Abstract
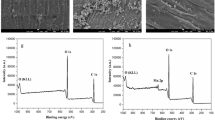
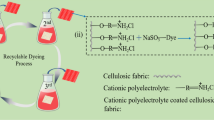
Pollution caused by organic dyes is of serious environmental and health concern to the population. Dyes are widely used in textile coloring applications. In the present work, cotton textile was coated with a conducting polymer, polypyrrole (PPy), in situ during the oxidative polymerization of pyrrole. The resulting materials were utilized as easily separated and recyclable adsorbent for the removal of methylene blue (MB) as a model of cationic dyes in alkaline solutions. It showed also some affinity to remove Acid Green 25 as an anionic dye in acidic medium. The adsorption was assessed by monitoring the decrease in dye concentration by UV–Visible absorption spectroscopy. The influence of various parameters such as initial dye concentration, contact time, pH, temperature, and adsorbent dose on the adsorption process was studied. The pseudo-second-order kinetic model and Freundlich isotherm model were found to describe the adsorption process. The thermodynamic study revealed that the adsorption of MB by PPy was feasible, spontaneous, and exothermic process. Investigation of the substrate regeneration revealed that PPy deposited on cotton textile can be reused for dye adsorption several times with good efficiency and it allows for the recovery of MB for recycling purposes.

















Similar content being viewed by others
References
Abramian L, El-Rassy H (2009) Adsorption kinetics and thermodynamics of azo-dye Orange II onto highly porous titania aerogel. Chem Eng J 150:403–410. https://doi.org/10.1016/j.cej.2009.01.019
Abdullah H, Kuo DH, Kuo YR, Yu FA, Cheng K-B (2016) Facile synthesis and recyclability of thin Nylon film-supported n-type ZnO/p-type Ag2O nano composite for visible light photocatalytic degradation of organic dye. J Phys Chem C 120:7144–7154. https://doi.org/10.1021/acs.jpcc.5b12153
Abrahart EN (1977) Dyes and their intermediates. Edward Arnold Ltd., London
Al-Anber MA (2011) Thermodynamics approach in the adsorption of heavy metals. In: Moreno-Pirajan JC (ed) Thermodynamics—interaction studies—solids, liquids and gases. InTech, Rijeka, Croatia, pp 737–764. https://doi.org/10.5772/21326
Alekseeva E, Bober P, Trchová M, Šeděnková I, Prokeš J, Stejskal J (2015) The composites of silver with globular or nanotubular polypyrrole: the control of silver content. Synth Met 209:105–111. https://doi.org/10.1016/j.synthmet.2015.07.003
Allen SJ, Mckay G, Porter JF (2004) Adsorption isotherm models for basic dye adsorption by peat in single and binary component systems. J Colloid Interface Sci 280:322–333. https://doi.org/10.1016/j.jcis.2004.08.078
Anirudhan T, Radhakrishnan P (2008) Thermodynamics and kinetics of adsorption of Cu (II) from aqueous solutions onto a new cation exchanger derived from tamarind fruit shell. J Chem Thermodyn 40:702–709. https://doi.org/10.1016/j.jct.2007.10.005
Ansari R, Mosayebzadeh Z (2010) Removal of basic dye methylene blue from aqueous solutions using sawdust and sawdust coated with polypyrrole. J Iran Chem Soc 7:339–350. https://doi.org/10.1007/BF03246019
Ayad MM, El-Nasr AA (2010) Adsorption of cationic dye (methylene blue) from water using polyaniline nanotubes base. J Phys Chem C 114:14377–14383. https://doi.org/10.1021/jp103780w
Ayad M, Zaghlol S (2012) Nanostructured crosslinked polyaniline with high surface area: synthesis, characterization and adsorption for organic dye. Chem Eng J 204:79–86. https://doi.org/10.1016/j.cej.2012.07.102
Ayad MM, El-Nasr AA, Stejskal J (2012) Kinetics and isotherm studies of methylene blue adsorption onto polyaniline nanotubes base/silica composite. J Ind Eng Chem 18:1964–1969. https://doi.org/10.1016/j.jiec.2012.05.012
