Abstract
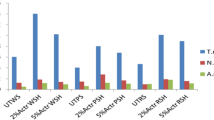
Cellulosic feedstocks for bioenergy differ in composition and processing requirements for efficient conversion to chemicals and fuels. This study discusses and compares the processing requirements for three lignocellulosic feedstocks—soybean hulls, wheat straw, and de-starched wheat bran. They were ground with a hammer mill to investigate how differences in composition and particle size affect the hydrolysis process. Enzyme hydrolysis was conducted using cellulase from Trichoderma reesei at 50°C and pH 5. Ground fractions were also subjected to dilute sulfuric acid treatment at 125°C, 15 psi for 30 min prior to cellulase treatment. Reducing particle size of biomass resulted in segregated components of feedstock. Grinding wheat straw to particle size <132 μm resulted in measured lignin content from 20% to ≈5% and reduced hemicellulose content. Reducing lignin content increased the effectiveness of enzyme hydrolysis of wheat straw. Particles sized <132 μm exhibited the highest soluble sugar release upon hydrolysis for all three feedstocks studied. Hemicellulose digestion improved with dilute sulfuric acid treatment with residual hemicellulose content <5% in all three feedstocks after acid treatment. This enhanced the cellulase action and resulted in approximately 1.6-fold increase in sugar availability in de-starched wheat bran and ≈1.5-fold for wheat straw and soybean hulls. Higher sugar availability in wheat bran after acid-mediated enzyme treatment correlated to higher ethanol yields during yeast fermentation compared with soybean hulls and wheat straw.





Similar content being viewed by others
References
McKendry P (2002) Energy production from biomass (Part 2): conversion technologies. Bioresour Technol 83:47–54
Chen Y, Sharma-Shivappa RR, Keshwani D, Chen C (2007) Potential of agricultural residue and hay for bio-ethanol production. Appl Biochem Biotechnol 142:276–290
Blasi DA, Titgemeyer EC, Drouillard J (2000) Soybean hull—composition and feeding value for beef and dairy cattle. Publication MF2438, Kansas State University, January 2000
Mandalari G, Craig B, Faulds AIS, Antonella S, Giuseppe B, Rosario L et al (2005) Fractionation and characterization of arabinoxylans from brewers’ spent grain and wheat bran. J Cereal Sci 42:205–212
Pan XJ, Sano Y (2005) Fractionation of wheat straw by atmospheric acetic acid process. Bioresour Technol 96:1256–1263
Li Y, Ruan R, Chen PL, Liu Z, Pan X, Lin X et al (2004) Enzymatic hydrolysis of corn stover pretreated by combined dilute alkaline treatment and homogenization. Trans ASAE 47:821–825
Moniruzzaman M (1996) Effect of steam explosion on the physicochemical properties and enzymatic saccharification of rice straw. Appl Biochem Biotechnol 59:283–297
Torget R, Walter P, Himmel M, Grohmann K (1991) Dilute acid pretreatment of corn residues and short rotation woody crops. Appl Biochem Biotechnol 28/29:75–85
Gharpuray MM, Lee YH, Fan LT (1983) Structural modification of lignocellulosics by pretreatments to enhance enzymatic hydrolysis. Biotechnol Bioeng 25:157–172
Sidiras D, Koukios E (2004) Simulation of acid-catalysed organosolv fractionation of wheat straw. Bioresour Technol 94:91–98
Mais U, Esteghlalian A, Saddler JN, Mansfield SD (2002) Enhancing the enzymatic hydrolysis of cellulosic materials using simultaneous ball milling. Appl Biochem Biotechnol 99:815–832
Lamsal B, Yoo J, Brijwani K, Alavi S (2010) Extrusion as a thermo-mechanical pre-treatment for lignocellulosic ethanol. Biomass Bioenergy 34:1703–1710
Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK (2010) Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuels 3:10. doi:10.1186/1754-6834-3-10
Sangseethong K, Meunier-Goddik L, Tantasucharit U, Liaw ET, Penner MH (1998) Rationale for particle size effect on rates of enzymatic saccharification of microcrystalline cellulose. J Food Biochem 22:321–330
Yeh AI, Huang YC, Chen SH (2010) Effect of particle size on the rate of enzymatic hydrolysis of cellulose. Carbohydr Polym 79:192–199
