Abstract
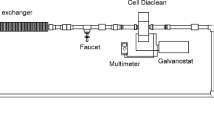
The goal of this research was the electrooxidation (EO) of a nonionic surfactant nonylphenol decaethoxylate (NP-10) in aqueous solution and denim wastewater. Three different configuration systems were evaluated in batch cells using a boron-doped diamond (BDD) anode; copper, iron, and BDD were used as cathodes. The EO process was carried out in a batch process, in a glass cell with a capacity of 1000 mL. The anode surface area was 0.0307 m2 and 1–3 A of current intensity were applied (3, 6, 10 mA/cm2) with an electrolysis time of 240 min for aqueous solution and 780 min for denim wastewater in order to investigate the degradation of the surfactant. The processes were analyzed in terms of chemical oxygen demand (COD) and total organic carbon (TOC). The maximum mineralization efficiency in aqueous solution for the BDD-Cu electrooxidation system was 92.2% for COD and 45.6% for TOC at pH 2 and 3 mA/cm2 of current intensity. For denim wastewater, the removal efficiency was 44.1% for COD and 26.5% for TOC at pH 4.5 and 6 mA/cm2 of current intensity, using a BDD-BDD system. The raw and treated (aqueous solution and denim) wastewater were characterized by UV-Vis and infrared spectroscopy.












Similar content being viewed by others
References
APHA & AWWA. (2012). Standard methods for examination of water and wastewater. Washington DC: American Public Health Association.
Ashar, A., Iqbal, M., Bhatti, I. A., Ahmad, M. Z., Qureshi, K., Nisar, J., & Bukhari, I. H. (2016). Synthesis, characterization and photocatalytic activity of ZnO flower and pseudo-sphere: nonylphenol ethoxylate degradation under UV and solar irradiation. Journal of Alloys and Compounds, 678, 126–136.
Babuponnusami, A., & Muthukumar, K. (2012). Advanced oxidation of phenol: a comparison between Fenton, electro-Fenton, sono-electro-Fenton and photo-electro-Fenton processes. Chemical Engineering Journal, 183, 1–9.
Bokare, A. D., & Choi, W. (2014). Review of iron-free Fenton-like systems for activating H2O2 in advanced oxidation processes. Journal of Hazardous Materials, 275, 121–135.
Chen, J., & Mullin, C. A. (2014). Determination of nonylphenol ethoxylate and octylphenol ethoxylate surfactants in beehive samples by high performance liquid chromatography coupled to mass spectrometry. Food Chemistry, 158, 473–479.
Chen, L., Zhou, H. Y., Liu, L., & Deng, Q. Y. (2007). Mechanism study on UV-induced photodegradation of nonylphenol ethoxylates by intermediate products analysis. Chinese Chemical Letters, 18, 473–475.
Cherniaev, A. P., Kondakova, A. S., & Zyk, E. N. (2016). Contents of 4-Nonylphenol in surface sea water of Amur Bay (Japan/East Sea). Achievements in the Life Sciences, 10, 65–71.
da Silva, S. W., Bordignon, G. L., Viegas, C., Rodrigues, M. A. S., Arenzon, A., & Bernardes, A. M. (2015a). Treatment of solutions containing nonylphenol ethoxylate by photoelectrooxidation. Chemosphere, 119 Supplement, S101–S108.
da Silva, S. W., Klauck, C. R., Siqueira, M. A., & Bernardes, A. M. (2015b). Degradation of the commercial surfactant nonylphenol ethoxylate by advanced oxidation processes. Journal of Hazardous Materials, 282, 241–248.
da Silva, S. W., Bortolozzi, J. P., Banús, E. D., Bernardes, A. M., & Ulla, M. A. (2016). TiO2 thick films supported on stainless steel foams and their photoactivity in the nonylphenol ethoxylate mineralization. Chemical Engineering Journal, 283, 1264–1272.
EPA. (2005). Ambient aquatic life water quality criteria: nonylphenol final (p. 96). Washington, DC: Office of Sciencie and Technology.
Fan, J., Yang, W., & Li, A. (2011). Adsorption of phenol, bisphenol A and nonylphenol ethoxylates onto hypercrosslinked and aminated adsorbents. Reactive and Functional Polymers, 71, 994–1000.
Gao, D., Li, Z., Guan, J., Li, Y., & Ren, N. (2014). Removal of surfactants nonylphenol ethoxylates from municipal sewage-comparison of an A/O process and biological aerated filters. Chemosphere, 97, 130–134.