Ayad M, El-Hefnawy G, Zaghlol S (2013) Facile synthesis of polyaniline nanoparticles; its adsorption behavior. Chem Eng J 217:460–465. https://doi.org/10.1016/j.cej.2012.11.099
Ayad M, Salahuddin N, Fayed A, Bastakoti BP, Suzuki N, Yamauchi Y (2014) Chemical design of a smart chitosan–polypyrrole–magnetite nanocomposite toward efficient water treatment. Phys Chem Chem Phys 16:21812–21819. https://doi.org/10.1039/c4cp03062a
Babu KF, Subramanian SS, Kulandainathan MA (2013) Functionalisation of fabrics with conducting polymer for tuning capacitance and fabrication of supercapacitor. Carbohydr Polym 94:487–495. https://doi.org/10.1016/j.carbpol.2013.01.021
Bajgar V, Penhaker M, Martinková L, Pavlovič A, Bober P, Trchová M, Stejskal J (2016) Cotton fabric coated with conducting polymers and its application in monitoring carnivorous plant response. Sensors 16:498. https://doi.org/10.3390/s16040498
Balathanigaimani M, Shim WG, Park KH, Lee JW, Moon H (2009) Effects of structural and surface energetic heterogeneity properties of novel corn grain-based activated carbons on dye adsorption. Microporous Mesoporous Mater 118:232–238. https://doi.org/10.1016/j.micromeso.2008.08.028
Ballav N, Debnath S, Pillay K, Maity A (2015) Efficient removal of Reactive Black from aqueous solution using polyaniline coated ligno-cellulose composite as a potential adsorbent. J Mol Liq 209:387–396. https://doi.org/10.1016/j.molliq.2015.05.051
Blinova NV, Stejskal J, Trchová M, Prokeš J, Omastová M (2007) Polyaniline and polypyrrole: a comparative study of the preparation. Eur Polym J 43:2331–2341. https://doi.org/10.1016/j.eurpolymj.2007.03.045
Bober P, Stejskal J, Šeděnková I, Trchová M, Martinková L, Marek J (2015) The deposition of globular polypyrrole and polypyrrole nanotubes on cotton textile. Appl Surf Sci 356:737–741. https://doi.org/10.1016/j.apsusc.2015.08.105
Bober P, Zasonska BA, Humpolíček P, Kuceková Z, Varga M, Horák D, Babayan V, Kazantseva N, Prokeš J, Stejskal J (2016) Polyaniline–maghemite based dispersion: electrical, magnetic properties and their cytotoxicity. Synth Met 214:23–29. https://doi.org/10.1016/j.synthmet.2016.01.010
Bulut Y, Aydın H (2006) A kinetics and thermodynamics study of methylene blue adsorption on wheat shells. Desalination 194:259–267. https://doi.org/10.1016/j.synthmet.2016.01.010
Chakrabarti S, Dutta BK (2005) On the adsorption and diffusion of methylene blue in glass fibers. J Colloid Interface Sci 286:807–811. https://doi.org/10.1016/j.jcis.2005.01.035
Chen KC, Wu JY, Liou DJ, Hwang SCJ (2003) Decolorization of the textile dyes by newly isolated bacterial strains. J Biotechnol 101:57–68. https://doi.org/10.1016/S0168-1656(02)00303-6
Chen J, Feng J, Yan W (2016) Influence of metal oxides on the adsorption characteristics of PPy/metal oxides for methylene blue. J Colloid Interface Sci 475:26–35. https://doi.org/10.1016/j.jcis.2016.04.017
Dave PN, Kaur S, Khosla E (2011) Removal of Eriochrome black-T by adsorption on to eucalyptus bark using green technology. Indian J Chem Technol 18:53–60 (WOS:000288733300007)
Debnath S, Ballav N, Maity A, Pillay K (2015) Development of a polyaniline-lignocellulose composite for optimal adsorption of Congo red. Int J Biol Macromol 75:199–209. https://doi.org/10.1016/j.ijbiomac.2015.01.011
Dubal DP, Patil SV, Kim WB, Lokhande CD (2011) Supercapacitors based on electrochemically deposited polypyrrole nanobricks. Mater Lett 65:2628–2631. https://doi.org/10.1016/j.matlet.2011.05.114
Ebrahimian A, Saberikhah E (2013) Biosorption of methylene blue onto Foumant tea waste: equilibrium and thermodynamic studies. Cell Chem Technol 47:657–666 (WOS: 000326418100017)
