Abstract
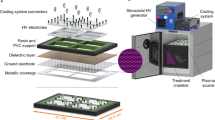
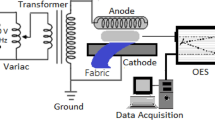
In this work, the hydrophilic improvement of a woven PET surface was accomplished by a plasma technique. The woven PET surface was plasma-treated by dielectric barrier discharge (DBD) under various operating conditions (electrode gap distance, plasma treatment time, input voltage, and input frequency) and various gaseous environments (air, O2, N2, and Ar) in order to improve its hydrophilicity. It was experimentally found that a decrease in electrode gap distance and an increase in input voltage increased the electric field strength, leading to higher hydrophilicity of the PET surface characterized by wickability and contact angle measurements. In comparisons among the studied environmental gases, air gave the highest hydrophilicity, being comparable to O2, while Ar and N2 gave lower hydrophilicity of the woven PET surface. The optimum conditions for a maximum hydrophilicity of the PET surface were an electrode gap distance of 4 mm, a plasma treatment time of 10 s, an output voltage of 15 kV, and a frequency of 350 Hz under air environment. After the plasma treatment under the obtained optimum conditions, the woven PET was loaded with Ag particles using a AgNO3 aqueous solution in order to obtain the antimicrobial property. The plasma-treated woven PET loaded with Ag particles exhibited good antimicrobial activity against both E. coli (gram-negative bacteria) and S. aureus (gram-positive bacteria).












Similar content being viewed by others
References
Wypych J (1988) Polymer modified textile materials. Wiley, New York
Chan CM (1994) Polymer surface modification and characterization. Hanser, New York
Ko TM, Cooper SL (1994) Frontiers of polymers and advanced materials. Plenum Press, New York
Strobel M, Lyons CS, Mittal KL (1994) Plasma surface modification of polymers: relevance to adhesion. VSP, Utrecht
Brooks D, Giles GA (2002) PET packaging technology. Sheffield Academic, Sheffield
Scheirs J, Long TE (2003) Modern polyesters: chemistry and technology of polyesters and copolyesters. Wiley, New York
Dai JH, Bruening ML (2002) Nano Lett 2:497
Lee HJ, Yeo SY, Jeong SH (2003) J Mater Sci 28:2199
Ignatova M, Labaye D, Lenoir S, Strivay D, Jerome R, Jerome C (2003) Langmuir 19:2759
Huh MW, Kang IK, Lee DH, Kim WS, Lee DH (2001) J Appl Polym Sci 81:2769
Yang MR, Chen KS, Tsai JCh, Tseng CC, Lin SF (2002) Mater Sci Eng C 20:167
Fu J, Ji J, Yuan W, Shen J (2005) Biomaterials 26:6684
Nakagawa Y, Hayashi H, Tawaratani T, Kourai H, Horie T, Shibasaki I (1984) Appl Environ Microbiol 47:513
Cen L, Neoh KG, Kang ET (2003) Langmuir 19:10295
Charton C, Fahland M (2001) Surf Coat Tech 142–144:175
Shi Z, Neoh KG, Kang ET (2004) Langmuir 20:6847
Wang J, Li J, Ren L, Zhao A, Li P, Leng Y, Sun H, Huang N (2007) Surf Coat Tech 201:6893
Li JX, Wang J, Shen LR, Xu ZJ, Li P, Wan GJ, Huang N (2007) Surf Coat Tech 201:8155
De Geyter N, Morent R, Leys C (2006) Surf Coat Tech 201:2460
Chavadej S, Kiattubolpaiboon W, Rangsunvigit P, Sreethawong T (2007) J Mol Catal A: Chem 263:128
Li Y, Leung P, Yao L, Song QW, Newton E (2006) J Hosp Infect 62:58
Morinaga K, Suzuki M (1962) Bull Chem Soc Jpn 35:204
Kogelschatz U (2003) Plasma Chem Plasma Process 23:1
Fang Z, Qiu Y, Luo Y (2003) Appl Phys 36:2980
Le QT, Pireaux JJ, Verbist JJ (1994) Surf Interface Anal 22:224
Inagaki N, Tasaka S, Narushima K, Kobayashi H (2002) J Appl Polym Sci 85:2845
Park JW, Kang M (2007) Int J Hydrogen Energy 32:4840
Weaver JF, Hoflund GB (1994) J Phys Chem 98:8519
Biniak S, Pakula M, Swiatkowski A (1999) J Appl Electrochem 29:421
Feng QL, Wu J, Chen GQ, Cui FZ, Kim TN, Kim JO (2000) J Biomed Mater Res 52:662
Cho KH, Park JE, Osaka T, Park SG (2005) Electrochim Acta 51:956
Acknowledgments
The authors would like to thank Thai Negoro Co., Ltd, Thailand; the Sustainable Petroleum and Petrochemicals Research Unit, Center for Petroleum, Petrochemicals, and Advanced Materials, Chulalongkorn University, Thailand; and the Petrochemical and Environmental Catalysis Research Unit under the Ratchadapisek Somphot Endowment Fund, Chulalongkorn University, Thailand. The authors would also like to thank Dr. Hideaki Nagahama, Department of Chemistry and Materials Engineering, Faculty of Chemistry, Materials, and Bioengineering, Kansai University, Japan for his assistance in XPS analysis.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Onsuratoom, S., Rujiravanit, R., Sreethawong, T. et al. Silver Loading on DBD Plasma-Modified Woven PET Surface for Antimicrobial Property Improvement. Plasma Chem Plasma Process 30, 191–206 (2010). https://doi.org/10.1007/s11090-009-9199-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-009-9199-6