Abstract
Aim
Plant-associated bacteria can improve phytoextraction by increasing plant growth and/or metal uptake. This study aimed to characterise the culturable rhizobacterial community associated with two Ni-hyperaccumulators and to obtain a collection of isolates for application in Ni phytomining.
Methods
Non-vegetated and rhizosphere soil samples were collected from the Ni-hyperaccumulator Alyssum serpyllifolium ssp. lusitanicum (three populations) and Alyssum serpyllifolium ssp. malacitanum (one population), as well as from non-hyperaccumulating plants (Dactylis glomerata, Santolina semidentata and Alyssum serpyllifolium ssp. serpyllifolium). Rhizobacteria were isolated and characterised genotypically (BOX-PCR, 16S rDNA sequencing) and phenotypically (Ni tolerance, plant growth promoting (PGP) traits, biosurfactant production).
Results
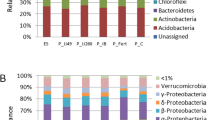
Hyperaccumulating Alyssum subspecies hosted higher densities of bacteria compared to either non-hyperaccumulators or non-vegetated soil. In some cases hyperaccumulators showed selective enrichment of Ni-tolerant bacteria. Most bacterial strains belonged to the Actinobacteria phylum and presented Ni resistance. Phosphorus-solubilisers were mostly associated with the hyperaccumulators, siderophore-producers with D. glomerata, and IAA-producers with both these species.
Conclusion
Taxonomic diversity and phenotypic characteristics were soil-, plant species- and plant population-specific. Moreover, differences were observed between the two Ni-hyperaccumulating subspecies and amongst plant populations. Several strains presented PGP characteristics which could be useful when selecting microorganisms for bioaugmentation trials.



Similar content being viewed by others
References
Abou-Shanab RI, Delorme TA, Angle JS, Chaney RL, Ghanem K, Moawad H, Ghozlan HA (2003) Phenotypic characterization of microbes in the rhizosphere of Alyssum murale. Int J Phytorem 5:367–379
Abou-Shanab RAI, van Berkum P, Angle JS, Delorme TA, Chaney RL, Ghozlan HA, Ghanem K, Moawad H (2010) Characterization of Ni-resistant bacteria in the rhizosphere of the hyperaccumulator Alyssum murale by 16S rRNA gene sequence analysis. World J Microbiol Biotechnol 26:101–108. doi:10.1007/s11274-009-0148-6
Acea M, Carballas T (1986) Estudio de la población microbiana de diversos tipos de suelos de zona húmeda (N.O. de España). Ann Edafol Agrobiol 45:381–398
Amir H, Pineau R (1998) Effects of metals on the germination and growth of fungal isolates from New Caledonian ultramafic soils. Soil Biol Biochem 30:2043–2054. doi:10.1016/S0038-0717(98)00079-0
Bååth E (1989) Effects of heavy metals in soil on microbial processes and populations (a review). Water Air Soil Pollut 47:335–379. doi:10.1007/BF00279331
Baker A, Brooks R (1989) Terrestrial higher plants which hyperaccumulate metallic elements. A review of their distribution, ecology and phytochemistry. Biorecovery 1:81–126
Becerra-Castro C (2006) Relacións planta – solo – microorganismo nunha especie hiperacumuladora de níquel endémica de solos serpentiníticos. B. Sc. Dissertation, Universidade de Santiago de Compostela (Spain)
Becerra-Castro C, Monterroso C, García-Lestón M, Prieto-Fernández A, Acea MJ, Kidd PS (2009) Rhizosphere microbial densities and trace metal tolerance of the nickel hyperaccumulator Alyssum serpyllifolium subsp. lusitanicum. Int J Phytorem 11:525–541. doi:10.1080/15226510902717549
Becerra-Castro C, Prieto-Fernández Á, Álvarez-López V, Monterroso C, Cabello-Conejo MI, Acea MJ, Kidd PS (2011) Nickel solubilizing capacity and characterization of rhizobacteria isolated from hyperaccumulating and non-hyperaccumulating subspecies of Alyssum serpyllifolium. Int J Phytorem 13(Suppl 1):229–244. doi:10.1080/15226514.2011.568545