Ertaş M, Acemioğlu B, Alma MH, Usta M (2010) Removal of methylene blue from aqueous solution using cotton stalk, cotton waste and cotton dust. J Hazard Mater 183:421–427. https://doi.org/10.1016/j.jhazmat.2010.07.041
Fan L, Wei C, Xu Q, Xu J (2017) Polypyrrole-coated cotton fabrics used for removal of methylene blue from aqueous solution. J Textile Inst 108:1847–1852. https://doi.org/10.1080/00405000.2017.1296989
Feng J, Li J, Lv W, Xu H, Yang H, Yan W (2014) Synthesis of polypyrrole nano-fibers with hierarchical structure and its adsorption property of Acid Red G from aqueous solution. Synth Met 191:66–73. https://doi.org/10.1016/j.synthmet.2014.02.013
Gil A, Assis F, Albeniz S, Korili S (2011) Removal of dyes from wastewaters by adsorption on pillared clays. Chem Eng J 168:1032–1040. https://doi.org/10.1016/j.cej.2011.01.078
Jangid NK, Chauhan NPS, Punjabi PB (2014) Novel dye-substituted polyanilines: conducting and antimicrobial properties. Polym Bull 71:2611–2630. https://doi.org/10.1007/s00289-014-1210-6
Jangid NK, Chauhan NPS, Punjabi PB (2015) Preparation and characterization of polyanilines bearing rhodamine 6-G and Azure B as pendant groups. J Macromol Sci A 52:95–104. https://doi.org/10.1080/10601325.2015.980714
Jumasiah A, Chuah TG, Gimbon J, Choong TSY, Azni I (2005) Adsorption of basic dye onto palm kernel shell activated carbon: sorption equilibrium and kinetics studies. Desalination 186:57–64. https://doi.org/10.1016/j.desal.2005.05.015
Kamboh MA, Solangi IB, Sherazi S, Memon S (2011) A highly efficient calix-[4]-arene based resin for the removal of azo dyes. Desalination 268:83–89. https://doi.org/10.1016/j.desal.2010.10.001
Khan AR, Tahir H, Uddin F, Hameed U (2005) Adsorption of methylene blue from aqueous solution on the surface of wool fiber and cotton fiber. J Appl Sci Environ Manage 9:29–35. https://doi.org/10.4314/jasem.v9i2.17287
Khare SK, Panday KK, Srivastava RM, Singh VN (1987) Removal of victoria blue from aqueous solution by fly ash. J Chem Technol Biotechnol 38:99–104. https://doi.org/10.1002/jctb.280380206
Kopecká J, Kopecký D, Vrňata M, Fitl P, Stejskal J, Trchová M, Bober P, Morávková Z, Prokeš J, Sapurina I (2014) Polypyrrole nanotubes: mechanism of formation. RSC Adv 4:1551–1558. https://doi.org/10.1039/c3ra45841e
Kumar R, Ansari MO, Parveen N, Barakat MA, Cho MH (2015) Simple route for the generation of differently functionalized PVC@ graphene–polyaniline fiber bundles for the removal of Congo red from wastewater. RSC Adv 5:61486–61494. https://doi.org/10.1039/c5ra10378a
Langmuir I (1916) The constitution and fundemental properties of solids and liquids. Part I. Solids. J Am Chem Soc 38:2221–2295. https://doi.org/10.1021/ja02268a002
Lay M, Méndez JA, Pèlach MÀ, Bun KN, Vilaseca F (2016) Combined effect of carbon nanotubes and polypyrrole on the electrical properties of cellulose-nanopaper. Cellulose 23:3925–3937. https://doi.org/10.1007/s10570-016-1060-5
Li J, Feng J, Yan W (2013) Excellent adsorption and desorption characteristics of polypyrrole/TiO2 composite for methylene blue. Appl Surf Sci 279:400–408. https://doi.org/10.1016/j.apsusc.2013.04.127
Liu K, Li H, Wang Y, Gou X, Duan Y (2015a) Adsorption and removal of rhodamine B from aqueous solution by tannic acid functionalized graphene. Colloids Surf A Physicochem Eng Asp 477:35–41. https://doi.org/10.1016/j.colsurfa.2015.03.048
Liu W, Li X, Li M, Li Y, Ge M (2015b) Preparation of polyaniline/filter-paper composite for removal of Coomassie Brilliant Blue. Polym Polym Compos 23:191–198 (WOS:000353862600010)
