Abstract
Transit compartments (TC) models are used to describe pharmacodynamic responses that involve drug action on cells undergoing differentiation and maturation. Such pharmacodynamic systems can also be described by lifespan based indirect response (LIDR) models. The purpose of this report is to investigate conditions under which the transit compartments models can be considered a special case of LIDR models. An integral representation of a solution to TC model has been used to determine the lifespan distribution for cell population described by this model. The distribution served as a basis for definition of new LIDRE (lifespan based indirect response with an effect on the lifespan distribution) models. Time courses of responses described by both types of models were simulated for a monoexponential pharmacokinetic function. The limit response was calculated as the number of transit compartments approached infinity. The difference between the limit response and TC responses were evaluated by computer simulations using MATLAB 7.7. TC models are a special case of LIDR models with the lifespan distribution described by the gamma function. If drug affects only the production of cells, then the cell lifespan distribution is time invariant. In this case an increase in the number of compartments results in a basic LIDR model with a point lifespan distribution. When the drug inhibits or stimulates cell aging, the cell lifespan distribution becomes time dependent revealing a new mechanism for drug effect on the gamma probability density function. The TC model with a large number of transit compartments converges to an LIDRE model. The limit LIDR models are approximated by the TC models when the number of compartments is at least 5. A moderate improvement in the approximation is observed if this number exceeds 20. The lifespan distribution for a cell population described by a TC model is described by the gamma probability density function. A drug affects this distribution only if it stimulates or inhibits the rate of cell maturation. If the number of transit compartments increases, then the TC model converges to a new type of LIDR model.






Similar content being viewed by others
References
Friberg LE, Karlsson MO (2003) Mechanistic models for myelosuppression. Invest New Drugs 21:183–194
Harker LA, Roskos LK, Marzec UM, Carter RA, Cherry JK, Sundell B, Cheung EL, Terry D, Sheridan W (2000) Effects of megakaryocyte growth and development factor on platelet production, platelet life span, and platelet function in healthy human volunteers. Blood 95:2514–2522
Friberg LE, Henningsson A, Maas H, Nguyen L, Karlsson MO (2002) Model of chemotherapy-induced myelosuppression with parameter consistency across drugs. J Clin Oncol 20:4713–4721
Roskos LK, Lum P, Lockbaum P, Schwab G, Yang B-B (2006) Pharmacokinetic/pharmacodynamic modeling of pegfilgrastim in healthy subjects. J Clin Pharmacol 46:747–757
Perez-Ruixo JJ, Krzyzanski W, Hing J (2008) Pharmacodynamic analysis of recombinant human erythropoietin effect on reticulocyte production rate and age distribution in healthy subjects. Clin Pharmacokinet 47:399–415
Krzyzanski W, Ramakrishnan R, Jusko WJ (1999) Basic pharmacodynamic models for agents that alter production of natural cells. J Pharmacokinet Biopharm 21:457–478
Uehlinger DE, Gotch FA, Sheiner LB (1992) A pharmacodynamic model of erythropoietin therapy for uremic anemia. Clin Pharmacol Ther 51:76–89
Chapel SH, Veng-Pedersen P, Schmidt RL, Widness JA (2000) A pharmacodynamic analysis of erythropoietin-stimulated reticulocyte response in phlebotomized sheep. J Pharmacol Exp Ther 295:346–351
Krzyzanski W, Woo S, Jusko WJ (2006) Pharmacodynamic models for agents that alter production of natural cells with various distributions of lifespans. J Pharmacokinet Pharmacodyn 33:125–165
Freise KJ, Widness JA, Schmidt RL, Veng-Pedersen P (2008) Modeling time variant distributions of cellular lifespans: increase in circulating reticulocyte lifespans following double phlebotomies in sheep. J Pharmacokinet Pharmacodyn 35:285–324
Freise KJ, Widness JA, Schmidt RL, Veng-Pedersen P (2008) Pharmacodynamic modeling of the effect of changes in the environment on cellular lifespan and response. J Pharmacokinet Pharmacodyn 35:527–552
Krzyzanski W, Perez-Ruixo JJ, Vermeulen A (2008) Basic pharmacodynamic models for agents that alter the lifespan distribution of natural cells. J Pharmacokinet Pharmacodyn 35:349–377
