Abstract
Aim
Rhizobacteria can influence plant growth and metal accumulation. The aim of this study was to evaluate the effect of rhizobacterial inoculants on the Ni phytoextraction efficiency of the Ni-hyperaccumulator Alyssum pintodasilvae.
Method
In a preliminary screening 15 metal-tolerant bacterial strains were tested for their plant growth promoting (PGP) capacity or effect on Ni bioaccumulation. Strains were selected for their Ni tolerance, plant growth promoting traits and Ni solubilizing capacity. In a re-inoculation experiment five of the previously screened bacterial isolates were used to inoculate A. pintodasilvae in two contrasting Ni-rich soils (a serpentine (SP) soil and a sewage sludge-affected agricultural (LF) soil).
Results
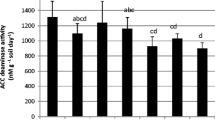
Plant growth was greater in serpentine soil (where it grows naturally) than in the LF soil, probably due to Cd phytotoxicity. Rhizobacterial inoculants influenced plant growth and Ni uptake and accumulation, but the effect of the strains was dependent upon soil type. The increase in plant biomass and/or Ni accumulation significantly promoted shoot Ni removal.
Conclusion
One strain (Arthrobacter nicotinovorans SA40) was able to promote plant growth and phytoextraction of Ni in both soil types and could be a useful candidate for future field-based trials.





Similar content being viewed by others
References
Abou-Shanab RA, Angle JS, Delorme TA, Chaney RL, van Berkum P, Moawad H, Ghanem K, Ghozlan HA (2003) Rhizobacterial effects on nickel extraction from soil and uptake by Alyssum murale. New Phytol 158:219–224. doi:10.1046/j.1469-8137.2003.00721.x
Abou-Shanab R, Angle J, Chaney R (2006) Bacterial inoculants affecting nickel uptake by Alyssum murale from low, moderate and high Ni soils. Soil Biol Biochem 38:2882–2889. doi:10.1016/j.soilbio.2006.04.045
Asensi A, Rodríguez N, Díez-Garretas B, Amils R, de la Fuente V (2004) Nickel hyperaccumulation of some subspecies of Alyssum serpyllifolium (Brassicaceae) from ultramafic soils of Iberian Peninsula. In: Boyd RS, Baker AJM, Proctor J (eds) Ultramafic Soils: Their Soils, Vegetation and Fauna. Science Reviews, St. Albans, UK, pp 263–266
Assunção AL, Bleeker P, Bookum W, Vooijs R, Schat H (2008) Intraspecific variation of metal preference patterns for hyperaccumulation in Thlaspi caerulescens: evidence from binary metal exposures. Plant Soil 303:289–299. doi:10.1007/s11104-007-9508-x
Baker AJM, McGrath SP, Sidoli CMD, Reeves RD (1994) The possibility of in situ heavy metal decontamination of polluted soils using crops of metal-accumulating plants. Resour Conserv Recycl 11:41–49
Bani A, Echevarria G, Sulçe S, Morel J, Mullai A (2007) In-situ phytoextraction of Ni by a native population of Alyssum murale on an ultramafic site (Albania). Plant Soil 293:79–89. doi:10.1007/s11104-007-9245-1
Barzanti R, Colzi I, Arnetoli M, Gallo A, Pignattelli S, Gabbrielli R, Gonnelli C (2011) Cadmium phytoextraction potential of different Alyssum species. J Hazard Mater 196:66–72. doi:10.1016/j.jhazmat.2011.08.075
Becerra-Castro C, Prieto-Fernández A, Álvarez-Lopez V, Monterroso C, Cabello-Conejo M, Acea M, Kidd P (2011) Nickel solubilizing capacity and characterization of rhizobacteria isolated from hyperaccumulating and non-hyperaccumulating subspecies of Alyssum serpyllifolium. Int J Phytoremediat 13:229–244
