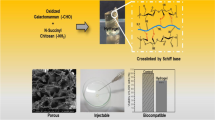
Summary
In order to investigate the strength, structure and cell cytocompatibility of injectable thermosensitive chitosan (CS)/poly(vinyl alcohol) (PVA) composite hydrogel, chitosan hydrochloride solution was transferred to a neutral pH and mixed with different proportions of PVA, then the gelation time and strength of these different hydrogels were tested and spatial structures were observed under a scanning electron microscopy (SEM) after freeze-drying. The cytocompatibility of the hydrogels was evaluated through cytotoxicity test and three-dimensional culture with bone marrow mesenchymal stem cells. The results showed that the CS/PVA solution kept in liquid state at low temperature (0–4°C) and turned into transparent elastomer about 15–20 min at 37°C. Gelation time was prolonged, the strength increased and porous structure became dense with the PVA content increased in the mixed hydrogel. The cytotoxicity grades of these gels were from 0 to 1. Rabbit bone marrow mesenchymal stem cells could survive and proliferate in the gel within 3 weeks, and the gel had good cytocompatibility. It was concluded that thermosensitive CS/PVA composite hydrogel not only has interpenetrating network structure and better mechanical strength, but also has good cytocompatibility, and may be used as an injectable scaffold for tissue engineering.
Similar content being viewed by others
References
Kim IY, Seo SJ, Moon HS, et al. Chitosan and its derivatives for tissue engineering applications. Biotechnol Adv, 2008,26(1):1–21
Kumar MN, Muzzarelli RA, Muzzarelli C, et al. Chitosan chemistry and pharmaceutical perspectives. Chem Rev, 2004,104(12):6017–6084
Khor E, Lim LY. Implantable applications of chitin and chitosan. Biomaterials, 2003,24(13):2339–2349
Sezer AD, Cevher E, Hatipoǧlu F, et al. Preparation of fucoidan-chitosan hydrogel and its application as burn healing accelerator on rabbits. Biol Pharm Bull, 2008,31(12):2326–2333
Gutowska A, Jeong B, Jasionowski M. Injectable gels for tissue engineering. Anat Rec, 2001,263(4):342–349
Shu XZ, Liu YC, Palumbo FS, et al. In situ crosslinkable hyaluronan hydrogels for tissue engineering. Biomaterials, 2004,25(7–8):1339–1348
Jeong B, Kim SW, Baeb YH. Thermosensitive sol-gel reversible hydrogels. Adv Drug Deliv Rev, 2002,54(1):37–51
Park J, Kim D. Release behavior of amoxicillin from glycol chitosan superporous hydrogels. J Biomater Sci Polym Ed, 2009,20(5–6):853–862
de Souza Costa-Júnior E, Pereira MM, Mansur HS. Properties and biocompatibility of chitosan films modified by blending with PVA and chemically crosslinked. J Mater Sci Mater Med, 2009,20(2):553–561
Mori Y, Tokura H, Yoshikawa M. Properties of hydrogels synthesized by freezing and thawing aqueous polyvinyl alcohol solutions and their applications. J Materials Sci, 1997,32(2):491–496
Kurkuri MD, Aminabhavi TM. Poly(vinyl alcohol) and poly(acrylic acid) sequential interpenetrating network pH-sensitive microspheres for the delivery of diclofenac sodium to the intestine. J Control Release, 2004,96(1):9–20
Park H, Kim D. Swelling and mechanical properties of glycolchitosan/poly(vinyl alcohol) IPN-type superporous hydrogels. J Biomed Mater Res A, 2006,78(4):662–667
Tang Y, Du Y, Li Y, et al. A thermosensitive chitosan/ poly(vinyl alcohol) hydrogel containing hydroxyapatite for protein delivery. J Biomed Mater Res A, 2009,91(4):953–963
Gong Z, Ding SS, Yin YJ, et al. Optimized design of hy drogels for tissue engineering. Hua Xue Gongye Yu Gong Cheng Jin Zhan (Chinese). 2008,27(11):1743–1748
Bureau of Technical Supervision. The People’s Republic of China National Standards: Biological evaluation of medical silicone materials test methods (GB/T 16175-1996)[S]. Beijing: China Standard Press, 1996:6,17–18
Kretlow JD, Klouda L, Mikos AG. Injectable matrices and scaffolds for drug delivery in tissue engineering. Adv Drug Deliv Rev, 2007,59(4–5):236–273
Balakrishnan B, Jayakrishnan A. Self-cross-linking biopolymers as injectable in situ forming biodegradable scaffolds. Biomaterials, 2005,26(18):3941–3951
Chenite A, Chaput C, Wang D, et al. Novel injectable neutral solutions of chitosan form biodegradable gels in situ. Biomaterials, 2000,21(21):2155–2161
Cui YL, Qi AD, Liu WG, et al. Biomimetic surface modification of poly(L-lactic acid) with chitosan and its effects on articular chondrocytes in vitro. Biomaterials, 2003,24(21):3859–3868
Donati I, Stredanska S, Silvestrini G, et al. The aggregation of pig articular chondrocyte and synthesis of extracellular matrix by a lactose-modified chitosan. Biomaterials, 2005,26(9):987–998
Hong Y, Song H, Gong Y, et al. Covalentlycrosslinked chitosan hydrogel: properties of in vitro degradation and chondrocyte encapsulation. Acta Biomaterialia, 2007,3(1): 23–31
Bhattarai N, Ramay HR, Gunn J, et al. PEG-grafted chitosan as an injectable thermosensitive hydrogel for sustained protein release. J Control Release, 2005,103(3):609–624
Drury JL, Mooney DJ. Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials, 2003,24(24):4337–4351
Tan H, Gong Y, Lao L, et al. Gelatin/chitosan/hyaluronan ternary complex scaffold containing basic fibroblast growth factor for cartilage tissue engineering. J Mater Sci Mater Med, 2007,18(10):1961–1968
Crompton KE, Goud JD, Bellamkonda RV, et al. Polylysine-functionalised thermoresponsive chitosan hydrogel for neural tissue engineering. Biomaterials,2007,28(3):441–449
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by grants from National Natural Sciences Foundation of China (No. 30770574) and Research Fund Project of Health Department of Hubei Province (No. JX3B21).
Rights and permissions
About this article
Cite this article
Qi, B., Yu, A., Zhu, S. et al. The preparation and cytocompatibility of injectable thermosensitive chitosan/poly(vinyl alcohol) hydrogel. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 30, 89–93 (2010). https://doi.org/10.1007/s11596-010-0116-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-010-0116-2