Krzyzanski W, Jusko WJ (2002) Multiple-pool cell lifespan model of hematologic effects of anticancer agents. J Pharmacokinet Pharmacodyn 29:311–337
Mager DE, Jusko WJ (2001) Pharmacodynamic modeling of time-dependent transduction systems. Clin Pharmacol Ther 70:210–216
Sun YN, Jusko WJ (1998) Transit compartments versus gamma distribution function to model signal transduction processes in pharmacodynamics. J Pharm Sci 87:732–737
Murphy EA, Francis ME (1969) The estimation of blood platelet survival. I. General principles of the study of cell survival. Thrombos Diath Haemorrh (Stuttg) 22:281–295
Murphy EA, Francis ME (1971) The estimation of blood platelet survival. II. The multiple hit model. Thrombos Diath Haemorrh (Stuttg) 25:53–80
Dayneka NL, Garg V, Jusko WJ (1993) Comparison of four basic models of indirect pharmacodynamic responses. J Pharmacokinet Biopharm 21:457–478
Rudin W (1973) Functional analysis. McGraw-Hill, Boston, p 157
Simeoni M, Magni P, Cammia C, De Nicolao G, Croci V, Pesenti E, Germani M, Poggesi I, Rocchetti M (2004) Predictive pharmacokinetic-pharmacodynamic modeling of tumor growth kinetics in xenograft models after administration of anticancer agents. Cancer Res 64:1094–1101
Zelen M, Severo NC (1972) Probability functions. In: Abramowitz M, Stegun I (eds) Handbook of mathematical functions with formulas, graphs, and mathematical tables. Dover Publications, New York
Davis PJ (1972) Gamma function and related functions. In: Abramowitz M, Stegun I (eds) Handbook of mathematical functions with formulas, graphs, and mathematical tables. Dover Publications, New York
Wiczling P, Krzyzanski W (2008) Flow cytometric assessment of homeostatic aging of reticulocytes in rats. Exp Hematol 36:112–127
Hunt RA (1994) Calculus. HarperCollins College Publishers, New York
Rudin W (1987) Real and complex analysis. McGraw-Hill, Boston, p 26
Walte K, Gabrilove J, Bronchud MH, Platzer E, Morstyn G (1996) Filgrastim (r-metHuG-CSF): the first 10 years. Blood 88:1907–1929
Wiczling P, Ait-Oudhia S, Krzyzanski W (2009) Flow cytometric analysis of reticulocyte maturation after erythropoietin administration in rats. Cytometry A 75A:584–592
Polenakovic M, Sikole A (1996) Is erythropoietin a survival factor for red blood cells? J Am Soc Nephrol 7:1178–1182
Krzyzanski W, Wiczling P, Lowe P, Pigeolet E, Fink M, Berghout A, Balser S (2010) Population modeling of filgrastim PK-PD in healthy adults following intravenous and subcutaneous administrations. J Clin Pharmacol 50:101S–112S
Samtani MN, Perez-Ruixo JJ, Brown K, Cerneus D, Molloy C (2009) Pharmacokinetic and pharmacodynamic modeling of pegylated thrombopoietin mimetic peptide (PEG-TPOm) after single intravenous dose in healthy subjects. J Clin Pharmacol 49:336–350
Wang Y-M, Krzyzanski W, Doshi S, Xiao JJ, Perez Ruixo JJ, Chow AT (2010) Pharmacodynamics-mediated drug disposition (PDMDD) and precursor pool lifespan model for single dose of romiplostim in healthy subjects. AAPS J 12:729–740
Acknowledgments
The author is grateful to Dr. Juan Jose Perez-Ruixo for reviewing this manuscript and providing insightful comments. This study was supported by the National Institute of General Medical Sciences, National Institutes of Health Grant GM 57980.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendices
Appendix 1
Derivation of Eq. 6
To simplify calculations we introduce the following variables
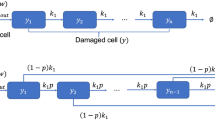
Then the transit compartments model Eqs. 1–2 become
Equation 45 can be solved using the integrating factor technique [24]:
We will show by mathematical induction that for \( {\text{i}} = 1, \ldots ,{\text{n}} \)
Since the case i = 1 reduces to Eq. 47, it remains to show that Eq. 48 is true for i + 1 assuming it holds for i. Using the same integrating factor as for Eq. 47 one can solve Eq. 46 with i + 1 to obtain the following representation of Yi+1:
One can substitute the right hand side of Eq. 48 for Yi(τ) in Eq. 49:
Equation 50 can be further reduced to
Changing the order of the first two integrals in Eq. 51 results in
The only part of the integrant that depends on τ is equal to
Substituting Eq. 53 in the integrant of Eq. 52 gives
Equation 11 is equal to Eq. 48 with i + 1, which completes the recursive proof that Eq. 48 holds true for \( {\text{i}} = 1, \ldots ,{\text{n}} \). Finally, to derive Eq. 6 one needs to replace Yi(t) and k(τ) in Eq. 48 with the original Pi and kin(t) using Eq. 44.
