Abstract
Background
Gastric electrical stimulation synchronized to the refractory period of gastric electrical activity and applied during meals was evaluated for safety and for improvement of body weight and glycemic control in obese type 2 diabetes.
Methods
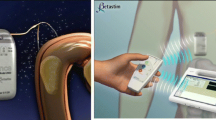
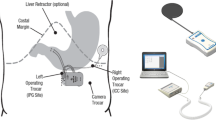
The study involved obese diabetic type 2 (ODM) patients in a multicenter open-label European feasibility trial. A total of 24 ODM (nine males, 15 females) treated with insulin and/or oral hyperglycemic agents and body mass index between 33.3 to 49.7 kg/m2 were implanted laparoscopically with a TANTALUS system.
Results
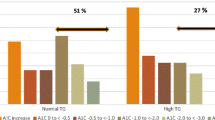
There were 18 adverse events related to the implant procedure or the device reported in 12 subjects. All were short lived and resolved with no sequelae. In the 21 subjects that reached the 1-year visit weight was reduced by 4.5 ± 2.7 kg (p < 0.05) and HbA1c by 0.5 ± 0.3% (p < 0.05). In a subgroup (n = 11) on stable or reduced oral medication, weight was reduced by 6.3 ± 3.4 kg (p < 0.05) and HbA1c by 0.9 ± 0.4% (p < 0.05). The group on insulin (n = 6) had no significant changes in weight and HbA1c.
Conclusions
The TANTALUS system is well tolerated in obese type 2 diabetic subjects. Gastric electrical stimulation can potentially improve glucose metabolism and induce weight loss in obese diabetic patients, who are not well controlled on oral antidiabetic therapy. Further evaluation is required to determine whether this effect is due to induced weight loss and/or to direct signal dependent mechanisms.




Similar content being viewed by others
Abbreviations
- EWL:
-
Excess weight loss
- TFEQ:
-
Three-Factor Eating Questionnaire
- BMI:
-
Body mass index
- GCM:
-
Gastric contractility modulation
- PP:
-
Postprandrial
- BT:
-
Bed time
- GE:
-
Gastric emptying
- FBG:
-
Fasting blood glucose
- BL:
-
Baseline
References
Engelgau MM, Geiss LS, Saaddine JB, et al. The evolving diabetes burden in the United States. Ann Intern Med. 2004;140:945–50.
Kopelman PG, Hitman GA. Diabetes: exploding type II. Lancet. 1998;352(Suppl. 4):5.
Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292:1724–37.
Moo TA, Rubino F. Gastrointestinal surgery as treatment for type 2 diabetes. Curr Opin Endocrinol Diabetes Obes. 2008;2:153–8.
Bohdjalian A, Prager G, Aviv R, et al. One-year experience with Tantalus: a new surgical approach to treat morbid obesity. Obes Surg. 2006;5:627–34.
Peles S, Petersen J, Aviv R, et al. Enhancement of antral contractions and vagal afferent signaling with synchronized electrical stimulation. Am J Physiol Gastrointest Liver Physiol. 2003;285:G577–85.
Sanmiguel CP, Aviv R, Policker S, et al. Association between gastric electromechanical activity and satiation in dogs. Obesity. 2007;15(12):2958–63.
Pocai A, Lam TK, Gutierrez-Juarez R, et al. Hypothalamic K(ATP) channels control hepatic glucose production. Nature. 2005;434:1026–31.
Libert C. Inflammation: a nervous connection. Nature. 2003;421:328–9.
Sanmiguel CP, Haddad W, Aviv R, et al. The TANTALUS trade mark system for obesity: effect on gastric emptying of solids and ghrelin plasma levels. Obes Surg. 2007;17:1503–9.
Neeley WE. Simply automated determination of serum or plasma glucose by a hexokinase/glucose-6-phosphate dehydrogenase method. Clin Chem. 1972;18:509–15.
Dunn PJ, Cole RA, Soeldner JS. Further development and automation of a high pressure liquid chromatography method for the determination of glycosylated hemoglobins. Metabolism. 1979;28:777–9.
Albert J, Stunkard AJ, Samuel M. The three factors eating questionnaire to measure dietary restraint, disinhibition and hunger. J Psychosom Res. 1985;29:71–83.
Lundberg GD. How to reduce laparoscopy-induced shoulder pain. Medscape J Med. 2008;10:173.
De Vries J, De Jongste MJ, Spincemaille G, Staal MJ. Spinal cord stimulation for ischemic heart disease and peripheral vascular disease. Adv Tech Stand Neurosurg. 2007;32:63–89.
Zhang J, Chen JD. Pacing the gut in motility disorders. Curr Treat Options Gastroenterol. 2006;9:351–60.
Policker S, Lu H, Haddad W, et al. Electrical stimulation of the gut for the treatment of type 2 diabetes: effect of automatic eating detection. J Diabetes Sci Technol. 2008;2:904–12.
Dockray GJ. The brain-gut axis. In: Yamada T, editor. Basic mechanisms of normal and abnormal gastrointestinal function. 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2003. p. 77–91.
Sobocki J, Thor PJ, Uson J, et al. Microchip vagal pacing reduces food intake and body mass. Hepatogastroenterology. 2001;48:1783–7.
Rocca AS, Brubaker PL. Role of the vagus nerve in mediating proximal nutrient-induced glucagon-like peptide-1 secretion. Endocrinology. 1999;140:1687–94.
Andrews PL, Sanger GJ. Abdominal vagal afferent neurones: an important target for the treatment of gastrointestinal dysfunction. Curr Opin Pharmacol. 2002;6:650–6.
Strader AD, Woods SC. Gastrointestinal hormones and food intake. Gastroenterology. 2005;128:175–91.
Liu S, Lei Y, Chen JD. Inhibitory effects and mechanisms of colonic electric stimulation on gastric and rectal tone in conscious dogs. Dis Colon Rectum. 2006;49:1749–54.
Acknowledgments
This work was supported by a research grant from MetaCure (USA) Inc, Orangeburg, NY. TANTALUS™ system is a registered trademark of MetaCure NV, Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bohdjalian, A., Prager, G., Rosak, C. et al. Improvement in Glycemic Control in Morbidly Obese Type 2 Diabetic Subjects by Gastric Stimulation. OBES SURG 19, 1221–1227 (2009). https://doi.org/10.1007/s11695-009-9901-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-009-9901-z