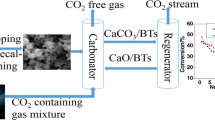
Abstract
Using lime mud (LM) purified by sucrose method, derived from paper-making industry, as calcium precursor, and using mineral rejects–bauxite-tailings (BTs) from aluminum production as dopant, the CaO-based sorbents for high-temperature CO2 capture were prepared. Effects of BTs content, precalcining time, and temperature on CO2 cyclic absorption stability were illustrated. The cyclic carbonation behavior was investigated in a thermogravimetric analyzer (TGA). Phase composition and morphologies were analyzed by XRD and SEM. The results reflected that the as-synthesized CaO-based sorbent doped with 10 wt% BTs showed a superior CO2 cyclic absorption–desorption conversion during multiple cycles, with conversion being >38 % after 50 cycles. Occurrence of Ca12Al14O33 phase during precalcination was probably responsible for the excellent CO2 cyclic stability.








Similar content being viewed by others
References
Angeli SD, Martavaltzi CS, Lemonidou AA (2014) Development of a novel-synthesized Ca-based CO2 sorbent for multicycle operation: parametric study of sorption. Fuel 127:62–69. doi:10.1016/j.fuel.2013.10.046
Borgwardt RH (1989) Sintering of nascent calcium oxide. Chem Eng Sci 44:53–60. doi:10.1016/0009-2509(89)85232-7
Broda M, Manovic V, Anthony EJ, Mueller CR (2014) Effect of pelletization and addition of steam on the cyclic performance of carbon-templated, CaO-based CO2 sorbents. Environ Sci Technol 48:5322–5328. doi:10.1021/es405668f
Butler JW, Lim CJ, Grace JR (2014) Kinetics of CO2 absorption by CaO through pressure swing cycling. Fuel 127:78. doi:10.1016/j.fuel.2013.09.058
Chen H, Zhao C, Yang Y (2013) Enhancement of attrition resistance and cyclic CO2 capture of calcium-based sorbent pellets. Fuel Process Technol 116:116–122. doi:10.1016/j.fuproc.2013.05.012
Dai Q-X, Ma L-P, Yan B, Xie L-G, Mao Y, Zhang H, Zi Z-C (2015) Purification of calcium oxide in phosphogypsum decomposition residue based on the sucrose-CO2 method. Sep Sci Technol 50:479–486. doi:10.1080/01496395.2014.950671
He D, Barr C (2004) Chinas pulp and paper sector: an analysis of supply–demand and medium term projections. Int For Rev 6:254–266. doi:10.1505/ifor.6.3.254.59970
Hu Y-C, Jia Q-M, Shan S-Y, Li S-M, Jiang L-H, Wang Y-M (2015) Development of CaO-based sorbent doped with mineral rejects–bauxite-tailings in cyclic CO2 capture. J Taiwan Inst Chem Eng 46:155–159. doi:10.1016/j.jtice.2014.09.020
Kargari N, Mastouri R (2011) Effect of nuclear power on CO2 emission from power plant sector in Iran. Environ Sci Pollut Res Int 18:116–122. doi:10.1007/s11356-010-0402-3
Li Y-J, Zhao C-S, Ren Q-Q, Duan L-B, Chen H-C, Chen X-P (2009) Effect of rice husk ash addition on CO2 capture behavior of calcium-based sorbent during calcium looping cycle. Fuel Process Technol 90:825–834. doi:10.1016/j.fuproc.2009.03.013
Li Y-J, Liu C-T, Sun R-Y, Liu H-L, Wu S-M, Lu C-M (2012) Sequential SO2/CO2 capture of calcium-based solid waste from the paper industry in the calcium looping process. Ind Eng Chem Res 51:16042–16048. doi:10.1021/ie301375g
Li B, Duan Y, Luebke D, Morreale B (2013) Advances in CO2 capture technology: a patent review. Appl Energy 102:1439–1447. doi:10.1016/j.apenergy.2012.09.009
Li Y-J, Su M-Y, Xie X, Wu S-M, Liu C-T (2015) CO2 capture performance of synthetic sorbent prepared from carbide slag and aluminum nitrate hydrate by combustion synthesis. Appl Energy 145:60–68. doi:10.1016/j.apenergy.2015.01.061
Liu W-Q, An H, Qin C-L, Yin J-J, Wang G-X, Feng B, Xu M-H (2012) Performance enhancement of calcium oxide sorbents for cyclic CO2 capture—a Review. Energy Fuel 26:2751–2767. doi:10.1021/ef300220x
Luo C, Zheng Y, Zheng C-G, Yin J-J, Qin C-L, Feng B (2013) Manufacture of calcium-based sorbents for high temperature cyclic CO2 capture via a sol–gel process. Int J Greenhouse Gas Control 12:193–199. doi:10.1016/j.ijggc.2012.11.011
MacKenzie A, Granatstein DL, Anthony EJ, Abanades JC (2007) Economics of CO2 capture using the calcium cycle with a pressurized fluidized bed combustor. Energy Fuel 21:920–926. doi:10.1021/ef0603378
