Abstract
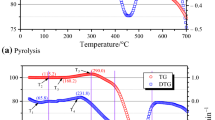
Thermal analysis of seven Jurassic coal samples from North Shaanxi in West China and three permo-carboniferous coal samples from East China was studied to identify ignition temperatures in the process of the oxidation and spontaneous combustion. The experiments were carried out under non-isothermal heating conditions up to 700 °C at the heating rates of 5, 10, 15, and 20 °C min−1 in an air atmosphere. Through the FTIR spectrometer experiments, the absorbance peaks of functional groups of coal samples were analyzed at the ignition temperatures, pre-ignition of the 10 °C, post-ignition of the 10 °C at the heating rate of 10 °C min−1. By the differential spectrum method, the changes of functional groups were discussed with the aim to determine characteristics and reactivity of the ignition temperature around. The results showed that ignition temperatures of experimental coal samples increased with the rising heating rates, and ignition temperatures of Jurassic coals were lower than that of the permo-carboniferous coal samples at the same heating rate. Apparent activation energy of experimental Jurassic coals at the ignition temperatures was calculated by Ozawa method based on the non-isothermal and differential heating rates, ranging from 80 to 105 kJ mol−1, which were lower than that of the eastern permo-carboniferous samples. On the basis of Pearson correlation coefficient method which can signify the degree of correlations ranging from −1 to 1, the correlation analyses were conducted between activation energy and functional groups variation within 10 °C before and after ignition temperature. It was concluded that the key functional groups of Jurassic coals in the oxidation and ignition reaction were methyl and alkyl ether within 10 °C before ignition temperature, and carboxyl and carbonyl within 10 °C after ignition temperature.











Similar content being viewed by others
References
Hong Zhang, Hong-tang Li, Cun-wei Xiong. Jurassic coal-bearing and accumulation regularity in Northwest China. Beijing: Geological Publishing House; 1998.
Hai-zhou Chang, Fan-gui Zeng, Wen-ying Li. Micro-FTIR study on structure of macerals from Jurassic Coals in Northwestern China. Spectrosc Spectr Anal. 2008;28:1535–8.
Wall TF, Gupta RP, Gururajan VS. The ignition of coal particles. Fuel. 1991;70:1011–6.
Zhang DK, Wall TF, Harris DJ, Smith IW, Stanmore BR. Experimental studies of ignition behaviour and combustion reactivity of pulverized fuel particles. Fuel. 1992;71:1239–46.
Chen John C, Taniguchi M, Ito K. Observation of laser ignition and combustion of pulverized coals. Fuel. 1996;74:323–30.
Meriste T, Yörük CR, Trikkel A. TG–FTIR analysis of oxidation kinetics of some solid fuels under oxy-fuel conditions. J Therm Anal Calorim. 2013;114:483–9.
Fan D, Zhu Z, Na Y. Thermogravimetric analysis of gasification reactivity of coal chars with steam and CO2 at moderate temperatures. J Therm Anal Calorim. 2013;113:599–607.
Mothé Cheila G, de Miranda Iara C. Study of kinetic parameters of thermal decomposition of bagasse and sugarcane straw using Friedman and Ozawa–Flynn–Wall isoconversional methods. J Therm Anal Calorim. 2013;113:497–505.
Babiński P, Łabojko G, Kotyczka-Morańska M. Kinetics of coal and char oxycombustion studied by TG–FTIR. J Therm Anal Calorim. 2008;28:388–91.
Hu RZ, Gao SL, Zhao XQ. Thermal analysis kinetics. Beijing: Science Press; 2008.
Kök MV. Recent developments in the application of thermal analysis techniques in fossil fuels. J Therm Anal Calorim. 2008;91:763–73.
Kök MV. Temperature-controlled combustion and kinetics of different rank coal samples. J Therm Anal Calorim. 2005;79:175–80.
Koga N. Ozawa’s kinetic method for analyzing thermoanalytical curves. J Therm Anal Calorim. 2013;113:1527–41.
Ozawa T. Applicability of Friedman plot. J Therm Anal Calorim. 1986;31:547–51.
Cui X, Zhang X, Yang. M. Study on the structure and reactivity of COREX coal. J Therm Anal Calorim. 2013;113:693–701.
Ge LM, Li JW. Evolution of functional groups in low-temperature oxidized Shenfu coal. J Xi’an Univ Sci Technol. 2003;22:187–90.
Kizgut S, Baran Y, Cuhadaroglu D. Reactivity and characterisation of various rank Turkish bituminous coal chars. J Therm Anal Calorim. 2003;71:857–65.
Kaljuvee T, Keelman M, Trikkel A, Petkova V. TG-FTIR/MS analysis of thermal and kinetic characteristics of some coal samples. J Therm Anal Calorim. 2013;113:1063–71.
Qin-lin He, De-ming Wang. Comprehensive study on the rule of spontaneous combustion coal in oxidation process by TG- DTA- FTIR technology. J China Coal Soc. 2005;30:53–7.
Jie F, Wen-ying L, Ke-chang X. Research on coal structure using FT-IR. J China Univ Min Technol. 2002;31:362–6.
Acknowledgements
This paper was supported by the Key Program of National Natural Science Foundation of China (No. 51134019), the National Basic Research Program of China (973 Program No. 2011CB411902), National Natural Science Foundation (No. 51244001), Shaanxi Innovative Research Team for Key Science and Technology (No. 2012KCT-09).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deng, J., Wang, K., Zhang, Y. et al. Study on the kinetics and reactivity at the ignition temperature of Jurassic coal in North Shaanxi. J Therm Anal Calorim 118, 417–423 (2014). https://doi.org/10.1007/s10973-014-3974-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-014-3974-1