Abstract
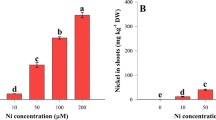
The influence of 50 and 100 μM Ni on the activities of nitrate reductase (NR), nitrite reductase (NiR), glutamine synthetase (GS), glutamate synthase (GOGAT), glutamate dehydrogenase (GDH), alanine aminotransferase (AlaAT) and aspartate aminotransferase (AspAT) was studied in the wheat roots. Root fresh weight, tissue Ni, nitrate, ammonium, glutamate and protein concentrations were also determined. Exposure to Ni resulted in a marked reduction in fresh weight of the roots accompanied by a rapid accumulation of Ni in these organs. Both nitrate and ammonium contents in the root tissue were considerably enhanced by Ni stress. While protein content was not significantly influenced by Ni application, glutamate concentration was slightly reduced on the first day after treatment with the higher Ni dose. Treatment of the wheat seedlings with 100 μM Ni led to a decrease in NR activity; however, it did not alter the activation state of this enzyme. Decline in NiR activity observed after application of 100 μM Ni was more pronounced than that in NR. The activities of GS and NADH-GOGAT also showed substantial decreases in response to Ni stress with the latter being more susceptible to this metal. Starting from the fourth day, both aminating and deaminating GDH activities in the roots of the seedlings supplemented with Ni were lower in comparison to the control. While the activity of AspAT remained unaltered after Ni application that of AlaAT showed a considerable enhancement. The results indicate that exposure of the wheat seedlings to Ni resulted in a general depression of nitrogen assimilation in the roots. Increase in the glutamate-producing activity of AlaAT may suggest its involvement in supplying the wheat roots with this amino acid under Ni stress.





Similar content being viewed by others
Abbreviations
- AlaAT:
-
Alanine aminotransferase
- AspAT:
-
Aspartate aminotransferase
- GDH:
-
Glutamate dehydrogenase
- GOGAT:
-
Glutamate synthase
- GS:
-
Glutamine synthetase
- NEA:
-
Naphthylenediamine dihydrochloride
- NiR:
-
Nitrite reductase
- NR:
-
Nitrate reductase
- PMSF:
-
Phenylmethylsulphonyl fluoride
- PVP:
-
Polyvinylpyrrolidone
- SA:
-
Sulfanilamide
References
Agbaria H, Heuer B, Zieslin N (1998) Rootstock-imposed alterations in nitrate reductase and glutamine synthetase activities in leaves of rose plants. Biol Plant 41:85–91
Atta-Aly MA (1999) Effect of nickel addition on the yield and quality of parsley leaves. Sci Hortic 82:9–24
Balestrasse KB, Gallego SM, Tomaro ML (2006) Oxidation of the enzymes involved in nitrogen assimilation plays an important role in the cadmium-induced toxicity in soybean plants. Plant Soil 284:187–194
Bhatia NP, Walsh KB, Baker AJM (2005) Detection and quantification of ligands involved in nickel detoxification in a herbaceous Ni hyperaccumulator Stackhousia tryonii Bailey. J Exp Bot 56:1343–1349
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brown PH, Welch RM, Madison JT (1990) Effect of nickel deficiency on soluble anion, amino acid, and nitrogen levels in barley. Plant Soil 125:19–27
Cataldo DA, Haroon M, Schrader LE, Youngs VL (1975) Rapid colorimetric determination of nitrate in plant tissue by nitration of salicylic acid. Commun Soil Sci Plant Anal 6:71–80
Chaffei C, Gouia H, Ghorbel MH (2003) Nitrogen metabolism in tomato plants under cadmium stress. J Plant Nutr 26:1617–1634
Chaffei C, Pageau K, Suzuki A, Gouia H, Ghorbel MH, Masclaux-Daubresse C (2004) Cadmium toxicity induced changes in nitrogen management in Lycopersicon esculentum leading to a metabolic safeguard through an amino acid storage strategy. Plant Cell Physiol 45:1681–1693
Debouba M, Gouia H, Suzuki A, Ghorbel MH (2006) NaCl stress effects on enzymes involved in nitrogen assimilation pathway in tomato “Lycopersicon esculentum” seedlings. J Plant Physiol 163:1247–1258
Dubois F, Tercé-Laforgue T, Gonzalez-Moro M-B, Estavillo J-M, Sangwan R, Gallais A, Hirel B (2003) Glutamate dehydrogenase in plants: is there a new story for an old enzyme? Plant Physiol Biochem 41:565–576
El-Shintinawy F, El-Ansary A (2000) Differential effect of Cd2+ and Ni2+ on amino acid metabolism in soybean seedlings. Biol Plant 43:79–84
Forde BG, Lea PJ (2007) Glutamate in plants: metabolism, regulation, and signaling. J Exp Bot 58:2339–2358
Freeman JL, Persans MW, Nieman K, Albrecht C, Peer W, Pickering IJ, Salt DE (2004) Increased glutathione biosynthesis plays a role in nickel tolerance in Thlaspi nickel hyperaccumulators. Plant Cell 16:2176–2191
Gajewska E, Skłodowska M (2008) Differential biochemical responses of wheat shoots and roots to nickel stress: antioxidative reactions and proline accumulation. Plant Growth Regul 54:179–188
Gajewska E, Słaba M, Andrzejewska R, Skłodowska M (2006) Nickel-induced inhibition of wheat root growth is related to H2O2 production, but not to lipid peroxidation. Plant Growth Regul 49:95–103
