y. Tok, F. Y. C. Boey, Z. Dong, and X. L. Sun (2007). J. Mater. Process. Tech. 190, 217.
Z. Wu, J. Zhang, R. E. Benfield, Y. Ding, D. Grandjean, Z. Zhang, and X. Ju (2002). J. Phys. Chem. B 106, 4569.
C. Kitiwiang and S. Phanichphant (2009). J. Microsc. Soc. Thailand 23, (1), 83.
M. Salavati-Niasari, G. Hosseinzadeh, and F. Davar (2011). J. Alloys. Compd. 509, 134.
M. Esmaeili-Zare, M. Salavati-Niasari, and A. Sobhani (2012). Ultrason. Sonochem. 19, 1079.
G. Kianpour, M. Salavati-Niasari, and H. Emadi (2012). Ultrason. Sonochem. 20, (1), 418.
M. Salavati-Niasari, D. Ghanbari, and M. R. Loghman-Estarki (2012). Polyhedron 35, 149.
T. J. Mason, J. P. Lorimer, and D. M. Bates (1992). Ultrasonics 30, 40.
Y. J. Chen, L. W. Huang, and T. S. Shih (2003). Mater. Trans. 44, 327.
S. Popinet and S. Zaleski (2002). J. Fluid Mech. 464, 137.
H.R. Momenian, S. Gholamrezaei, M. Salavati-Niasari, B. Pedram, F. Mozaffar, D.,and Ghanbari (2013) J. Clust. Sci., DOI 10.1007/s10876-013-0595-y.
S. R. Shirsath, D. V. Pinjari, P. R. Gogate, S. H. Sonawane, and A. B. Pandit (2013). Ultrason. Sonochem. 20, 277.
D. V. Pinjari and A. B. Pandit (2011). Ultrason. Sonochem. 18, 1118.
K. Prasad, D. V. Pinjari, A. B. Pandit, and S. T. Mhaske (2010). Ultrason. Sonochem. 17, 697.
Acknowledgments
Authors are grateful to the council of Iran National Science Foundation and University of Kashan for supporting this work by Grant No (159271/80).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jamshidi, P., Salavati-Niasari, M., Ghanbari, D. et al. Synthesis, Characterization, Photoluminescence and Photocatalytic Properties of CeO2 Nanoparticles by the Sonochemical Method. J Clust Sci 24, 1151–1162 (2013). https://doi.org/10.1007/s10876-013-0605-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-013-0605-0