Miller GL (1959) Use of dinitrosalicylic reagent for determination of reducing sugars. Anal Chem 31:426–428
AOCS (2009) Official methods and recommended practices of the AOCS, 6th edn. American Oil Chemists’ Society, Urbana, IL
AACC International (1999) Approved methods of the AACC, 10th edn. American Association of Cereal Chemists, St. Paul, MN
Kristensen JB, Thygesen LG, Felby C, Jørgensen H, Elder T (2008) Cell-wall structural changes in wheat straw pretreated for bioethanol production. Biotechnol Biofuels 1:5. doi:10.1186/1754-6834-1-5
Csoka L, Lorincz A, Winkler A (2008) Sonochemically modified wheat straw for pulp and papermaking to increase its economical performance and reduce environmental issues. Bioresources 3:91–97
Mielenz JR, Bardsley JS, Wyman CE (2009) Fermentation of soybean hulls to ethanol while preserving protein value. Bioresour Technol 100(2009):3532–3539
Dasari RK, Berson RE (2007) The effect of particle size on hydrolysis reaction rates and rheological properties in cellulosic slurries. Appl Biochem Biotechnol 136–140:289–299
McMillan JD (1994) Pretreatment of lignocellulosic biomass. In: Himmel ME, Baker JO, Overend RP (eds) Enzymatic conversion of biomass for fuels production. American Chemical Society, Washington DC, pp 292–324
Qi BK, Chen XR, Wan YH (2010) Pretreatment of wheat straw by nonionic surfactant-assisted dilute acid for enhancing enzymatic hydrolysis and ethanol production. Bioresour Technol 101:4875–4883
Schell DJ, Farmer J, Newman M, McMillan JD (2003) Dilute sulfuric acid pretreatment of corn stover in pilot scale reactor: investigation of yields, kinetics, and enzyme digestabilities of solids. Appl Biochem Biotechnol 105:69–85
Digman MF, Shinners KJ, Muck RE, Dien BS (2010) Pilot-scale on-farm pretreatment of perennial grasses with dilute acid and alkali for fuel ethanol production. Trans ASABE 53:1007–1014
Hsu TC, Guo GL, Chen WH, Hwang WS (2010) Effect of dilute acid pretreatment of rice straw on structural properties and enzymatic hydrolysis. Bioresour Technol 101:4907–4913
Jensen JR, Morinelly JE, Gossen KR, Brodeur-Campbell MJ, Shonnard DR (2010) Effects of dilute acid pretreatment conditions on enzymatic hydrolysis monomer and oligomer sugar yields for aspen, balsam, and switchgrass. Bioresour Technol 101:2317–2325
Jeong TS, Um BH, Kim JS, Oh KK (2010) Optimizing dilute-acid pretreatment of rapeseed straw for extraction of hemicellulose. Appl Biochem Biotechnol 161:22–33
Li CL, Knierim B, Manisseri C, Manisseri C, Arora R, Scheller HV et al (2010) Comparison of dilute acid and ionic liquid pretreatment of switchgrass: biomass recalcitrance, delignification and enzymatic saccharification. Bioresour Technol 101:4900–4906
Wei GY, Gao W, Jin IH, Yoo SY, Lee JH, Chung CH et al (2009) Pretreatment and saccharification of rice hulls for the production of fermentable sugars. Biotechnol Bioprocess Eng 14:828–834
Yang Y, Sharma-Shivappa RR, Burns JC, Cheng J (2009) Saccharification and fermentation of dilute-acid-pretreated freeze-dried switchgrass. Energy Fuels 23:5626–5635
Acknowledgments
This project was supported by the Joint KSU/Specialty Enzymes Project at Grain Science and Industry Department. We thank Specialty Enzymes and Biochemicals, Inc., Chino, CA, for its generous contribution of the enzymes used in the study. We thank Dr. Donghai Wang and his laboratory at KSU Biological and Agricultural Engineering Department for ethanol fermentation runs on our treated samples. We also appreciate Khushal Brijwani, graduate student at KSU Grain Science and Industry for his initial help in manuscript preparation. This manuscript is Kansas Agricultural Experiment Station manuscript No. 08-356-J. The Iowa State University Agricultural Experiment Station supported senior author's salary during manuscript revision and editing.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lamsal, B.P., Madl, R. & Tsakpunidis, K. Comparison of Feedstock Pretreatment Performance and Its Effect on Soluble Sugar Availability. Bioenerg. Res. 4, 193–200 (2011). https://doi.org/10.1007/s12155-010-9112-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-010-9112-4