Becerra-Castro C, Monterroso C, Prieto-Fernández A, Rodríguez-Lamas L, Loureiro-Viñas M, Acea MJ, Kidd PS (2012) Pseudometallophytes colonising Pb/Zn mine tailings: a description of the plant-microorganism-rhizosphere soil system and isolation of metal-tolerant bacteria. J Hazard Mater 217–218:350–359. doi:10.1016/j.jhazmat.2012.03.039
Becerra-Castro C, Kidd P, Kuffner M, Prieto-Fernández A, Hann S, Monterroso C, Sessitsch A, Wenzel W, Puschenreiter M (2013) Bacterially induced weathering of ultramafic rock and its implications for phytoextraction. Appl Environ Microbiol 79:5094–5103
Braud A, Jézéquel K, Vieille E, Tritter A, Lebeau T (2006) Changes in extractability of Cr and Pb in a polycontaminated soil after bioaugmentation with microbial producers of biosurfactants, organic acids and siderophores. Water Air Soil Pollut Focus 6:261–279
Brooks RR (1987) Serpentine and its vegetation: a multidisciplinary approach. Croom Helm, Dioscorides Press
Cabello-Conejo MI (2015) Nickel hyperaccumulating plants: strategies to improve phytoextraction and a characterisation of Alyssum endemic to the Iberian Peninsula. PhD Thesis, Universidade de Santiago de Compostela (Spain)
Cabello-Conejo MI, Becerra-Castro C, Prieto-Fernández A, Monterroso C, Saavedra-Ferro A, Mench M, Kidd PS (2014) Rhizobacterial inoculants can improve nickel phytoextraction by the hyperaccumulator Alyssum pintodasilvae. Plant Soil 379:35–50
Carballeira A, Devesa C, Retuerto R, Santillán E, Ucieda F (1983) Bioclimatología de Galicia. Fundación Pedro Barrié de la Maza, Conde de Fenosa, A Coruña
Castillo-Martín A (2000) Parque nacional de Sierra Nevada clima e hidrología. In: Canseco (ed) Parque nacional de Sierra Nevada
Chaney RL, Angle JS, Broadhurst CL, Peters CA, Tappero RV, Sparks DL (2007) Improved understanding of hyperaccumulation yields commercial phytoextraction and phytomining technologies. J Environ Qual 36:1429–1443. doi:10.2134/jeq2006.0514
Chen C-Y, Baker SC, Darton RC (2007) The application of a high throughput analysis method for the screening of potential biosurfactants from natural sources. J Microbiol Methods 70:503–510. doi:10.1016/j.mimet.2007.06.006
Cole JR, Wang Q, Cardenas E, Fish J, Chai B, Farris RJ, Kulam-Syed-Mohideen AS, McGarrell DM, Marsh T, Garrity GM, Tiedje JM (2009) The Ribosomal Database Project: improved alignments and new tools for rRNA analysis. Nucleic Acids Res 37:D141–D145. doi:10.1093/nar/gkn879
Cox CD (1994) Deferration of laboratory media and assays for ferric and ferrous ions. Methods Enzymol 235:315–329
DeGrood SH, Claassen VP, Scow KM (2005) Microbial community composition on native and drastically disturbed serpentine soils. Soil Biol Biochem 37:1427–1435. doi:10.1016/j.soilbio.2004.12.013
Delorme TA, Gagliardi JV, Angle JS, Chaney RL (2001) Influence of the zinc hyperaccumulator Thlaspi caerulescens J. & C. Presl. and the nonmetal accumulator Trifolium pratense L. on soil microbial populations. Can J Microbiol 47:773–776. doi:10.1139/cjm-47-8-773
Fitz WJ, Wenzel WW, Zhang H, Nurmi J, Štipek K, Fischerova Z, Schweiger P, Köllensperger G, Ma LQ, Stingeder G (2003) Rhizosphere characteristics of the arsenic hyperaccumulator Pteris vittata L. and monitoring of phytoremoval efficiency. Environ Sci Technol 37:5008–5014. doi:10.1021/es0300214
Glick BR (2014) Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol Res 169:30–39
Gómez-Zotano J, Román-Requena F, Hidalgo-Triana N, Pérez-Latorre A (2014) Biodiversity and conservation values of the serpentine ecosystems in Spain: Sierra Bermeja (Málaga province). Bol Asoc Geógr Esp 65:451–456
Grayston SJ, Wang S, Campbell CD, Edwards AC (1998) Selective influence of plant species on microbial diversity in the rhizosphere. Soil Biol Biochem 30:369–378. doi:10.1016/S0038-0717(97)00124-7
