Abstract
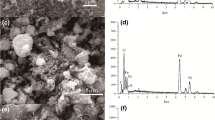
Porous chitosan(CS)/magnetic(Fe3O4)/ferric hydroxide(Fe(OH)3) microsphere as novel and low-cost adsorbents for the removal of As(III) have been synthesized via the electrospraying technology by a simple process of two steps. Characterization of the obtained adsorbents was studied by transmission electron microscopy (TEM), scanning electron microscopy (SEM), X-ray photoelectron spectroscopy (XPS), and X-ray diffraction (XRD). The adsorption kinetics and equilibrium isotherms were investigated in batch experiments. The Langmuir, Freundlich isotherm and pseudo-second order kinetic models agree well with the experimental data. The adsorption of As(III) onto CS/Fe3O4/Fe(OH)3 microspheres occurred rapidly and reached adsorption equilibrium after about 45 min. The maximum adsorption capacity of CS/Fe3O4/Fe(OH)3 microspheres, calculated by the Langmuir isotherm model, was 8.47 mg g−1, which is higher than that of CS/Fe3O4/Fe(OH)3 prepared by the conventional method (4.72 mg g−1). The results showed that the microspheres had a high adsorption capacity for As(III) and a high separation efficiency due to their microporous structure and superparamagnetic characteristics. Present research may eventually lead to a simple and low cost method for fabricating microporous materials and application for removal of arsenic from aqueous solution.
Similar content being viewed by others
References
Bang SB, Patel M, Lippincott L, Meng XG. Removal of arsenic from groundwater by granular titanium dioxide adsorbent. Chemosphere, 2005, 60: 389–397
Penna M, Meng XG, Korfiatis GP, Jing CY. Adsorption mechanism of arsenic on nanocrystalline titanium dioxide. Environ Sci Technol, 2006, 40: 1257–1262
Camacho LM, Gutierrez MT, Alarcon-Herrera ML, Deng SG. Occurrence and treatment of arsenic in groundwater and soil in northern Mexico and southwestern USA. J Hazard Mater, 2011, 83: 211–225
WHO, Guidelines for drinking water quality, World Health Organization, Geneva, Switzerland, 1993
Guan XH, Du JS, Meng XG, Sun YK, Sun B, Hu QH. Application of titanium dioxide in arsenic removal from water: A review. J Hazard Mater, 2012, 215–216: 1–16
Elson CM, Bem EM, Ackman RG. Removal of arsenic from contaminated drinking water by a chitosan/chitin mixture. Water Res, 1980, 14: 1307–1311
Wan Ngah WS, Fatinathan S. Adsorption of Cu(II) ions in aqueous solution using chitosan beads, chitosan-GLA beads and chitosanalginate beads. Chem Eng J, 2008, 143: 62–72
Gerente C, Andres Y, McKay G, Cloirec PL. Removal of As(V) onto chitosan. From sorption mechanism explanation to dynamic water treatment process. Chem Eng J, 2010, 158: 593–598
Miller SM, Spaulding ML, Zimmerman JB. Optimization of capacity and kinetics for a novel bio-based arsenic sorbent, TiO2-impregnated chitosan bead. Water Res, 2011, 45: 5745–5754
Boddu VM, Abburi K, Talbott JL, Smith ED, Haasch R. Removal of arsenic(III) and arsenic(V) from aqueous medium using chitosancoated biosorbent. Water Res, 2008, 42: 633–642
Gang DD, Deng BL, Lin LS. As(III) removal using an iron-impregnated chitosan sorbent. J Hazard Mater, 2010, 182: 156–161
Prevost MC, Nour SKF, Jekel MT, Gallagher PM, Blumenschein CD. Kinetic and thermodynamic aspects of adsorption of arsenic onto granular ferric hydroxide (GFH). Water Res, 2008, 42: 3371–3378
Pierce ML, Moore CB. Adsorption of arsenite and arsenate on amorphous iron hydroxide. Water Res, 1982, 16: 1247–1253
Zhang K, Dwivedi VN, Chi CY, Wu JS. Graphene oxide/ferric hydroxide composites for efficient arsenate removal from drinking water. J Hazard Mater, 2010, 182: 162–168
Liu Z, Yi G, Zhang H, Ding J, Zhang Y and Xue J. Monodisperse silica nanoparticles encapsulating upconversion fluorescent and superparamagnetic nanocrystals. Chem Commun, 2008, 694–696
Liu Z, Ding J, and Xue J. A new family of biocompatible and stable magnetic nanoparticles: silica cross-linked pluronic F127 micelles loaded with iron oxides. New J Chem, 2009, 33: 88–92
