Abstract
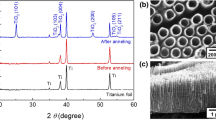
The mechanism of the conversion of titanate nanotubes into nanoribbons is of considerable interest. The details of the transformation processes involved when nanoribbons are produced from a P25 TiO2 powder precursor by alkaline hydrothermal treatment have been investigated systematically by transmission electron microscopy. A multistep attachment model is proposed for the growth at the early stage of coarsening. The treatment duration has a strong effect on the change in product morphology from hollow nanotubes into nanoribbons, since the nanotubes cannot retain their morphology in the strong alkaline solution for extended periods of time. Most of the nanotubes were etched and dissolved, providing the nutrients for subsequent nanoribbon growth. Some stable nanotubes grew spirally internally to form nanowires or became connected together to form rafts which acted as the grains for nanoribbon growth. With increasing hydrothermal time, a large number of nanotubes and other fragments became attached to the grains which began to grow larger and eventually formed the nanoribbons, in a process in which the stepped faces and kinked faces became fused and were eliminated while the flat faces were retained in the nanoribbon morphology.
Similar content being viewed by others
References
Kasuga T, Hiramatsu M, Hoson A, Sekino T, Niihara K. Formation of titanium oxide nanotube. Langmuir, 1998, 14: 3160–3163
Chen GHD, Zhang S, Peng LM. The structure of trititanate nanotubes. Acta Crystallographica Section B, 2002, 58: 587–593
Yuan ZY, Colomer JF, Su BL. Titanium oxide nanoribbons. Chem Phys Lett, 2002, 363: 362–366
Bavykin JMF, Walsh FC. Protonated titanates and TiO2 nanostruc-tured materials: Synthesis, properties, and applications. Adv Mater, 2006,18: 2807–2824
Chen Q, Zhou WZ, Du GH, Peng LM. Trititanate nanotubes made via a single alkali treatment. Adv Mater, 2002, 14: 1208–1211
Tsai CC, Teng H. Structural features of nanotubes synthesized from NaOH treatment on TiO2 with different post-treatments. Chem Mater, 2005, 18: 367–373
Wu D, Liu J, Zhao X, Li A, Chen Y, Ming N. Sequence of events for the formation of titanate nanotubes, nanofibers, nanowires, and nanobelts. Chem Mater, 2005, 18: 547–553
Wen BM, Liu CY, Liu Y. Solvothermal synthesis of ultralong single-crystalline TiO2 nanowires. New J Chem, 2005, 29: 969–971
Armstrong AR, Armstrong G, Canales J, Bruce PG. TiO2-B nanowires. Angew Chem-Int Edit, 2004, 43: 2286–2288
Song SA, Liu ZW, He ZQ, Zhang AL, Chen JM. Impacts of morphology and crystallite phases of titanium oxide on the catalytic ozonation of phenol. Environ Sci Technol, 2010, 44: 3913–3918
Xiao D. Template synthesis of N-F-codoped TiO2 nanotubes with high visible light activity. Sci China Ser B-Chem, 2009, 52: 2043–2046
Bavykin DV, Lapkin AA, Plucinski PK, Friedrich JM, Walsh FC. Reversible storage of molecular hydrogen by sorption into multilayered TiO2 nanotubes. J Phys Chem B, 2005, 109: 19422–19427
Xu JW, Ha CH, Cao B, Zhang WF. Electrochemical properties of anatase TiO2 nanotubes as an anode material for lithium-ion batteries. Electrochimica Acta, 2007, 52: 8044–8047
Adachi M, Okada I, Ngamsinlapasathian S, Murata Y, Yoshikawa S. Dye-sensitized solar cells using semiconductor thin film composed of titania nanotubes. Electrochemistry, 2002, 70: 449–452
Uchida S, Chiba R, Tomiha M, Masaki N, Shirai M. Application of titania nanotubes to a dye-sensitized solar cell. Electrochemistry, 2002, 70: 418–420
Sheng J, Hu L, Xu S, Liu W, Mo Le, Tian H, Dai S. Characteristics of dye-sensitized solar cells based on the TiO2 nanotube/nanoparticle composite electrodes. J Mater Chem, 2011, 21: 5457–5463
Riss A, Elser MJ, Bernardi J, Diwald O. Stability and photoelectronic properties of layered titanate nanostructures. J Amer Chem Soc, 2009, 131: 6198–6206
Humar M, Arcon D, Umek P, Skarabot M, Musevic I, Bregar G. Mechanical properties of titania-derived nanoribbons. Nanotechnology, 2006, 17: 3869–3872
Yuan ZY, Su BL. Titanium oxide nanotubes, nanofibers and nanowires. Colloids Surf A, 2004, 241: 173–183
Wei M, Konishi Y, Zhou H, Sugihara H, Arakawa H. A simple method to synthesize nanowires titanium dioxide from layered titanate particles. Chem Phys Lett, 2004, 400: 231–234
Ma RZ, Fukuda K, Sasaki T, Osada M, Bando Y. Structural features of titanate nanotubes/nanobelts revealed by Raman, X-ray absorption fine structure and electron diffraction characterizations. J Phys Chem B, 2005, 109: 6210–6214
Yu HG, Yu JG, Cheng B, Zhou MH. Effects of hydrothermal post-treatment on microstructures and morphology of titanate nanoribbons. J Solid State Chem, 2006, 179: 349–354
Bavykin DV, Parmon VN, Lapkin AA, Walsh FC. The effect of hydrothermal conditions on the mesoporous structure of TiO2 nano-tubes. J Mater Chem, 2004, 14: 3370–3377
Elsanousi A, Elssfah EM, Zhang J, Lin J, Song HS, Tang C. Hydrothermal treatment duration effect on the transformation of titanate nanotubes into nanoribbons. J Phys Chem C, 2007, 111: 14353–14357
Feng XJ, Shankar K, Varghese OK, Paulose M, Latempa TJ, Grimes CA. Vertically aligned single crystal TiO2 nanowire arrays grown directly on transparent conducting oxide coated glass: Synthesis details and applications. Nano Lett, 2008, 8: 3781–3786
Papa AL, Millot N, Saviot L, Chassagnon R, Heintz O. Effect of reaction parameters on composition and morphology of titanate nanomaterials. J Phys Chem C, 2009, 113: 12682–12689
Bavykin DV, Walsh FC. Elongated titanate nanostructures and their applications. Eur J Inorg Chem, 2009, 977-997
Yao BD, Chan YF, Zhang XY, Zhang WF, Yang ZY, Wang N. Formation mechanism of TiO2 nanotubes. Appl Phys Lett, 2003, 82: 281–283
Pina CM, Becker U, Risthaus P, Bosbach D, Putnis A. Molecular-scale mechanisms of crystal growth in barite. Nature, 1998, 395: 483–486
Koutsopoulos S. Kinetic study on the crystal growth of hydroxyapatite. Langmuir, 2001, 17: 8092–8097
Cahn JW, Hoffman DW. Vector thermodynamics for anisotropic surfaces curved and faceted surfaces. Acta Metallurgica, 1974, 22: 1205–1214
Donnay JDH, Harker D. A new law of crystal morphology extending the law of bravais. Amer Miner, 1937, 22: 446–467
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sheng, J., Hu, L., Mo, L. et al. A multistep attachment process: Transformation of titanate nanotubes into nanoribbons. Sci. China Chem. 55, 368–372 (2012). https://doi.org/10.1007/s11426-011-4362-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-011-4362-3