Abstract
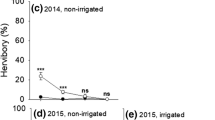
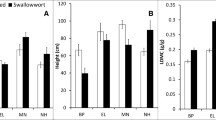
Unpalatable plants can protect palatable neighbor plants from grazing pressure, but morphological evolution of a palatable species might change its interactions with unpalatable plants. We predicted that when a palatable species has locally adapted to grazing by expressing a dwarf phenotype that reduces grazer accessibility, the dwarf plants experience relatively more competitive effects than facilitative effects from large, well-defended, unpalatable species. We used a transplant experiment, in which both dwarf and large ecotypes of a palatable annual species, Persicaria longiseta, were transplanted outside and inside the canopy of an unpalatable nettle, Urtica thunbergiana, in a long-term deer grazing habitat of Nara Park, Japan. The dwarf ecotype of Persicaria has adapted to the grazing environments of the park by exhibiting inherently short shoots and small leaves, whereas the large ecotype is found in habitats with no grazing history. A previous common-garden study suggested that the phenotypic differences were genetically based and that phenotypic plasticity contributed little to the morphological difference. The large-phenotype of Persicaria experienced significantly increased morphological size, survival, and reproductive output under the Urtica canopy compared to outside the canopy, whereas these traits of the dwarf phenotype were reduced under the Urtica canopy compared to outside. These results indicate that the net effects of Urtica on Persicaria were positive for the large ecotype and negative for the dwarf ecotype. Thus, the morphological adaptation of a palatable species to avoid grazing altered its interactions with a large, well-defended neighbor.



Similar content being viewed by others
References
Alberti J, Escapa M, Iribarne O, Silliman B, Bertness M (2008) Crab herbivory regulates plant facilitative and competitive processes in Argentinean marshes. Ecology 89:155–164
Baraza E, Zamora R, Hodar JA (2006) Conditional outcomes in plant-herbivore interactions: neighbours matter. Oikos 113:148–156
Bossuyt B, De Fre B, Hoffmann M (2005) Abundance and flowering success patterns in a short-term grazed grassland: early evidence of facilitation. J Ecol 93:1104–1114
Briske DD, Anderson VJ (1992) Competitive ability of the bunchgrass Schizachyrium scoparium as affected by grazing history and defoliation. Vegetatio 103:41–49
Bronstein JL (2009) The evolution of facilitation and mutualism. J Ecol 97:1160–1170
Brooker RW, Scott D, Palmer SCF, Swaine E (2006) Transient facilitative effects of heather on Scots pine along a grazing disturbance gradient in Scottish moorland. J Ecol 94:637–645
Brooker RW, Maestre FT, Callaway RM, Lortie CL, Cavieres LA, Kunstler G, Liancourt P, Tielbörger K, Travis JMJ, Anthelme F, Armas C, Coll L, Corcket E, Delzon S, Forey E, Kikvidze Z, Olofsson J, Pugnaire F, Quiroz CL, Saccone P, Schiffers K, Seifan M, Touzard B, Michalet R (2008) Facilitation in plant communities: the past, the present, and the future. J Ecol 96:18–34
Callaway RM (1997) Positive interactions in plant communities and the individualistic-continuum concept. Oecologia 112:143–149
Callaway RM, Kikvidze Z, Kikodze D (2000) Facilitation by unpalatable weeds may conserve plant diversity in overgrazed meadows in the Caucasus Mountains. Oikos 89:275–282
Callaway RM, Kikodze D, Chiboshvili M, Khetsuriani L (2005) Unpalatable plants protect neighbors from grazing and increase plant community diversity. Ecology 86:1856–1862
Detling JK, Painter EL (1983) Defoliation responses of western wheatgrass populations with diverse histories of prairie dog grazing. Oecologia 57:65–71
Eccles NS, Esler KJ, Cowling RM (1999) Spatial pattern analysis in Namaqualand desert plant communities: evidence for general positive interactions. Plant Ecol 142:71–85
Ehlers BK, Thompson J (2004) Do co-occurring plant species adapt to one another? The response of Bromus erectus to the presence of different Thymus vulgaris chemotypes. Oecologia 141:511–518
Fahnestock JC, Detling JK (2000) Morphological and physiological responses of perennial grasses to long-term grazing in the Pryor mountains, Montana. Am Mid Nat 143:312–320
Graff P, Aguiar MR, Chaneton EJ (2007) Shifts in positive and negative plant interactions along a grazing intensity gradient. Ecology 88:188–199
Hartvigsen G, McNaughton SJ (1995) Tradeoff between height and relative growth rate in a dominant grass from the Serengeti ecosystem. Oecologia 102:273–276
Ishikawa N, Yokoyama J, Ikeda H, Takabe E, Tsukaya H (2006) Evaluation of morphological and molecular variation in Plantago asiatica var. densiuscula, with special reference to the systematic treatment of Plantago asiatica var. yakusimensis. J Plant Res 119: 385–395