Appendix 2
Derivation of Eq. 16
Equation 6 with i = n implies that the solution for the n-th compartment is
Changing the variables τ → t − τ in the first integral results in
The difference in the integrand can be integrated separately as follows
The change of the variable
reduces the integral in the second term of Eq. 57 to the following form
The properties of the gamma distribution imply that [21]
Consequently,
where the original integration variable τ was restored in the integral in Eq. 60. The baseline conditions Eq. 3 imply that for τ > t
Hence
One can insert In back in Eq. 57 resulting in
After multiplying both sides of Eq. 64 by nE(t)/TR one arrives at Eq. 16.
Appendix 3
Derivation of Eq. 19
For given times t and τ let us define
Since the derivative
Equation 17 can be simplified to the following form
Applying the change of variables \( s \to \hat{{\text{E}}}_{{\text{t}}} (\tau ) \) in the following integral, one can further reduce it to the integral of the gamma function γn(s) defined in Eq. 18:
The latter is a consequence of γn(s) as a probability density function. This proves Eq. 19.
Appendix 4
Derivation of Eq. 29
To prove Eq. 29 we use the definition of the Dirac delta function δτ0(τ) = δ(τ − τ0) stating that for any test function ϕ(τ) (an indefinitely differentiable function with a compact support contained in the interval (0,∞)) the following relationship holds [19]:
Let ϕ(τ) be a test function, then according to Eq. 24
The substitution \( {\text{s}} \to \Upphi_{{\text{t}}} (\tau ) \) transforms the last integral in Eq. 70 to the following
where \( \Upphi_{{\text{t}}}^{ - 1} ({\text{s}}) \) is the inverse of \( \Upphi_{{\text{t}}} (\tau ) \). The integral in Eq. 71 is evaluated according to Eq. 69. Consequently, for any test function ϕ(τ)
This proves Eq. 29.
Appendix 5
Derivation of Eqs. 30, 32, and 33
A general form for the basic LIDRE model for drug affecting the point distribution according to Eqs. 27 and 28 will be described as follows
where after applying Eqs. 24, 65 and 66:
Therefore Eq. 73 can be further simplified to
To calculate the integral in Eq. 75 let us introduce a transformation Ψt(τ)
One can verify that for any time t
The change of variables s → Ψt(τ) transforms the integral in Eq. 74 to
where the derivative of the inverse is
and the definition of the delta function δ(τ − TR) has been utilized. To abbreviate the right hand side of Eq. 78 the following function of time t is introduced:
Then Eq. 73 simplifies to Eq. 30
Notice that Eq. 81 was derived without an assumption of any specific form for kin(t) like one specified by Eqs. 9 and 10. Equations 76 and 80 imply
At t = 0, Eq. 82 becomes
Since TR > 0 and E(z) > 0, then TE(0) < 0. For z < 0, E(z) = 1 (see Eq. 3), which implies
This proves Eq. 33. To derive a differential equation for TE(t), calculate the derivative with respect to t of both sides of Eq. 82. The derivative of the right-hand side is zero, and the derivative of the integral in the left-hand side results in the following equation:
That results in Eq. 32.
Appendix 6
Proof of Eqs. 37–39
Integration of both sides of Eq. 26 leads to
Similarly, integration of Eq. 30 results in
Upon subtraction of both sides of Eqs. 87 and 86:
Following the definition of Ψt(τ), Eq. 88 implies that
Analogously, Eq. 80 implies
Hence
and Eq. 37 follows. To show that Eq. 37 holds true one can transform Eq. 91 further utilizing the fact that γn(τ) is a p.d.f.
Since Eqs. 7–10 imply that ϕs(τ) is a bound function of s and τ, i.e. there is a constant M > 0 such that for any s and τ
Hence
The dominated convergence theorem [25] implies that for any t the right hand side of Eq. 92 approaches 0 as n → ∞, for any s
Equation 95 is true due to the approximate identity property of the gamma distribution p.d.f. γn(τ) cited in Eq. 36, which we will prove for an arbitrary continuous and bound function f(τ). Let ε > 0 be a small number and M > 0 be such |f(τ)| ≤ M for all τ. Then the continuity of f(τ) implies that there is a small ε > 0 such that for any TR-ε < τ < TR + ε
The integral in Eq. 95 can be partitioned into a sum of three integrals:
The first and third integral in Eq. 97 can be bound as follows:
and
The bound for the second integral in Eq. 97 is
Since
the first and third integral in Eq. 97 can be bound by ε for large enough n, and consequently
Rights and permissions
About this article
Cite this article
Krzyzanski, W. Interpretation of transit compartments pharmacodynamic models as lifespan based indirect response models. J Pharmacokinet Pharmacodyn 38, 179–204 (2011). https://doi.org/10.1007/s10928-010-9183-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-010-9183-z