Abstract
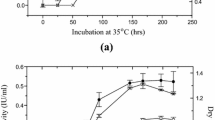
A fungal strain, marked as ECU0913, producing high activities of both cellulase and xylanase was newly isolated from soil sample collected near decaying straw and identified as Penicillium sp. based on internal transcribed spacer sequence homology. The cultivation of this fungus produced both cellulase (2.40 FPU/ml) and xylanase (241 IU/ml) on a stepwisely optimized medium at 30 °C for 144 h. The cellulase and xylanase from Penicillium sp. ECU0913 was stable at an ambient temperature with half-lives of 28 and 12 days, respectively. Addition of 3 M sorbitol greatly improved the thermostability of the two enzymes, with half-lives increased by 2.3 and 188-folds, respectively. Catalytic performance of the Penicillium cellulase and xylanase was evaluated by the hydrolysis of corn stover pretreated by steam explosion. With an enzyme dosage of 50 FPU/g dry substrate, the conversions of cellulose and hemicellulose reached 77.2% and 47.5%, respectively, without adding any accessory enzyme.






Similar content being viewed by others
References
Adsul, M. G., Bastawde, K. B., Varma, A. J., & Gokhale, D. V. (2007). Strain improvement of Penicillium janthinellum NCIM 1171 for increased cellulase production. Bioresource Technology, 98, 1467–1473.
Ahamed, A., & Vermette, P. (2008). Culture-based strategies to enhance cellulase enzyme production from Trichoderma reesei RUT-C30 in bioreactor culture conditions. Biochemical Engineering Journal, 40, 399–407.
Ahamed, A., & Vermette, P. (2008). Enhanced enzyme production from mixed cultures of Trichoderma reesei RUT-C30 and Aspergillus niger LMA grown as fed batch in a stirred tank bioreactor. Biochemical Engineering Journal, 42, 41–46.
Bailey, M. J., Biely, P., & Poutanen, K. (1992). Interlaboratory testing of methods for assay of xylanase activity. Journal of Biotechnology, 23, 257–270.
Berghem, L. E. R., & Pettersson, L. G. (1974). Mechanism of enzymatic cellulose degradation: isolation and some properties of a beta-glucosidase from Trichoderma viride. European Journal of Biochemistry, 46, 295–305.
Camassola, M., & Dillon, A. J. P. (2007). Production of cellulases and hemicellulases by Penicillium echinulatum grown on pretreated sugar cane bagasse and wheat bran in solid-state fermentation. Journal of Applied Microbiology, 103, 2196–2204.
Camassola, M., & Dillon, A. J. P. (2009). Biological pretreatment of sugar cane bagasse for the production of cellulases and xylanases by Penicillium echinulatum. Industrial Crops and Products, 29, 642–647.
Cobos, A., & Estrada, P. (2003). Effect of polyhydroxylic cosolvents on the thermostability and activity of xylanase from Trichoderma reesei QM 9414. Enzyme and Microbial Technology, 33, 810–818.
George, S. P., Ahmad, A., & Rao, M. B. (2001). A novel thermostable xylanase from Thermomonospora sp.: influence of additives on thermostability. Bioresource Technology, 78, 221–224.
Ghose, T. K. (1987). Measurement of cellulase activities. Pure and Applied Chemistry, 59, 257–268.
Heck, J. X., Hertz, P. F. M., & Ayub, A. Z. (2002). Cellulase and xylanase production by isolated amazon Bacillus strains using soybean industrial residue based solid-state cultivation. Brazilian Journal of Microbiology, 33, 213–218.
Ikeda, Y., Hayashi, H., Okuda, N., & Park, E. Y. (2007). Efficient Cellulase Production by the Filamentous Fungus Acremonium cellulolyticus. Biotechnology Progress, 23, 333–338.
Jørgensen, H., Mørkeberg, A., Krogh, K. B. R., & Olsson, L. (2005). Production of cellulases and hemicellulases by three Penicillium species: effect of substrate and evaluation of cellulase adsorption by capillary electrophoresis. Enzyme and Microbial Technology, 36, 42–48.
Kevin, A. G., Zhao, L. S., & Mark, E. (2006). Bioethanol Current Opinion in Chemical Biology, 10, 141–146.
Kovács, K., Megyeri, L., Szakacs, G., Kubicek, C. P., Galbe, M., & Zacchi, G. (2008). Trichoderma atroviride mutants with enhanced production of cellulase and β-glucosidase on pretreated willow. Enzyme and Microbial Technology, 43, 48–55.
Kovács, K., Szakacs, G., & Zacchi, G. (2009). Comparative enzymatic hydrolysis of pretreated spruce by supernatants, whole fermentation broths and washed mycelia of Trichoderma reesei and Trichoderma atroviride. Bioresource Technology, 100, 1350–1357.
