Abstract
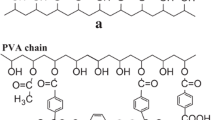
Thin sheets of polyallylamine (PAAm) and polyvinyl alcohol (PVA) blend were prepared by employing solution casting technique for potential membranes application. The blends were characterized by Fourier transform infrared (FTIR) and 1H-NMR spectroscopy, scanning electron microscopy, thermogravimetric analysis, ultraviolet–visible spectroscopy, X-ray diffraction, and mechanical properties. The zeta potential, conductivity and rheological properties of PAAm/PVA blends were also studied. The FTIR spectrum reveals that the C–H asymmetric stretching vibration band of PVA at 2,928 cm−1 disappeared in all the blend samples. Thermal stability of the blend membrane was better than pure polymers. The crystallinity of the PAAm/PVA blends was decreased, which may be due to the entanglement of PAAm in to PVA chains, which is also responsible for the improvement in the mechanical properties of the blends. Zeta potential decreases where as the conductivity increases as a function of temperature. Hydrophilicity is improved by addition of PVA to PAAm, which may be due to hydroxyl group of PVA. The blend solution shows non-Newtonian character of the liquid. By applying shear stress, increase in the effect of rarefaction was observed. The knowledge about the investigated parameters will be of vital importance for use of the blended material in membrane applications, especially where CO2 separation is in focus. The membrane performance (separation properties) of the PAAm/PVA blended material is, however, not reported in the current article.











Similar content being viewed by others
References
Fang H, Mighri F, Ajji A (2007) J Appl Polym Sci 105:2955–2962
Pawde SM, Deshmukh K (2009) J Appl Polym Sci 114:2169–2179
Shen J, Qiu J, Wu L, Gao C (2006) Sep and Purif Tech 51:345–351
Cai Y, Wang Z, Yi C, Bai Y, Wang J, Wang S (2008) J Membr Sci 310:184–196
Robsen LM (1991) J Membr Sci 62:165–185
Kim TJ, Li B, Hagg MB (2004) J Polym Sci Part B Polym Phys 42:4326–4336
Dong C, Wang Z, Yi C, Wang S (2006) J Appl Polym Sci 101:1885–1891
Zhang Y, Wang Z, Wang S (2002) J Appl Polym Sci 86:2222–2226
Zhang Y, Wang Z, Wang S (2002) Chem Lett 31:430–431
Yamasaki A, Shinbo T, Mizoguchi K (1997) J Appl Polym Sci 64:1061–1065
Shibayama M, Sato M, Kimura Y, Fujiwara H, Nomura S (1988) Polymer 29:336–340
Zidan HM (2003) J Appl Polym Sci 88:104–111
Shahawy MAE (2003) Polym Inter 52:1919–1924
Selim SM, Seodi R, Shabaka AA (2005) Mater Lett 59:2650–2654
Galindo-Rosales FJ, Rubio-Hernandez FJ, Velazquez-Navarro JF, Gomez-Merino AI (2007) J Amer Cer Soc 90:1641–1643
Amiri A, Gisle O, Sjoblom S (2009) Colloids Surf A Physicochem Eng Asp 349:43–54
Lianyu L, Fubing P, Zhougyi J, Jianghui W (2006) J Appl Polym Sci 101:167–173
Pawde SM, Deshmukh K, Parab S (2008) J Appl Polym Sci 109:1328–1337
Pawde SM, Deshmukh K (2008) J Appl Polym Sci 109:3431–3437
Shin EJ, Lee YH, Choi SC (2004) J Appl Polym Sci 91:2407–2415
Selvasekarapandian S, Hirankumar G, Kawamura J, Kuwata N, Hattori T (2005) Mater Lett 59:2741–2745
Yu D, Yang J, Xie J (1989) Handbook of analytical chemistry. Part 5: nuclear magnetic resonance. Chemistry Industry Press, Beijing
Petit JM, Zhu XX (1996) Macromolecules 29:2075–2081
Bugay DE (1993) Pharm Res 10:317–327
Guiping M, Dongzhi Y, Dandan S, Xueyan M, John FK, Jun N (2010) Polym Adv Tech 21:189–195
Finch CA (1973) Polyvinyl alcohol properties and application. Wiley, Chichester, p 493
Sperling LH (1992) Introduction to physical polymer science, vol 4, 2nd edn. Wiley Interscience, New York, p 122
Chang MC, Tanaka J (2002) Biomaterials 23:4811–4818
Chunxue Z, Xiaoyan Y, Lili W, Jing S (2006) Acta Polym Sin 1:294–297
Zhang Y, Huang X, Duan B, Wu L, Li S, Yuan X (2007) Coll Polym Sci 285:855–863
ASTM (1985) Zeta potential of colloids in water and waste water. ASTM standard D 4187-82. American Society for Testing and Materials, West Conshohocken
Toshima N, Hara S (1995) Prog Polym Sci 20:155–183
Benseddik E, Makhlouski M, Bernede JC, Lefrant S, Pron A (1995) Syn Met 72:237–242
Dutta P, Biswas S, Ghosh M, De SK, Chatterjee S (2001) Synth Met 122:455–461
Tominaga Y, Asai S, Sumita M, Panero S, Scrosati B (2005) J Pow Sour 146:402–406
Deshmukh RR, Shetty AR (2008) J Appl Polym Sci 107:3707–3717
Pawde SM, Deshmukh K (2009) Polym Eng Sci 49:808–818
Deng L, Kim TJ, Hagg MB (2009) J Membr Sci 340:154–163
Hede PD, Bach P, Jensen AD (2009) Indust Eng Chem Res 48:1905–1913
Bogun M, Mikolajczyk T (2009) J Appl Polym Sci 114:3452–3457
Acknowledgments
Authors wish to thank the Norwegian Research Council for financial support to the work through CCERT project. Sincere thanks to Sondre Volden, Iva Kralowa, Signe Hakonsen, and Asal Amiri from Ugelstad Laboratory at Department of Chemical Engineering, Norwegian University of Science and Technology (NTNU), for their help in using various analytical instruments. The authors would further like to thank Dr. S. L. Kamath from the Indian Institute of Technology (IIT) Mumbai for SEM analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deshmukh, K., Ahmad, J. & Hägg, M.B. Fabrication and characterization of polymer blends consisting of cationic polyallylamine and anionic polyvinyl alcohol. Ionics 20, 957–967 (2014). https://doi.org/10.1007/s11581-013-1062-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-013-1062-3