Gremion F, Chatzinotas A, Harms H (2003) Comparative 16S rDNA and 16S rRNA sequence analysis indicates that Actinobacteria might be a dominant part of the metabolically active bacteria in heavy metal-contaminated bulk and rhizosphere soil. Environ Microbiol 5:896–907. doi:10.1046/j.1462-2920.2003.00484.x
Idris R, Trifonova R, Puschenreiter M, Wenzel WW, Sessitsch A (2004) Bacterial communities associated with flowering plants of the Ni hyperaccumulator Thlaspi goesingense. Appl Environ Microbiol 70:2667–2677. doi:10.1128/aem.70.5.2667-2677.2004
Jeong S, Moon HS, Shin D, Nam K (2013) Survival of introduced phosphate-solubilizing bacteria (PSB) and their impact on microbial community structure during the phytoextraction of Cd-contaminated soil. J Hazard Mater 263(Part 2):441–449. doi:10.1016/j.jhazmat.2013.09.062
Kidd P, Barceló J, Bernal MP, Navari-Izzo F, Poschenrieder C, Shilev S, Clemente R, Monterroso C (2009) Trace element behaviour at the root–soil interface: implications in phytoremediation. Environ Exp Bot 67:243–259. doi:10.1016/j.envexpbot.2009.06.013
Kozdrój J (1995) Microbial responses to single or successive soil contamination with Cd or Cu. Soil Biol Biochem 27:1459–1465. doi:10.1016/0038-0717(95)00070-U
Kunito T, Saeki K, Nagaoka K, Oyaizu H, Matsumoto S (2001) Characterization of copper-resistant bacterial community in rhizosphere of highly copper-contaminated soil. Eur J Soil Biol 37:95–102. doi:10.1016/S1164-5563(01)01070-6
Lane D (1991) 16s/23s rRNA sequencing. In: Stackerbrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Wiley, Chichester, pp 115–175
Lebeau T, Braud A, Jezequel K (2008) Performance of bioaugmentation-assisted phytoextraction applied to metal contaminated soils: a review. Environ Pollut 153:497–522. doi:10.1016/j.envpol.2007.09.015
Lodewyckx C, Mergeay M, Vangronsveld J, Clijsters H, Van der Lelie D (2002) Isolation, characterization, and identification of bacteria associated with the zinc hyperaccumulator Thlaspi caerulescens subsp. calaminaria. Int J Phytorem 4:101–115. doi:10.1080/15226510208500076
Ma Y, Rajkumar M, Vicente JAF, Freitas H (2010) Inoculation of Ni-resistant plant growth promoting bacterium Psychrobacter sp. strain SRS8 for the improvement of nickel phytoextraction by energy crops. Int J Phytorem 13:126–139. doi:10.1080/15226511003671403
Menezes de Sequeira E, Pinto da Silva AR (1992) Ecology of serpentinized areas of north-east Portugal. In: Roberts BA, Proctor J (eds) The Ecology of Areas with Serpentinized Rocks, vol 17. Geobotany. Springer Netherlands, pp 169–197. doi:10.1007/978-94-011-3722-5_7
Mengoni A, Barzanti R, Gonnelli C, Gabbrielli R, Bazzicalupo M (2001) Characterization of nickel-resistant bacteria isolated from serpentine soil. Environ Microbiol 3:691–698. doi:10.1046/j.1462-2920.2001.00243.x
Mengoni A, Schat H, Vangronsveld J (2010) Plants as extreme environments? Ni-resistant bacteria and Ni-hyperaccumulators of serpentine flora. Plant Soil 331:5–16
Mergeay M, Nies D, Schlegel H, Gerits J, Charles P, Van Gijsegem F (1985) Alcaligenes eutrophus CH34 is a facultative chemolithotroph with plasmid-bound resistance to heavy metals. J Bacteriol 162:328–334
Nautiyal CS (1999) An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol Lett 170:265–270. doi:10.1111/j.1574-6968.1999.tb13383.x
Oline DK (2006) Phylogenetic comparisons of bacterial communities from serpentine and nonserpentine soils. Appl Environ Microbiol 72:6965–6971
Pal A, Dutta S, Mukherjee PK, Paul AK (2005) Occurrence of heavy metal-resistance in microflora from serpentine soil of Andaman. J Basic Microbiol 45:207–218. doi:10.1002/jobm.200410499
Pal A, Wauters G, Paul AK (2007) Nickel tolerance and accumulation by bacteria from rhizosphere of nickel hyperaccumulators in serpentine soil ecosystem of Andaman, India. Plant Soil 293:37–48. doi:10.1007/s11104-007-9195-7
