Summary
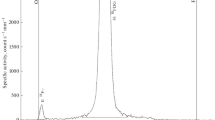
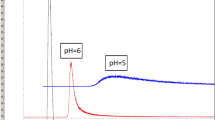
To control virtually the toxic compounds and to improve quality control of the solution of 2-deoxy-2-[18F]fluoro-d-glucose (2-[18F]FDG), the products of its autoradiolysis were analyzed by high-performance liquid chromatography with electrospray mass spectrometric and radiometric detectors (HPLC/MS/RAD), thin layer chromatography on TLC silica plate and HPTLC on amino modified silica plate. Except Kryptofix™ 2.2.2, glucose and fluoride anion, no by-products and impurities were observed by LC/MS analysis of fresh 2-[18F]FDG samples. The analysis performed in the time interval of 6 to 48 hours after the end of 2-[18F]FDG synthesis indicated that the activity of the autoradiolysis products separated by HPLC did not exceed 1.3%. As the main autoradiolysis products of 3.3 . 10-5 to 4.4 . 10-5M 2-[18F]FDG solution of original specific activity 0.5-1.5 GBq . cm-3 were established: arabinose - 2.8 μM (G= 0.07/100 eV), gluconic and glucuronic acids 1.8-0.5 μM (G =0.01-0.05/100 eV), arabinose and araburonic acids occurred under 0.5 μM concentration at residual glucose contents about 0.14 mM. Radiation chemical yields of active products were calculated from molar activity of 2-[18F]FDG and the percentage of their activity: 0.5% radiochemical yield of 2-[18F]fluoroglucuronic acid corresponds to the G = 0.004/100 eV and 0.3% yield of 2-[18F]fluorogluconic acid issues G = 0.003/100 eV.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Búriová, E., Macášek, F., Melichar, F. et al. Autoradiolysis of the 2-deoxy-2-[18F]fluoro-D-glucose radiopharmaceutical. J Radioanal Nucl Chem 264, 595–602 (2005). https://doi.org/10.1007/s10967-005-0759-9
Issue Date:
DOI: https://doi.org/10.1007/s10967-005-0759-9