Yavuz CT, Mayo JT, Yu WW, Prakash A, Falkner JC, Yean S, Cong L, Shipley HJ, Kan A, Tomson M, Natelson D, Colvin VL. Low-field magnetic separation of monodisperse Fe3O4 nanocrystals. Science, 2006, 314: 964–967
Simeonidisa K, Gkinisa Th, Tresintsib S, Martinez-Boubetac C, Vourliasa G, Tsiaoussisa I, Stavropoulosb G, Mitrakasb M, Angelakerisa M. Magnetic separation of hematite-coated Fe3O4 particles used as arsenic adsorbents. Chem Eng J, 2011, 168: 1008–1015
Mayo J T, Yavuz C, Yean S, Cong L, Shipley H, Yu W, Falkner J, Kan A, Tomson M, Colvin, V L. The effect of nanocrystalline magnetite size on arsenic removal. Sci Technol Adv Mater, 2007, 8: 71–75
Chen R, Zhi C, Yang H, Bando Y, Zhang Z, Sugiur N, Golberg D. Arsenic(V) adsorption on Fe3O4 nanoparticle-coated boron nitride nanotubes. J Colloid Interf Sci, 2011, 359: 261–268
Tian Y, Wu M, Lin X, Huang P, Huang Y. Synthesis of magnetic wheat straw for arsenic adsorption. J Hazard Mater, 2011, 193: 10–16
Savina IN, English, CJ, Whitby RLD, Zheng YS, Leistner A, Mikhalovsky SV, Cundy AB. High efficienct removal of dissolved As(III) using iron nanoparticle-embedded macroporous polymer composites. J Hazard Mater, 2011, 192: 1002–1008
Wu Y Q, Clark R L. Controllable porous polymer particles generated by electrospraying, J Colloid Interf Sci, 2007, 310: 529–535
Jaworek A, Sobczyk AT. Electrospraying route to nanotechnology: An overview. J Electrostatics, 2008, 66: 197–219
Liu ZY, Bai HW, Sun DD. Facile fabrication of porous chitosan/TiO2/Fe3O4 microspheres with multifunction for water purifications. New J Chem, 2011, 35: 137–140
Gang ZY, Yu SH, Dong PS, Qin HM. Synthesis, characterization and properties of ethylenediamine-functionalized Fe3O4 magnetic polymers for removal of Cr(VI) in wastewater. J Hazard Mater, 2010, 182: 295–302
Ngah WSW, Ghani SA, Kamari A, Adsorption behaviour of Fe(II) and Fe(III) ions in aqueous solution on chitosan and cross-linked chitosan beads. Bioresour Technol, 2005, 96: 443–451
Wang LL, Jiang JS. Preparation of Fe3O4 spherical nanoporous particles facilitated by polyethylene glycol 4000. Nanoscale Res Lett, 2009, 4: 1439–1446
Sing KSW, Everett DH, Haul RAW, Moscou L. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl Chem, 1985, 57, 603–619
Brunauer S, Emmett PH, Teller E. Adsorption of Gases in Multimolecular Layers. J Am Chem Soc, 1938, 60: 309–319
Wang JH, Zheng SR, Shao Y, Liu JL. Amino-functionalized Fe3O4@ SiO2 core-shell magnetic nanomaterial as a novel adsorbent for aqueous heavy metals removal. J Colloid Interf Sci, 2010, 349: 293–299
Peng Z G, Hidajat K, Uddin M S. Adsorption of bovine serum albumin on nanosized magnetic particle. J Colloid Interf Sci, 2004, 271: 277–283
Ho YS, McKay G. Comparative of chemisorption kinetics models applied to pollutant removal on various sorbents. J Environ Sci and Health. Part A, 1999, 34: 1179–1204
Zheng Y, Wang WB, Huang DJ, Wang AQ. Kapok fiber oriented-polyaniline nanofibers for efficient Cr(VI) removal. Chem Eng J, 2012, 191: 154–161
Langmuir I. The adsorption of gases on plane surfaces of glass, mica and platinium. J Am Chem Soc, 1918, 40: 1361–1403
Freundlich H M F. Uber die adsorption in lösungen, Zeitschrift für Physikalische Chemie (Leipzig), 1906, 57A: 385–470
Vu D, Li ZY, Zhang HN, Wang W, Wang ZJ, Xu XR, Dong B. Adsorption of Cu(II) from aqueous solution by anatase mesoporous TiO2 nanofibers prepared via electrospinning. J Colloid Interf Sci, 2012, 367: 429–435
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Vu, D., Li, X. & Wang, C. Adsorption of As(III) from aqueous solution based on porous magnetic/chitosan/ferric hydroxide microspheres prepared via electrospraying. Sci. China Chem. 56, 678–684 (2013). https://doi.org/10.1007/s11426-012-4817-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-012-4817-1