Proctor J (1999) Toxins, nutrient shortages and droughts: the serpentine challenge. Trends Ecol Evol 14:334–335. doi:10.1016/S0169-5347(99)01698-5
Puschenreiter M, Schnepf A, Millán IM, Fitz WJ, Horak O, Klepp J, Schrefl T, Lombi E, Wenzel WW (2005) Changes of Ni biogeochemistry in the rhizosphere of the hyperaccumulator Thlaspi goesingense. Plant Soil 271:205–218. doi:10.1007/s11104-004-2387-5
Rivas-Martinez S, Rivas-Saenz S (1996–2009) Worldwide bioclimatic classification system. Phytosociological Research Center, Spain. http://www.globalbioclimatics.org
Rodrı́guez H, Fraga R (1999) Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol Adv 17:319–339. doi:10.1016/S0734-9750(99)00014-2
Schlegel HG, Cosson JP, Baker AJM (1991) Nickel-hyperaccumulating plants provide a niche for nickel-resistant bacteria. Bot Acta 104:18–25. doi:10.1111/j.1438-8677.1991.tb00189.x
Schwartz C, Morel JL, Saumier S, Whiting SN, Baker AJM (1999) Root development of the zinc-hyperaccumulator plant Thlaspi caerulescens as affected by metal origin, content and localization in soil. Plant Soil 208:103–115
Schwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160:47–56. doi:10.1016/0003-2697(87)90612-9
Sessitsch A, Kuffner M, Kidd P, Vangronsveld J, Wenzel WW, Fallmann K, Puschenreiter M (2013) The role of plant-associated bacteria in the mobilization and phytoextraction of trace elements in contaminated soils. Soil Biol Biochem 60:182–194. doi:10.1016/j.soilbio.2013.01.012
Sheng X-F, Xia J-J, Jiang C-Y, He L-Y, Qian M (2008) Characterization of heavy metal-resistant endophytic bacteria from rape (Brassica napus) roots and their potential in promoting the growth and lead accumulation of rape. Environ Pollut 156:1164–1170. doi:10.1016/j.envpol.2008.04.007
Turgay OC, Gormez A, Bilen S (2012) Isolation and characterization of metal resistant-tolerant rhizosphere bacteria from the serpentine soils in Turkey. Environ Monit Assess 184:515–526. doi:10.1007/s10661-011-1984-z
Venkatesh NM, Vedaraman N (2012) Remediation of soil contaminated with copper using rhamnolipids produced from Pseudomonas aeruginosa MTCC 2297 using waste frying rice bran oil. Ann Microbiol 62:85–91. doi:10.1007/s13213-011-0230-9
Versalovic J, Schneider M, De Bruijn FJ, Lupski JR (1994) Genomic fingerprinting of bacteria using repetitive sequence-based polymerase chain reaction. Methods Mol Cell Biol 5:25–40
Whiting SN, Leake JR, McGrath SP, Baker AJM (2001) Assessment of Zn mobilization in the rhizosphere of Thlaspi caerulescens by bioassay with non-accumulator plants and soil extraction. Plant Soil 237:147–156
Zaidi S, Usmani S, Singh BR, Musarrat J (2006) Significance of Bacillus subtilis strain SJ-101 as a bioinoculant for concurrent plant growth promotion and nickel accumulation in Brassica juncea. Chemosphere 64:991–997. doi:10.1016/j.chemosphere.2005.12.057
Acknowledgments
This research was supported by the Spanish Ministerio de Economía y Competitividad (CTM2012-39904-C02-01) and FEDER, and by the 7th Framework Program of the European Commission (FP7-KBBE-266124, GREENLAND).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Antony Van der Ent.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online resource 1
(DOCX 29 kb)
Online resource 2
(DOCX 24 kb)
Online resource 3
(DOCX 27 kb)
Online resource 4
(DOCX 28 kb)
Online resource 5
(DOCX 22 kb)
Online resource 6
(DOCX 19 kb)
Online resource 7
(DOCX 48 kb)
Online resource 8
(DOCX 17 kb)
Rights and permissions
About this article
Cite this article
Álvarez-López, V., Prieto-Fernández, Á., Becerra-Castro, C. et al. Rhizobacterial communities associated with the flora of three serpentine outcrops of the Iberian Peninsula. Plant Soil 403, 233–252 (2016). https://doi.org/10.1007/s11104-015-2632-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-015-2632-0