Gerendás J, Polacco JC, Freyermuth SK, Sattelmacher B (1999) Significance of nickel for plant growth and metabolism. J Plant Nutr Soil Sci 162:241–256
Gouia H, Ghorbal MH, Meyer C (2000) Effects of cadmium on activity of nitrate reductase and on other enzymes of the nitrate assimilation pathway in bean. Plant Physiol Biochem 38:629–638
Gouia H, Suzuki A, Brulfert J, Ghorbal MH (2003) Effects of cadmium on the co-ordination of nitrogen and carbon metabolism in bean seedlings. J Plant Physiol 160:367–376
Groat RG, Vance CP (1981) Root nodule enzymes of ammonia assimilation in alfalfa (Medicago sativa L.). Developmental patterns and response to applied nitrogen. Plant Physiol 67:1198–1203
Jha AB, Dubey RS (2004) Arsenic exposure alters activity behaviour of key nitrogen assimilatory enzymes in growing rice plants. Plant Growth Regul 43:259–268
Kevrešan S, Petrović N, Popović M, Kandrač J (1998) Effect of heavy metals on nitrate and protein metabolism in sugar beet. Biol Plant 41:235–240
Kwinta J, Bielawski W (1998) Glutamate dehydrogenase in higher plants. Acta Physiol Plant 20:453–463
Kwinta J, Koźlik D (2006) Glutamine synthetase and glutamate dehydrogenase in cadmium-stressed triticale seedlings. Acta Physiol Plant 28:339–347
Lam H-M, Coschigano KT, Oliveira IC, Melo-Oliveira R, Coruzzi GM (1996) The molecular-genetics of nitrogen assimilation into amino acids in higher plants. Annu Rev Plant Physiol Plant Mol Biol 47:569–593
Lightfoot DA, Mungur R, Ameziane R, Nolte S, Long L, Bernhard K, Colter A, Jones K, Iqbal MJ, Varsa E, Young B (2007) Improved drought tolerance of transgenic Zea mays plants that express the glutamate dehydrogenase gene (gdhA) of E. coli. Euphytica 156:103–116
Llamas A, Sanz A (2008) Organ-distinctive changes in respiration rates of rice plants under nickel stress. Plant Growth Regul 54:63–69
Llorens N, Arola L, Bladé C, Mas A (2000) Effects of copper exposure upon nitrogen metabolism in tissue cultured Vitis vinifera. Plant Sci 160:159–163
Matraszek R (2008) Nitrate reductase activity of two leafy vegetables as affected by nickel and different nitrogen forms. Acta Physiol Plant 30:361–370
Miflin BJ, Habash DZ (2002) The role of glutamine synthetase and glutamate dehydrogenase in nitrogen assimilation and possibilities for improvement in the nitrogen utilization of crops. J Exp Bot 53:979–987
Molins-Legua C, Meseguer-Lloret S, Moliner-Martinez Y, Campíns-Falcó P (2006) A guide for selecting the most appropriate method for ammonium determination in water analysis. Trends Anal Chem 25:282–290
Parida BK, Chhibba IM, Nayyar VK (2003) Influence of nickel-contaminated soils on fenugreek (Trigonella corniculata L.) growth and mineral composition. Sci Hortic 98:113–119
Robinson SA, Slade AP, Fox GG, Phillips R, Ratcliffe RG, Stewart GR (1991) The role of glutamate dehydrogenase in plant nitrogen metabolism. Plant Physiol 95:509–516
Ruiz JM, Rivero RM, Romero L (2007) Comparative effect of Al, Se, and Mo toxicity on NO3 − assimilation in sunflower (Helianthus annuus L.) plants. J Environ Manag 83:207–212
Samarakoon AB, Rauser WE (1979) Carbohydrate levels and photoassimilate export from leaves of Phaseolus vulgaris exposed to excess cobalt, nickel, and zinc. Plant Physiol 63:1165–1169
Sgherri C, Quartacci MF, Izzo R, Navari-Izzo F (2002) Relation between liponic acid and cell redox status in wheat grown in excess copper. Plant Physiol Biochem 40:591–597
Sharma P, Dubey RS (2005) Modulation of nitrate reductase activity in rice seedlings under aluminium toxicity and water stress: role of osmolytes as enzyme protectant. J Plant Physiol 162:854–864
Sousa CAF, Sodek L (2003) Alanine metabolism and alanine aminotransferase activity in soybean (Glycine max) during hypoxia of the root system and subsequent return to normoxia. Environ Exp Bot 50:1–8
Stitt M, Müller C, Matt P, Gibon Y, Carillo P, Morcuende R, Scheible W-R, Krapp A (2002) Steps towards an integrated view of nitrogen metabolism. J Exp Bot 53:959–970
Tripathy BC, Bhatia B, Mohanty P (1981) Inactivation of chloroplast photosynthetic electron-transport activity by Ni2+. Biochim Biophys Acta 638:217–224
Xiong Z-T, Liu C, Geng B (2006) Phytotoxic effects of copper on nitrogen metabolism and plant growth in Brassica pekinensis Rupr. Ecotoxicol Environ Saf 64:273–280
Yu CC, Hung KT, Kao CH (2005) Nitric oxide reduces Cu toxicity and Cu-induced NH4 + accumulation in rice leaves. J Plant Physiol 162:1319–1330
Acknowledgments
This work was partly supported by University of Łódź, Grants Nos 505/402 and 506/819.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Klobus.
Rights and permissions
About this article
Cite this article
Gajewska, E., Wielanek, M., Bergier, K. et al. Nickel-induced depression of nitrogen assimilation in wheat roots. Acta Physiol Plant 31, 1291–1300 (2009). https://doi.org/10.1007/s11738-009-0370-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-009-0370-8