Summary
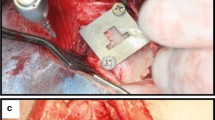
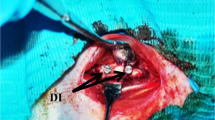
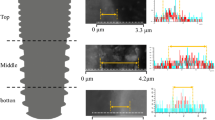
The clinical success of dental implants can be improved by achieving optimum implant properties, such as their biomechanical and surface characteristics. Nano-structured coatings can play an important role in improving the implant surface. The purpose of the present study was to determine the most appropriate conditions for electrophoretic deposition (EPD) of nano-zirconia coatings on Ti-6Al-7Nb substrates and to evaluate the structural and biomechanical characteristics of these deposited coatings on the dental implants. EPD was used with different applied voltages and time periods to obtain a uniform layer of nano-zirconia on Ti-6Al-7Nb samples. The coated samples were weighed and the thickness of the product layer was measured. Surface analysis was performed by using optical microscopical examination, scanning electron microscope and X-ray diffraction phase analysis. For in vivo examination, 48 screw-designed implants (24 uncoated and 24 nano-zirconia coated) were implanted in both tibiae of 12 white New Zealand rabbits and evaluated biomechanically after 4- and 12-week healing intervals. Results revealed that the use of different conditions for EPD affected the final coating film properties. Increasing the applied voltage and coating time period increased the deposited nano-zirconia film thickness and weight. By selecting the appropriate coating conditions, and analyzing scanning electron microscopical examination and XRD patterns, this technique could produce a thin and continuous nano-zirconia layer with a uniform thickness of the Ti-6Al-7Nb samples. Mechanically, the nano-zirconia-coated implants showed a highly statistically significant difference in removal torque values, while histologically these coated implants enhanced and promoted osseointegration after 4 and 12 weeks of healing, compared with the uncoated ones. In conclusion, EPD is an effective technique for providing a high quality nano-zirconia coating film on dental implant surfaces. Moreover, the osseointegration of these coated dental implants is improved compared with that of uncoated ones.
Similar content being viewed by others
References
Amarante ES, Lima LA. Optimization in surfaces of implants: titanium plasma and jetting with sand conditioned by acid-actual state. Implantodontia, 2001,15(2): 166–173
Guerra Neto CL. Avaliação da osseointegração de implantes de Ti Nitretados em Plasma. Natal: Universidade Federal do Rio Grande do Norte, Brazil, 2005.
Sä JC, de Brito RA, Moura CE, et al. Influence of argon-ion bombardment of titanium surfaces on the cell behavior. Surf Coat Technol, 2009,203(13):1765–1770
Turky RN, Jassim RK. The Electrophoretic Deposition of Nano A12O3 and AgNO3 on CpTi Dental Implant (An in vitro and in vivo study). J Bagh Coll Dent, 2016,28(1):41–47
Kelly JR, Denry I. Stabilized zirconia as a structural ceramic: an overview. Dent Mater, 2008,24(3):289–298
Liu X, Chu PK, Ding C. Surface nanofunctionalization of biomaterials. Mater Sei Eng R Rep, 2010,70(3):275–302
Ravichandran KS, An K, Dutton RE, et al. Thermal Conductivity of Plasma-Sprayed Monolithic and Multilayer Coatings of Alumina and Yttri-aStabilized Zirconia. J Am Ceram Soc, 2010,82(3):673–682
Manicone PF, Iommetti PR, Raffaelli L. An overview of zirconia ceramics: basic properties and clinical applications. J Dent, 2007,35(l1):819–826
Wang J, Yin W, He X, et al. Good Biocompatibility and Sintering Properties of Zirconia Nanoparticles Synthesized via Vapor-phase Hydrolysis. Sei Rep, 2016,6:35020
Thamaraiselvi T, Rajeswari S. Biological evaluation of bioceramic materials-a review. Carbon, 2004,24(31):172
Suzuki T, Fujibayashi S, Nakagawa Y, et al. Ability of zirconia double coated with titanium and hydroxyapatite to bond to bone under load-bearing conditions. Biomaterials, 2006,27(7):996–1002