Japan Meteorological Agency (2011) http://www.jma.go.jp/jma/ (in Japanese). Accessed 11 April 2011
Kato T, Ishida K, Sato H (2008) The evolution of nettle resistance to heavy deer browsing. Ecol Res 23:339–345
Kotanen PM, Bergelson J (2000) Effects of simulated grazing on different genotypes of Bouteloua gracilis: how important is morphology? Oecologia 123:66–74
Liancourt P, Callaway RM, Michalet R (2005) Stress tolerance and competitive-response ability determine the outcome of biotic interactions. Ecology 86:1611–1618
Maestre FT, Callaway RM, Valladares F, Lortie CJ (2009) Refining the stress-gradient hypothesis for competition and facilitation in plant communities. J Ecol 97:199–205
Manier D, Hobbs NT (2006) Large herbivores influence the composition and diversity of shrub-steppe communities in the Rocky Mountains, USA. Oecologia 146:641–651
McGuire R, Agrawal AA (2005) Trade-offs between the shade-avoidance response and plant resistance to herbivores? Tests with mutant Cucumis sativus. Funct Ecol 19:1025–1031
McKinney KK, Fowler NL (1991) Genetic adaptations to grazing and mowing in the unpalatable grass Cenchrus incertus. Oecologia 88:238–242
McNaughton SJ (1984) Grazing lawns: animals in herds, plant form, and coevolution. Am Nat 124:863–886
Michalet R, Xiao S, Touzard B, Smith DS, Cavieres LA, Callaway RM, Whitham TG (2011) Phenotypic variation in nurse traits and community feedbacks define an alpine community. Ecol Lett 14(5):433–443
Oesterheld M, Oyarzabal M (2004) Grass-to-grass protection from grazing in a semi-arid steppe. Facilitation, competition, and mass effect. Oikos 107:576–582
Osem Y, Perevolotsky A, Kigel J (2004) Site productivity and plant size explain the response of annual species to grazing exclusion in a Mediterranean semi-arid rangeland. J Ecol 92:297–309
Osem Y, Perevolotsky A, Kigel J (2007) Interactive effects of grazing and shrubs on the annual plant community in semi-arid Mediterranean shrublands. J Veg Sci 18:869–878
Polley HW, Detling JK (1988) Herbivory tolerance of Agropyron smithii populations with different grazing histories. Oecologia 77:261–267
R development Core Team (2008) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rebollo S, Milchunas DG, Noy-Meir I, Chapman PL (2002) The role of a spiny plant refuge in structuring grazed shortgrass steppe plant communities. Oikos 98:53–64
Russell FL, Fowler NL (2004) Effects of white-tailed deer on the population dynamics of acorns, seedlings and small saplings of Quercus buckleyi. Plant Ecol 173:59–72
Smit C, Vandenberghe C, den Ouden J, Müller-Schärer H (2007) Nurse plants, tree saplings and grazing pressure: changes in facilitation along a biotic environmental gradient. Oecologia 152:265–273
Suzuki RO (2008) Dwarf morphology of the annual plant Persicaria longiseta as a local adaptation to a grazed habitat, Nara Park, Japan. Plant Spec Biol 23:174–182
Suzuki RO, Suzuki SN (2011a) Facilitative and competitive effects of a large species with defensive traits on a grazing-adapted, small species in a long-term deer grazing habitat. Plant Ecol 212:343–351
Suzuki SN, Suzuki RO (2011b) Distance-dependent shifts in net effects by an unpalatable nettle on a palatable plant species. Acta Oecologica 37:386–392
Suzuki RO, Kato T, Maesako Y, Furukawa A (2009) Morphological and population responses to deer grazing for herbaceous species in Nara Park, western Japan. Plant Spec Biol 24:145–155
Thorpe AS, Aschehoug ET, Atwater DZ, Callaway RM (2011) Interactions among plants and evolution. J Ecol 99:729–740
Tirado R, Pugnaire FI (2003) Shrub spatial aggregation and consequences for reproductive success. Oecologia 136:296–301
Vandenberghe C, Smit C, Pohl M, Buttler A, Freléchoux F (2009) Does the strength of facilitation by nurse shrubs depend on grazing resistance of tree saplings? Basic Appl Ecol 10:427–436
Acknowledgments
We thank the Toudai-ji Temple and the Nara Park Management Office for permission to conduct research within Nara Park. We also thank Harumi Torii for permission to use an experimental garden in at the Nara University of Education, Yuki Imanishi and Miho Kamakura for research assistance, and Yuri Maesako and Teiko Kato for their valuable advice on field research. We appreciate two anonymous reviewers and the associate editor, Dr. William E. Rogers whose helpful comments significantly improved our manuscript. This study was supported by the Japan Society for the Promotion of Science [Grant-in-Aid for Young Scientists (B) 19770016 to R. O. Suzuki].
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suzuki, R.O., Suzuki, S.N. Morphological adaptation of a palatable plant to long-term grazing can shift interactions with an unpalatable plant from facilitative to competitive. Plant Ecol 213, 175–183 (2012). https://doi.org/10.1007/s11258-011-0012-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-011-0012-2