Summary
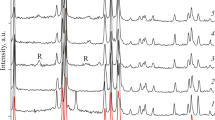
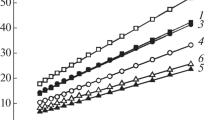
A mixed metal oxalate, manganese(II)bis(oxalato)nickelate(II)tetrahydrate, has been synthesized and characterized by elemental analysis, IR spectral and X-ray powder diffraction (XRD) studies. Thermal decomposition studies (TG, DTG and DTA) in air showed that the compound decomposed mainly to Mn2O3, MnO2 and NiO at ca.1000°C, via. the formation of several intermediates. DSC study in nitrogen upto 500°C showed the endothermic decomposition. The tentative mechanism for the thermal decomposition in air is proposed.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Deb, N. Thermal decomposition of manganese(II)bis(oxalato)nickelate(II)tetrahydrate. J Therm Anal Calorim 81, 61–65 (2005). https://doi.org/10.1007/s10973-005-0746-y
Issue Date:
DOI: https://doi.org/10.1007/s10973-005-0746-y