Manovic V, Anthony EJ (2009) Screening of binders for pelletization of CaO-based sorbents for CO2 capture. Energy Fuel 23:4797–4804. doi:10.1021/ef900266d
Reddy GK, Quillin S, Smirniotis P (2014) Influence of the synthesis method on the structure and CO2 adsorption properties of Ca/Zr sorbents. Energy Fuel 28:3292–3299. doi:10.1021/ef402573u
Salman M et al (2014) Effect of accelerated carbonation on AOD stainless steel slag for its valorisation as a CO2-sequestering construction material. Chem Eng J 246:39–52. doi:10.1016/j.cej.2014.02.051
Sanchez-Jimenez PE, Perez-Maqueda LA, Valverde JM (2014) Nanosilica supported CaO: a regenerable and mechanically hard CO2 sorbent at Ca-looping conditions. Appl Energy 118:92–99. doi:10.1016/j.apenergy.2013.12.024
Sun R-Y, Li Y-J, Liu C-T, Xie X, Lu C-M (2013) Utilization of lime mud from paper mill as CO2 sorbent in calcium looping process. Chem Eng J 221:124–132. doi:10.1016/j.cej.2013.01.068
Tian S, Jiang J, Yan F, Li K, Chen X (2015) Synthesis of highly efficient CaO-based, self-stabilizing CO2 sorbents via structure-reforming of steel slag. Environ Sci Technol 49:7464–7472. doi:10.1021/acs.est.5b00244
Wang M, Lee C, Ryu C (2008) CO2 sorption and desorption efficiency of Ca2SiO4. Int J Hydrog Energy 33:6368–6372. doi:10.1016/j.ijhydene.2008.07.114
Wang Y, Zhu Y-Q, Wu S-F (2013) A new nano CaO-based CO2 adsorbent prepared using an adsorption phase technique. Chem Eng J 218:39–45. doi:10.1016/j.cej.2012.11.095
Witoon T (2011) Characterization of calcium oxide derived from waste eggshell and its application as CO2 sorbent. Ceram Int 37:3291–3298. doi:10.1016/j.ceramint.2011.05.125
Yu C-T, Chen W-C (2014) Hydrothermal preparation of calcium–aluminum carbonate sorbent for high-temperature CO2 capture in fixed-bed reactor. Fuel 122:179–185. doi:10.1016/j.fuel.2014.01.022
Yu F-C, Fan L-S (2011) Kinetic study of high-pressure carbonation reaction of calcium-based sorbents in the calcium looping process (CLP). Ind Eng Chem Res 50:11528–11536. doi:10.1021/ie200914e
Zhang H-W, Hong X (2011) An overview for the utilization of wastes from stainless steel industries. Resour Conserv Recycl 55:745–754. doi:10.1016/j.resconrec.2011.03.005
Zhang G-C, Li X, Li Y-J, Wu T, Sun D-J, Lu F-J (2011) Removal of anionic dyes from aqueous solution by leaching solutions of white mud. Desalination 274:255–261. doi:10.1016/j.desal.2011.02.016
Zhang J-S, Wang Q-Q, Jiang J-G (2013) Lime mud from paper-making process addition to food waste synergistically enhances hydrogen fermentation performance. Int J Hydrog Energy 38:2738–2745. doi:10.1016/j.ijhydene.2012.12.048
Zhang J-S, Zheng P-W, Wang Q-Q (2014a) Lime mud from papermaking process as a potential ameliorant for pollutants at ambient conditions: a review. J Clean Prod 103:828–836. doi:10.1016/j.jclepro.2014.06.052
Zhang X-Y, Li Z-G, Peng Y, Su W-K, Sun X-X, Li J-H (2014b) Investigation on a novel CaO–Y2O3 sorbent for efficient CO2 mitigation. Chem Eng J 243:297–304. doi:10.1016/j.cej.2014.01.017
Zhenissova A, Micheli F, Rossi L, Stendardo S, Foscolo PU, Gallucci K (2014) Experimental evaluation of Mg- and Ca-based synthetic sorbents for CO2 capture. Chem Eng Res Des 92:727–740. doi:10.1016/j.cherd.2013.11.005
Zhou Z-M, Qi Y, Xie M-M, Cheng Z-M, Yuan W-K (2012) Synthesis of CaO-based sorbents through incorporation of alumina/aluminate and their CO2 capture performance. Chem Eng Sci 74:172–180. doi:10.1016/j.ces.2012.02.042
Acknowledgments
The National Natured Science Foundation of China (No. 2014FB129, 51104075, 51364023, and 31160146) provided financial supports for this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Ma, A., Jia, Q., Su, H. et al. Study of CO2 cyclic absorption stability of CaO-based sorbents derived from lime mud purified by sucrose method. Environ Sci Pollut Res 23, 2530–2536 (2016). https://doi.org/10.1007/s11356-015-5477-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-015-5477-4