Kristensen, J. B., Borjesson, J., Bruun, M. H., Tjerneld, F., & Jørgensen, H. (2007). Use of surface active additives in enzymatic hydrolysis of wheat straw lignocellulose. Enzyme and Microbial Technology, 40, 888–895.
Kumar, R., & Wyman, C. E. (2009). Effect of xylanase supplementation of cellulase on digestion of corn stover solids prepared by leading pretreatment technologies. Bioresource Technology, 100, 4203–4213.
Li, Y., Liu, Z., Cui, F., Xu, Y., & Zhao, H. (2007). Production of xylanase from a newly isolated Penicillium sp. ZH-30. World Journal of Microbiology & Biotechnology, 23, 837–843.
Lin, Y., & Tanaka, S. (2006). Ethanol fermentation from biomass resources: current state and prospects. Applied Microbiology and Biotechnology, 69, 627–642.
Liu, J., Yuan, X. Z., Zeng, G. M., Shi, J. G., & Chen, S. (2006). Effect of biosurfactant on cellulase and xylanase production by Trichoderma viride in solid substrate fermentation. Process Biochemistry, 41, 2347–2351.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the folin phenol reagent. The Journal of Biological Chemistry, 193, 265–275.
Malherbe, S., & Cloete, T. E. (2002). Lignocellulose biodegradation: Fundamentals and applications. Reviews in Environmental Science & Biotechnology, 1, 105–114.
Mandels, M., & Weber, J. (1969). The production of cellulases. Advances in Chemistry Series, 5, 391–414.
Martins, L. F., Kolling, D., Camassola, M., Dillon, A. J. P., & Ramos, L. P. (2008). Comparison of Penicillium echinulatum and Trichoderma reesei cellulases in relation to their activity against various cellulosic substrates. Bioresource Technology, 99, 1417–1424.
Miller, G. L. (1959). Use of dinitrosalicylic acid for determination of reducing sugar. Analytical Chemistry, 31, 426–430.
Mishra, C. S., Keskar, & Rao, M. (1984). Production and properties of extracellular endoxylanase from Neurospora crassa. Applied and Environmental Microbiology, 48, 224–228.
Sehnem, N. T., Bittencourt, L. R., Camassola, M., & Dillon, A. J. P. (2006). Cellulase production by Penicillium echinulatum on lactose. Applied Microbiology and Biotechnology, 72, 163–167.
Selig, M., Weiss, N., Ji, Y. (2008). Enzymatic Saccharification of Lignocellulosic Biomass. NREL Laboratory Analytical Procedure. http://www.eere.energy.gov/biomass/analyticalprocedures.html#LAP- 009.
Shah, A. R., & Madamwar, D. (2005). Xylanase production by a newly isolated Aspergillus foetidus strain and its characterization. Process Biochemistry, 40, 1763–1771.
Sun, Y., & Cheng, J. Y. (2002). Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresource Technology, 83, 1–11.
Väljamäe, P., Sild, V., Pettersson, G., & Johansson, G. (1998). The initial kinetics of hydrolysis by cellobiohydrolases Ι and ΙΙ is consistent with a cellulose surface-erosion model. European Journal of Biochemistry, 253, 469–475.
Vlasenko, E. Y., Ding, H., Labavitch, J. M., & Shoemaker, S. P. (1997). Enzymatic hydrolysis of pretreated rice straw. Bioresource Technology, 59, 109–119.
Vries, R. (2003). Regulation of Aspergillus genes encoding plant cell wall polysaccharide-degrading enzymes: relevance for industrial production. Applied Microbiology and Biotechnology, 61, 10–20.
Whitehurst, R. J., & Law, B. A. (2002). The nature of enzymes and their action in foods. Enzymes in Food Technology, 23, 1–18.
Zhang, J., Chu, D., Huang, J., Yu, Z., Dai, G., & Bao, J. (2010). Simultaneous saccharification and ethanol fermentation at high corn stover solids loading in a helical stirring bioreactor. Biotechnology and Bioengineering, 105, 718–728.
Acknowledgments
This research was financially supported by National Natural Science Foundation of China (grant nos. 20902023), National Special Fund for State Key Laboratory of Bioreactor Engineering (grant no. 2060204), Shanghai Leading Academic Discipline Project (No. B505), the Excellence Initiative of East China University of Science and Technology and Open Funding Project of the State Key Laboratory of Bioreactor Engineering.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shi, QQ., Sun, J., Yu, HL. et al. Catalytic Performance of Corn Stover Hydrolysis by a New Isolate Penicillium sp. ECU0913 Producing both Cellulase and Xylanase. Appl Biochem Biotechnol 164, 819–830 (2011). https://doi.org/10.1007/s12010-011-9176-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-011-9176-4