Abstract
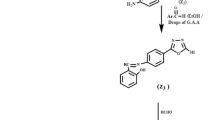
Green corrosion inhibitor, maleic acid mono-(1-alkyl-1H-[1, 2, 3]-triazol-4-ylmethyl) ester (Mal-Tr-R) with three different lengths of carbon chain were synthesized by nucleophilic substitution reaction, click coupling reaction, and esterification reaction. The final products were characterized by elemental analysis, FT-IR and 1H NMR spectra, indicating that the products were successfully synthesized. The corrosion inhibition efficiencies of Mal-Tr-R in hydrochloric acid solution were evaluated by weight loss and electrochemical methods. The adsorption equilibrium constant (K 0) and standard thermodynamic parameters of adsorption were calculated. The surface microstructure of the mild steel was studied through scanning electron microscope. The results indicated that the Mal-Tr-R can act as effective corrosion inhibitors in hydrochloric acid solution for mild steel. It was also determined that the inhibition efficiency of Mal-Tr-R increases with the increase of the carbon chain at low temperature, but the opposite trend appears at high temperature.











Similar content being viewed by others
References
P.M. Krishnegowda, V.T. Venkatesha, P.K.M. Krishnegowda, S.B. Shivayogiraju, Ind. Eng. Chem. Res. 52, 722 (2013)
M.L. Zheludkevich, D.G. Shchukin, K.A. Yasakau, H. Möhwald, M.G.S. Ferreira, Chem. Mater. 19, 402 (2007)
B.M. Mistry, N.S. Patel, M.J. Patel, S. Jauhari, Res. Chem. Intermed. 37, 660 (2011)
P.B. Raja, A.K. Qureshi, A.A. Rahim, K. Awang, M.R. Mukhtar, H. Osman, JMEPEG 22, 1072 (2013)
C. Kamal, M.G. Sethuraman, Ind. Eng. Chem. Res. 51, 10399 (2012)
K.D. Demadis, C. Mantzaridis, P. Lykoudis, Ind. Eng. Chem. Res. 45, 7795 (2006)
E.E. Oguzie, S.G. Wang, Y. Li, F.H. Wang, J. Phys. Chem. C 113, 8420 (2009)
M.-K. Hsieh, D.A. Dzombak, R.D. Vidic, Ind. Eng. Chem. Res. 49, 7313 (2010)
X. Sheng, Y.-P. Ting, S.O. Pehkonen, Ind. Eng. Chem. Res. 46, 7117 (2007)
D.-Q. Ng, T.J. Strathmann, Y.-P. Lin, Environ. Sci. Technol. 46, 11063 (2012)
A. Zarrouk, B. Hammouti, A. Dafali, F. Bentiss, Ind. Eng. Chem. Res. 52, 2560 (2012)
C.J. Zou, Q.W. Tang, P.W. Zhao, E.D. Guan, X. Wud, H. Ye, J. Pet. Sci. Eng. 103, 29 (2013)
C.J. Zou, Q.W. Tang, G.H. Lan, Q. Tian, T.Y. Wang, J. Inclu. Phenom. Macrocycl. Chem. (2013). doi:10.1007/s10847-012-0173-1
N. Saoudi, A. Bellaouchou, A. Guenbour, A. Ben Bachir, E.M. Essassi, M.E.L. Achouri, Bull. Mater. Sci. (2010). doi:10.1007/s12034-010-0048-2
N. Nava, N.V. Likhanova, O. Olivares-Xometl, E.A. Flores, I.V. Lijanova, Hyperfine Interact (2011). doi:10.1007/s10751-011-0360-2
P.C. Okafor, C.B. Liu, Y.J. Zhu, Y.G. Zheng, Ind. Eng. Chem. Res. 50, 7273 (2011)
G. Ji, S.K. Shukla, P. Dwivedi, S. Sundaram, R. Prakash, Ind. Eng. Chem. Res. 50, 11954 (2011)
J. Zhao, N. Zhang, Q. Chengtun, J. Zhang, X. Zhang, Ind. Eng. Chem. Res. 49, 12452 (2010)
H. Gerengi, H.I. Sahin, Ind. Eng. Chem. Res. 51, 780–787 (2012)
A. Gülşen, Res. Chem. Intermed. 38, 1312 (2012)
S.-Y. Baek, Y.-W. Kim, S.-H. Yoo, K. Chung, N.-K. Kim, J.-S. Kim, Ind. Eng. Chem. Res. 51, 9670 (2012)
S.L.A. Kumar, M. Gopiraman, M.S. Kumar, A. Sreekanth, Ind. Eng. Chem. Res. 50, 7824 (2011)
B.M. Mistry, N.S. Patel, M.J. Patel, S. Jauhari, Res. Chem. Intermed. 37, 663 (2011)
H.B. Ouici, O. Benali, Y. Harek, L. Larabi, B. Hammouti, A. Guendouzi, Res. Chem. Intermed. 39, 2780 (2013)
Acknowledgment
The authors thank the Engineering Research Center of Oilfield Chemistry, Ministry of Education Key for experiment condition support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, T., Cao, S., Quan, H. et al. Synthesis and corrosion inhibition performance of alkyl triazole derivatives. Res Chem Intermed 41, 2709–2724 (2015). https://doi.org/10.1007/s11164-013-1381-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-013-1381-z