Refaat MM, Hamad TI. Evaluation of Mechanical and Histological Significance of Nano Hydroxyapatite and Nano Zirconium Oxide Coating On the Osseointegration of CP Ti Implants. J Bagh Coll Dent, 2016,28(3):30–37
Calasans-Maia MD, Monteiro ML, Áscoli FO, et al. The rabbit as an animal model for experimental surgery. Acta Cir Bras, 2009,24(4):325–328
Muschler GF, Raut VP, Patterson TE, et al. The design and use of animal models for translational research in bone tissue engineering and regenerative medicine. Tissue Eng Part B Rev, 2010,16(1):123–145
Botticelli D, Lang NP. Dynamics of osseointegration in various human and animal models -a comparative analysis. Clin Oral Implants Res, 2017,28(6):742–748
Zhitomirsky I. Ceramic Films Using Cathodic Electro Deposition. JOM, 2000,52(1): 1–11
Ghiban B, Jicmon G, Cosmeleata G. Structural Investigation of Electrodeposited Hydroxyapatite on titanium supports. Rumanian J Physics, 2006,51:173–180
Mondragon-Cortez P, Vargas-Gutierrez G. Electrophoretic deposition of hydroxyapatite submicron particles at high voltages. Mater Lett, 2004,58:1336–1339
Meng X, Kwon T, Kim K. Hydroxyapatite coating by electrophoretic deposition at dynamic voltage. Dent Mater, 2008,27(5):666–671
Jassim R, Salman Y, Alzubaydi T. Studying the coating of alumina and hydroxy apatite on tapered dental implant (in vivo and in vitro study). Iraqi Dent J, 2016,38(1): 12–20
Hans JG. Thin film coatings for biomaterials and biomedical applications, 1st Edition. Woodhead Publishing, 2016,19–130
Tang Q, Brooks R, Rushton N, et al. Production and characterization of HA and SiHA coatings. J Mater Sci Mater Med, 2010,21(1):173–181
Zhang F, Chupas PJ, Lui SLA, et al. In situ Study of the Crystallization from Amorphous to Cubic Zirconium Oxide: Riveted and Reverse Monte Carlo Analyses. Chem Mater, 2007,19(13):3118–3126
Roshan GA, Arnoldi J, Procter P, et al. In vivo assessment of local effects after application of bone screws delivering bisphosphonates into a compromised cancellous bone site. Clin Biomech, 2011,26:1039–1043
Refaat MM, Hamad TI. Evaluation of Mechanical and Histological Significance of Nano Hydroxyapatite and Nano Zirconium Oxide Coating On the Osseointegration of CP Ti Implants. J Bagh Coll Dent, 2016,28(3):30–37
Park SH, Park KS, Cho SA. Comparison of removal torques of SLActive® implant and blasted, lasertreated titanium implant in rabbit tibia bone healed with concentrated growth factor application. J Adv Prosthodont, 2016,8(2):110–115
Azzawi ZG, Hamad TI, Kadhim SA. A Modified Laser Deposition Technique for Depositing Titania Nanoparticles on Commercially Pure Titanium Substrates (Mechanical Evaluation). IJIRSET, 2017,6(1):350–370
Mistry G, Shetty O, Shetty S, et al. Measuring implant stability: A review of different methods. J Dent Implant, 2014,4:165–169
Sennerby L, Thomsen P, Ericson LE. Early tissue response to titanium implants inserted in rabbit cortical bone. J Mater Sci Mater Med, 1993,4(3):240–250
Al-Habib SS. Evaluation of bone-Implant Interface of Commercially Pure Titanium and Ti-6Al-7Nb Alloy Dental Implants (Experimental Study in Rabbits). PhD thesis, College of Dentistry, University of Baghdad, 2005.
Al-Hejazee AY, Salim SA, Abdul-Hameed BA, et al. Histological evaluation of bone-implant interface associated with biomimetic coated commercially pure titanium and Ti-6Al-7Nb alloy implants. J Bagh Coll Dent, 2008,20(2):59–66
Alzubaydi TA, AlAmeer SS, Ismaeel T, et al. In vivo studies of the ceramic coated titanium alloy for enhanced osseointegration in dental applications. J Mater Sci Mater Med, 2009,20(Suppl 1):35
Albrektsson T. Direct bone anchorage of dental implants. J Prosth Dent, 1983,50:255–261
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hamad, T.I., Fatalla, A.A., Waheed, A.S. et al. Biomechanical Evaluation of Nano-Zirconia Coatings on Ti-6Al-7Nb Implant Screws in Rabbit Tibias. CURR MED SCI 38, 530–537 (2018). https://doi.org/10.1007/s11596-018-1911-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-018-1911-4