Becerra-Castro C, Monterroso C, Prieto-Fernández A, Rodríguez-Lamas L, Loureiro-Viñas M, Acea MJ, Kidd PS (2012) Pseudometallophytes colonising Pb/Zn mine tailings: a description of the plant–microorganism–rhizosphere soil system and isolation of metal-tolerant bacteria. J Hazard Mater 217–218:350–359. doi:10.1016/j.jhazmat.2012.03.039
Becerra-Castro C, Kidd P, Kuffner M, Prieto-Fernandez A, Hann S, Monterroso C, Sessitsch A, Wenzel W, Puschenreiter M (2013) Bacterially induced weathering of ultramafic rock and its implications for phytoextraction. Appl Environ Microbiol 79:5094–5103. doi:10.1128/aem.00402-13
Boisson J, Mench M, Sappin-Didier V, Solda P, Vangronsveld J (1998) Short-term in situ immobilization of Cd and Ni by beringite and steel shots application to long-term sludged plots. Agronomie 18:347–359
Brooks RR, Shaw S, Marfil AA (1981) Some observations on the ecology, metal uptake and nickel tolerance of Alyssum serpyllifolium subspecies from the Iberian peninsula. Plant Ecol 45:183–188. doi:10.1007/bf00054673
Cabello-Conejo M, Centofanti T, Kidd P, Prieto-Fernández Á, Chaney R (2013) Evaluation of plant growth regulators to increase nickel phytoextraction by Alyssum species. Int J Phytoremediat 15:365–375
Chaney RL, Malik M, Li YM, Brown SL, Brewer EP, Angle JS, Baker AJM (1997) Phytoremediation of soil metals. Curr Opin Biotechnol 8:279–284. doi:10.1016/s0958-1669(97)80004-3
Chaney RL, Angle JS, Broadhurst CL, Peters CA, Tappero RV, Sparks DL (2007) Improved understanding of hyperaccumulation yields commercial phytoextraction and phytomining technologies. J Environ Qual 36:1429–1443. doi:10.2134/jeq2006.0514
Chaney RL, Chen KY, Li YM, Angle JS, Baker AJM (2008) Effects of calcium on nickel tolerance and accumulation in Alyssum species and cabbage grown in nutrient solution. Plant Soil 311:131–140. doi:10.1007/s11104-008-9664-7
Dell’Amico E, Cavalca L, Andreoni V (2008) Improvement of Brassica napus growth under cadmium stress by cadmium-resistant rhizobacteria. Soil Biol Biochem 40:74–84. doi:10.1016/j.soilbio.2007.06.024
Everhart JL, McNear D Jr, Peltier E, van der Lelie D, Chaney RL, Sparks DL (2006) Assessing nickel bioavailability in smelter-contaminated soils. Sci Total Environ 367:732–744. doi:10.1016/j.scitotenv.2005.12.029
Ewers U (1991) Standards, guidelines and legislative regulations concerning metals and their compounds. In: Merian E (ed) Metals and their Compound in the Environment. VCH, Weinheim, pp 687–711
Gabbrielli P, Pandolfini T, Vergnano O, Palandri MR (1990) Comparison of two serpentine species with different nickel tolerance strategies. Plant Soil 122:271–277. doi:10.1007/BF02851985
Gadd GM (2010) Metals, minerals and microbes: geomicrobiology and bioremediation. Microbiology 156:609–643. doi:10.1099/mic.0.037143-0
Glick BR (2003) Phytoremediation: synergistic use of plants and bacteria to clean up the environment. Biotechnol Adv 21:383–393. doi:10.1016/S0734-9750(03)00055-7
Glick BR, Penrose DM, Li J (1998) A model for the lowering of plant ethylene concentrations by plant growth-promoting bacteria. J Theor Biol 190:63–68. doi:10.1006/jtbi.1997.0532
He S, He Z, Yang Z, Baligar VC (2012) Mechanisms of nickel uptake and hyperaccumulation by plants and implications for soil remediation. In: Advances in Agronomy, Vol 117. Elsevier, USA