Gao, D., Li, Z., Guan, J., & Liang, H. (2017). Seasonal variations in the concentration and removal of nonylphenol ethoxylates from the wastewater of a sewage treatment plant. Journal of Environmental Sciences., 54, 217–223.
García-García, A., Martínez-Miranda, V., Martínez-Cienfuegos, I. G., Almazán-Sánchez, P. T., Castañeda-Juárez, M., & Linares-Hernández, I. (2015). Industrial wastewater treatment by electrocoagulation–electrooxidation processes powered by solar cells. Fuel, 149, 46–54.
González, M. M., Martín, J., Santos, J. L., Aparicio, I., & Alonso, E. (2010). Occurrence and risk assessment of nonylphenol and nonylphenol ethoxylates in sewage sludge from different conventional treatment processes. Science of the Total Environment, 408, 563–570.
Iqbal, M., & Bhatti, I. A. (2015). Gamma radiation/H2O2 treatment of a nonylphenol ethoxylates: Degradation, cytotoxicity, and mutagenicity evaluation. Journal of Hazardous Materials, 299, 351–360.
Korsman, J. C., Schipper, A. M., de Vos, M. G., van den Heuvel-Greve, M. J., Vethaak, A. D., de Voogt, P., & Hendriks, A. J. (2015). Modeling bioaccumulation and biomagnification of nonylphenol and its ethoxylates in estuarine–marine food chains. Chemosphere, 138, 33–39.
Kwon, K. D., & Kubicki, J. D. (2004). Molecular orbital theory study on surface complex structures of phosphates to iron hydroxides: calculation of vibrational frequencies and adsorption energies. Langmuir, 20, 9249–9254.
Lu, J., Jin, Q., He, Y., & Wu, J. (2008). Enhanced anaerobic biodegradation of nonylphenol ethoxylates by introducing additional sulfate or nitrate as terminal electron acceptors. International Biodeterioration & Biodegradation, 62, 214–218.
Lyu, L., Zhang, L., & Hu, C. (2015). Enhanced Fenton-like degradation of pharmaceuticals over framework copper species in copper-doped mesoporous silica microspheres. Chemical Engineering Journal, 274, 298–306.
Ömeroğlu, S., Kara Murdoch, F., & Dilek Sanin, F. (2015). Investigation of nonylphenol and nonylphenol ethoxylates in sewage sludge samples from a metropolitan wastewater treatment plant in Turkey. Talanta, 131, 650–655.
Perrotta, I., & Tripepi, S. (2012). Ultrastructural alterations in the ventricular myocardium of the adult italian newt (Lissotriton italicus) following exposure to nonylphenol ethoxylate. Micron, 43, 183–191.
Pessala, P., Keränen, J., Schultz, E., Nakari, T., Karhu, M., Ahkola, H., Knuutinen, J., Herve, S., Paasivirta, J., & Ahtiainen, J. (2009). Evaluation of biodegradation of nonylphenol ethoxylate and lignin by combining toxicity assessment and chemical characterization. Chemosphere, 75, 1506–1511.
Yavuz, Y., & Koparal, A. S. (2006). Electrochemical oxidation of phenol in a parallel plate reactor using ruthenium mixed metal oxide electrode. Journal of Hazardous Materials, 136, 296–302.
Yu, Y., Zhai, H., Hou, S., & Sun, H. (2009). Nonylphenol ethoxylates and their metabolites in sewage treatment plants and rivers of Tianjin, China. Chemosphere, 77, 1–7.
Zhang, Q., Wang, F., Xue, C., Wang, C., Chi, S., & Zhang, J. (2016). Comparative toxicity of nonylphenol, nonylphenol-4-ethoxylate and nonylphenol-10-ethoxylate to wheat seedlings (Triticum aestivum L.) Ecotoxicology and Environmental Safety, 131, 7–13.
Acknowledgments
The authors appreciate the support given by CONACyT through project no. 219743 and the scholarship granted for Vences-Benitez (CVU 622200).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vences-Benitez, J.C., Martínez-Miranda, V., Linares-Hernández, I. et al. Electrooxidation Performance of Aqueous Solution of Nonylphenol Decaethoxylate and Denim Wastewater. Water Air Soil Pollut 228, 393 (2017). https://doi.org/10.1007/s11270-017-3576-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-017-3576-1