Maráková N, Humpolíček P, Kašpárková V, Capáková Z, Martinková L, Bober P, Trchová M, Stejskal J (2017) Antimicrobial activity and cytotoxicity of cotton fabric coated with conducting polymers, polyaniline or polypyrrole, and with deposited silver nanoparticles. Appl Surf Sci 396:169–176. https://doi.org/10.1016/j.apsusc.2016.11.024
Matheickal J, Yu Q (1997) Biosorption of lead (II) from aqueous solutions by Phellinus badius. Miner Eng 10:947–957. https://doi.org/10.1016/S0892-6875(97)00076-9
McKay G (1979) Waste color removal from textile effluents. Am Dyestuff Rep 68:29–36 (WOS:A1979GT25600002)
Namasivayam C, Sumithra S (2005) Removal of Direct Red 12B and methylene blue from water by adsorption onto Fe(III)/Cr(III) hydroxide, an industrial solid waste. J Environ Manage 74:207–215. https://doi.org/10.1016/j.jenvman.2004.08.016
Nassar MY, Moustafa MM, Taha MM (2016) Hydrothermal tuning of the morphology and particle size of hydrozincite nanoparticles using different counterions to produce nanosized ZnO as an efficient adsorbent for textile dye removal. RSC Adv 6:42180–42195. https://doi.org/10.1039/c6ra04855b
Nassar MY, Ayman AA, Amin AS (2017a) A facile Pechini sol–gel synthesis of TiO2/Zn2TiO2/ZnO/C nanocomposite: an efficient catalyst for the photocatalytic degradation of Orange G textile dye. RSC Adv 7:30411–30421. https://doi.org/10.1039/c7ra90074k
Nassar MY, Mohamed TY, Ahmed IS, Samir I (2017b) MgO nanostructure via a sol–gel combustion synthesis method using different fuels: an efficient nano-adsorbent for the removal of some anionic textile dyes. J Mol Liq 225:730–740. https://doi.org/10.1016/j.molliq.2016.10.135
Nassar MY, Alia EI, Zakaria ES (2017c) Tunable auto-combustion preparation of TiO2 nanostructures as efficient adsorbents for the removal of an anionic textile dye. RSC Adv 7:8034–8050. https://doi.org/10.1039/c6ra27924d
Nassar MY, Mohamed TY, Ahmed IS, Ibrahim SA, Mohamed NM, Khatab M (2017d) Hydrothermally synthesized Co3O4, α-Fe2O3, and CoFe2O4 nanostructures: efficient nano-adsorbents for the removal of Orange G textile dye from aqueous media. J Inorg Organomet Polym 27:1526–1537. https://doi.org/10.1007/s10904-017-0613-x
Ofomaja AE, Ho YS (2007) Equilibrium sorption of anionic dye from aqueous solution by palm kernel fibre as sorbent. Dyes Pigm 74:60–66. https://doi.org/10.1016/j.dyepig.2006.01.014
Ovando Medina VM, Díaz Flores PE, Martínez Gutiérrez H, Moreno Ruiz LA, Antonio Carmona ID, Hernández Ordoñez M (2014) Composite of cellulosic agricultural waste coated with semiconducting polypyrrole as potential dye remover. Polym Compos 35:186–193. https://doi.org/10.1002/pc.22649
Palanisamy P, Agalya A, Sivakumar P (2013) Polypyrrole composite—a potential material for the removal of acid dyes. Asian J Chem 25:5891–5896 (WOS:000325089600002)
Riaz U, Ashraf S, Aqib M (2014) Microwave-assisted degradation of acid orange using a conjugated polymer, polyaniline, as catalyst. Arab J Chem 7:79–86. https://doi.org/10.1016/j.arabjc.2013.07.001
Rußler A, Sakakibara K, Rosenau T (2011) Cellulose as matrix component of conducting films. Cellulose 18:937–944. https://doi.org/10.1007/s10570-011-9555-6
Salavati H, Kohestani T (2013) Preparation, characterization and photochemical degradation of dyes under UV light irradiation by inorganic–organic nanocomposite. Mater Sci Semicond Process 16:1904–1911. https://doi.org/10.1016/j.mssp.2013.07.014
Sasso C, Bruyant N, Benevenri D, Faure-Vincent J, Zeno E, Petit-Conil M, Chaussy D, Belgacem MN (2011) Polypyrrole (PPy) chemical synthesis with xylan in aqueous medium and production of highly conducting PPy/nanofibrillated cellulose films and coatings. Cellulose 18:1455–1467. https://doi.org/10.1007/s10570-011-9583-2
Sharma YC (2009) Optimization of parameters for adsorption of methylene blue on a low-cost activated carbon. J Chem Eng Data 55:435–439. https://doi.org/10.1021/je900408s