Jiang C-y, Sheng X-f, Qian M, Wang Q-y (2008) Isolation and characterization of a heavy metal-resistant Burkholderia sp. from heavy metal-contaminated paddy field soil and its potential in promoting plant growth and heavy metal accumulation in metal-polluted soil. Chemosphere 72:157–164. doi:10.1016/j.chemosphere.2008.02.006
Kidd P, Barceló J, Bernal MP, Navari-Izzo F, Poschenrieder C, Shilev S, Clemente R, Monterroso C (2009) Trace element behaviour at the root-soil interface: implications in phytoremediation. Environ Exp Bot 67:243–259
Kukier U, Peters CA, Chaney RL, Angle JS, Roseberg RJ (2004) The effect of pH on metal accumulation in two species. J Environ Qual 33:2090–2102. doi:10.2134/jeq2004.2090
Lebeau T, Braud A, Jézéquel K (2008) Performance of bioaugmentation-assisted phytoextraction applied to metal contaminated soils: a review. Environ Pollut 153:497–522. doi:10.1016/j.envpol.2007.09.015
Li Y-M, Chaney R, Brewer E, Roseberg R, Angle JS, Baker A, Reeves R, Nelkin J (2003) Development of a technology for commercial phytoextraction of nickel: economic and technical considerations. Plant Soil 249:107–115. doi:10.1023/a:1022527330401
Ma Y, Rajkumar M, Freitas H (2009) Improvement of plant growth and nickel uptake by nickel resistant-plant-growth promoting bacteria. J Hazard Mater 166:1154–1161. doi:10.1016/j.jhazmat.2008.12.018
Ma Y, Prasad MNV, Rajkumar M, Freitas H (2011) Plant growth promoting rhizobacteria and endophytes accelerate phytoremediation of metalliferous soils. Biotechnol Adv 29:248–258. doi:10.1016/j.biotechadv.2010.12.001
Mastretta C, Taghavi S, van der Lelie D, Mengoni A, Galardi F, Gonnelli C, Barac T, Boulet J, Weyens N, Vangronsveld J (2009) Endophytic bacteria from seeds of Nicotiana tabacum can reduce cadmium phytotoxicity. Int J Phytoremediat 11:251–267
Mench M, Renella G, Gelsomino A, Landi L, Nannipieri P (2006) Biochemical parameters and bacterial species richness in soils contaminated by sludge-borne metals and remediated with inorganic soil amendments. Environ Pollut 144:24–31. doi:10.1016/j.envpol.2006.01.014
Mench M, Schwitzguebel JP, Schroeder P, Bert V, Gawronski S, Gupta S (2009) Assessment of successful experiments and limitations of phytotechnologies: contaminant uptake, detoxification and sequestration, and consequences for food safety. Environ Sci Pollut Res 16:876–900. doi:10.1007/s11356-009-0252-z
Menezes de Sequeira E (1969) Toxicity and movement of heavy metals in serpentinic soils (North-Eastern Portugal). Agron Lusit 30:115–154
Mergeay M, Nies D, Schlegel HG, Gerits J, Charles P, Van Gijsegem F (1985) Alcaligenes eutrophus CH34 is a facultative chemolithotroph with plasmid-bound resistance to heavy metals. J Bacteriol 162:328–334
Moreno-Jiménez E, Esteban E, Carpena-Ruiz RO, Lobo MC, Penalosa JM (2012) Phytostabilisation with Mediterranean shrubs and liming improved soil quality in a pot experiment with a pyrite mine soil. J Hazard Mater 201:52–59
Olsen SR, Cole CV, Watanabe FS (1954) Estimation of available phosphorus in soils by extraction with sodium bicarbonate. USDA, Washington
Rajkumar M, Freitas H (2008a) Effects of inoculation of plant-growth promoting bacteria on Ni uptake by Indian mustard. Bioresour Technol 99:3491–3498. doi:10.1016/j.biortech.2007.07.046
Rajkumar M, Freitas H (2008b) Influence of metal resistant-plant growth-promoting bacteria on the growth of Ricinus communis in soil contaminated with heavy metals. Chemosphere 71:834–842. doi:10.1016/j.chemosphere.2007.11.038