Stejskal J, Trchová M, Bober P, Morávková Z, Kopecký D, Vrňata M, Prokeš J, Varga M, Watzlová E (2016) Polypyrrole salts and bases: superior conductivity of nanotubes and their stability towards the loss of conductivity by deprotonation. RSC Adv 6:88382–99391. https://doi.org/10.1039/c6ra19461c
Tan I, Ahmad A, Hameed B (2008) Adsorption of basic dye using activated carbon prepared from oil palm shell: batch and fixed bed studies. Desalination 225:13–28. https://doi.org/10.1016/j.desal.2007.07.005
Tavoli F, Alizadeh N (2014) In situ UV–vis spectroelectrochemical study of dye doped nanostructure polypyrrole as electrochromic film. J Electroanal Chem 720:128–133. https://doi.org/10.1016/j.jelechem.2014.03.022
Temkin M, Pyzhev V (1940) Kinetics of ammonia synthesis on promoted iron catalysts. Acta Physiochim URSS 12(3):217–222
Vadivelan V, Kumar KV (2005) Equilibrium, kinetics, mechanism, and process design for the sorption of methylene blue onto rice husk. J Colloid Interface Sci 286:90–100. https://doi.org/10.1016/j.jcis.2005.01.007
Walker G, Hansen L, Hanna J-A, Allen S (2003) Kinetics of a reactive dye adsorption onto dolomitic sorbents. Water Res 37:2081–2089. https://doi.org/10.1016/S0043-1354(02)00540-7
Wang S, Lu S, Li X, Zhang X, He S, He T (2013) Study of H2SO4 concentration on properties of H2SO4 doped polyaniline counter electrodes for dye-sensitized solar cells. J Power Sources 242:438–446. https://doi.org/10.1016/j.jpowsour.2013.05.060
Wang T et al (2014) Adsorption removal of organic dyes on covalent triazine framework (CTF). Microporous Mesoporous Mater 187:63–70. https://doi.org/10.1016/j.micromeso.2013.12.016
Wang N, Li J, Lv W, Feng J, Yan W (2015) Synthesis of polyaniline/TiO2 composite with excellent adsorption performance on Acid Red G. RSC Adv 5:21132–21141. https://doi.org/10.1039/c4ra16910g
Xiong J, Jiao C, Li C, Zhang D, Lin H, Chen Y (2014) A versatile amphiprotic cotton fiber for the removal of dyes and metal ions. Cellulose 21:3073–3087. https://doi.org/10.1007/s10570-014-0318-z
Yagub MT, Sen TK, Afroze S, Ang HM (2014) Dye and its removal from aqueous solution by adsorption: a review. Adv Colloid Interface Sci 209:172–184. https://doi.org/10.1016/j.cis.2014.04.002
Yang CX, Lei L, Zhou PX, Zhang Z, Lei ZQ (2015) Preparation and characterization of poly(AA co PVP)/PGS composite and its application for methylene blue adsorption. J Colloid Interface Sci 443:97–104. https://doi.org/10.1016/j.jcis.2014.11.040
Zhang S, Zhao L, Zeng M, Li J, Xu J, Wang X (2014) Hierarchical nanocomposites of polyaniline nanorods arrays on graphitic carbon nitride sheets with synergistic effect for photocatalysis. Catal Today 224:114–121. https://doi.org/10.1016/j.cattod.2013.12.008
Zhang Y, Yu F, Cheng W, Wang J, Ma J (2017) Adsorption equilibrium and kinetics of the removal of ammoniacal nitrogen by zeolite X/activated carbon composite synthesized from elutrilithe. J Chem 2017:1–9. https://doi.org/10.1155/2017/1936829
Zhu WJ, Lin L, Liao Q, Chen X, Qian ZQ, Shen JY, Liang JL, Yao JM (2016) Functionalization of cellulose with hyperbranched polyethylenimine for selective dye adsorption and separation. Cellulose 23:3785–3797. https://doi.org/10.1007/s10570-016-1045-4
Acknowledgements
The authors would like to thank Tanta University and the Technology Agency of the Czech Republic (TE01020022) for their support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ayad, M.M., Amer, W.A., Zaghlol, S. et al. Polypyrrole-coated cotton textile as adsorbent of methylene blue dye. Chem. Pap. 72, 1605–1618 (2018). https://doi.org/10.1007/s11696-018-0442-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-018-0442-6