Sessitsch A, Kuffner M, Kidd P, Vangronsveld J, Wenzel WW, Fallmann K, Puschenreiter M (2013) The role of plant-associated bacteria in the mobilization and phytoextraction of trace elements in contaminated soils. Soil Biol Biochem 60:182–194. doi:10.1016/j.soilbio.2013.01.012
Shilev S, Fernández A, Benlloch M, Sancho E (2006) Sunflower growth and tolerance to arsenic is increased by the rhizospheric bacteria Pseudomonas fluorescens. In: Morel JL, Echevarria G, Goncharova N (eds) Phytoremediation of Metal-Contaminated Soils. Springer, Netherlands, pp 315–326. doi:10.1007/1-4020-4688-X_12
Tappero R, Peltier E, Gräfe M, Heidel K, Ginder-Vogel M, Livi KJT, Rivers ML, Marcus MA, Chaney RL, Sparks DL (2007) Hyperaccumulator Alyssum murale relies on a different metal storage mechanism for cobalt than for nickel. New Phytol 175:641–654. doi:10.1111/j.1469-8137.2007.02134.x
Van der Ent A, Baker AJ, Reeves RD, Pollard AJ, Schat H (2013) Hyperaccumulators of metal and metalloid trace elements: facts and fiction. Plant Soil 362:319–334
Vangronsveld J and Clijsters H (1994) Toxic effects of metals. In: Farago ME (ed) Plants and the Chemical Elements. Biochemistry, Uptake,Tolerance and Toxicity. VCH, Weinheim, pp 149–177
Vangronsveld J, Herzig R, Weyens N, Boulet J, Adriaensen K, Ruttens A, Thewys T, Vassilev A, Meers E, Nehnevajova E, van der Lelie D, Mench M (2009) Phytoremediation of contaminated soils and groundwater: lessons from the field. Environ Sci Pollut Res Int 16:765–794. doi:10.1007/s11356-009-0213-6
Weissenhorn I, Mench M, Leyval C (1995) Bioavailability of heavy metals and arbuscular mycorrhiza in a sewage-sludge-amended sandy soil. Soil Biol Biochem 27:287–296. doi:10.1016/0038-0717(94)00179-5
Weyens N, van der Lelie D, Taghavi S, Newman L, Vangronsveld J (2009a) Exploiting plant-microbe partnerships to improve biomass production and remediation. Trends Biotechnol 27:591–598. doi:10.1016/j.tibtech.2009.07.006
Weyens N, van der Lelie D, Taghavi S, Vangronsveld J (2009b) Phytoremediation: plant-endophyte partnerships take the challenge. Curr Opin Biotechnol 20:248–254. doi:10.1016/j.copbio.2009.02.012
Zaidi S, Usmani S, Singh BR, Musarrat J (2006) Significance of Bacillus subtilis strain SJ-101 as a bioinoculant for concurrent plant growth promotion and nickel accumulation in Brassica juncea. Chemosphere 64:991–997. doi:10.1016/j.chemosphere.2005.12.057
Acknowledgments
This work was supported by the Spanish Ministerio de Economía e Competitividad (CTM2012-39904-C02-01) and FEDER, the Fundación Mapfre (Ayudas a la Investigación 2012), and by the 7th Framework Program of the European Commission (FP7-KBBE-266124, GREENLAND).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Juan Barcelo.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. 1S
Concentrations of macro- and micro-nutrients in the shoots (mean ±SE) of Alyssum pintodasilvae grown in sand/perlite mixtures and inoculated with 15 rhizobacterial strains. Values of non-inoculated controls are indicated by a continuous line (±SE (broken lines)). Asterisks indicate significant differences from the control (p <0.05) (PNG 386 kb)
Rights and permissions
About this article
Cite this article
Cabello-Conejo, M.I., Becerra-Castro, C., Prieto-Fernández, A. et al. Rhizobacterial inoculants can improve nickel phytoextraction by the hyperaccumulator Alyssum pintodasilvae . Plant Soil 379, 35–50 (2014). https://doi.org/10.1007/s11104-014-2043-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-014-2043-7