Abstract
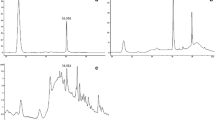
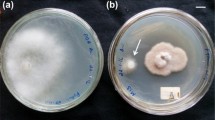
Taxol is a highly functionalized anticancer drug widely used in hospitals and clinics. The leaf spot fungus, Phyllosticta dioscoreae was isolated from diseased leaves of Hibiscus rosa-sinensis and screened for extracellular production of taxol in M1D (Modified liquid medium) and PDB (Potato dextrose broth) medium for the first time. The fungus was identified by its morphological and conidial features in the culture growth. The presence of taxol in the fungal culture filtrate was confirmed by different spectroscopic and chromatographic analyses. The amount of taxol produced was quantified by HPLC. The maximum amount of taxol produced was found to be 298 μg/L in M1D medium. Production rate was 5.96 × 103 times faster than that found in culture broth of earlier reported fungus, Taxomyces andreanae. The extracted fungal taxol also showed strong cytotoxic activity in vitro in the cultures of human cancer cells tested by apoptotic assay. The results indicate that P. dioscoreae is an excellent source of taxol production, which suggests that the fungus has potential to undergo genetic engineering in order to improve its production level.
Similar content being viewed by others
References
Wani, M. C., H. L. Taylor, M. E. Wall, P. Coggon, and A. T. McPhail (1971) Plant antitumor agents. VI. Isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J. Am. Chem. Soc. 93: 2325–2327.
Horwitz, S. B. (1992) Mechanism of action of taxol. Trends Pharmacol. Sci. 13: 134–136.
Nicolaou, K. C., Z. Yang, J. J. Liu, H. Ueno, P. G. Nantermet, R. K. Guy, C. F. Claiborne, J. Renaud, E. A. Couladouros, K. Paulvannan, and E. J. Sorensen (1994) Total synthesis of taxol. Nature 367: 630–634.
Jennewein, S. and R. Croteau (2001) Taxol: biosynthesis, molecular genetics, and biotechnological applications. Appl. Microbiol. Biotechnol. 57: 13–19.
Stierle, A., G. A. Strobel, and D. Stierle (1993) Taxol and taxane production by Taxomyces andreanae, an endophytic fungus of Pacific yew. Science 260: 214–216.
Strobel, G. A., X. Yang, J. Sears, R. Kramer, R. S. Sidhu, and W. M. Hess (1996) Taxol from Pestalotiopsis microspora, an endophytic fungus of Taxus wallachiana. Microbiology 142: 435–440.
Strobel, G. A., W. M. Hess, E. Ford, R. S. Sidhu, and X. Yang (1996) Taxol from fungal endophytes and the issue of biodiversity. J. Ind. Mircobiol. 17: 417–423.
Strobel, G. A. (2003) Endophytes as sources of bioactive products. Microbes Infect. 5: 535–544.
Ji, Y., J. N. Bi, B. Yan, and X. D. Zhu (2006) Taxolproducing fungi: a new approach to industrial production of taxol. Chin. J. Biotechnol. 22: 1–6.
Van der Aa, H. A. (1973) Studies in Phyllosticta I. Stud. Mycol. 5: 1–110.
Pinkerton, F. and G. A. Strobel (1976) Serinol as an activator of toxin production in attenuated cultures of Helminthosporium sacchari. Proc. Natl. Acad. Sci. USA 73: 4007–4011.
Cardellina, J. H. (1991) HPLC separation of taxol and cephalomannine. J. Liq. Chromatogr. 14: 659–665.
Ruckdeschel, K., A. Roggenkamp, V. Lafont, P. Mangeat, J. Heesemann, and B. Rouot (1997) Interaction of Yersinia enterocolitica with macrophages leads to macrophage cell death through apoptosis. Infect. Immun. 65: 4813–4821.
Li, J. Y., G. A. Strobel, R. Sidhu, W. M. Hess, and E. J. Ford (1996) Endophytic taxol-producing fungi from bald cypress, Taxodium distichum. Microbiology 142: 2223–2226.
Strobel, G. A., W. M. Hess, J. Y. Li, E. Ford, J. Sears, R. S. Sidhu, and B. Summerell (1997) Pestalotiopsis guepinii, a taxol-producing endophyte of the Wollemi pine, Wollemia nobilis. Aust. J. Bot. 45: 1073–1082.
McClure, T. D., K. H. Schram, and M. L. J. Reimer (1992) The mass spectrometry of taxol. J. Am. Soc. Mass Spectrom. 3: 672–679.
Chmurny, G. N., B. D. Hilton, S. Brobst, S. A. Look, K. M. Witherup, and J. A. Beutler (1992) 1H- and 13CNMR assignments for taxol, 7-epi-taxol, and cephalomannine. J. Nat. Prod. 55: 414–423.
Strobel, G. A., A. Stierle, D. Stierle, and W. M. Hess (1993) Taxomyces andreanae, a proposed new taxon for a bulbilliferous hyphomycete associated with Pacific yew. Mycotaxon 47: 71–78.
Li, J. Y., R. S. Sidhu, A. Bollon, and G. A. Strobel (1998) Stimulation of taxol production in liquid cultures of Pestalotiopsis microspora. Mycol. Res. 102: 461–464.
Li, J. Y., R. S. Sidhu, E. J. Ford, D. M. Long, W. M. Hess, and G. A. Strobel (1998) The induction of taxol production in the endophytic fungus-Periconia sp. from Torreya grandifolia. J. Ind. Microbiol. Biotechnol. 20: 259–264.
Kim, S. U., G. A. Strobel, and E. Ford (1999) Screening of taxol-producing endophytic fungi from Ginkgo biloba and Taxus cuspidate in Korea. Agric. Chem. Biotechnol. 42: 97–99.
Noh, M. J., J. G. Yang, K. S. Kim, Y. M. Yoon, K. A. Kang, H. Y. Han, S. B. Shim, and H. J. Park (1999) Isolation of a novel microorganism, Pestalotia heterocornis, producing paclitaxel. Biotechnol. Bioeng. 64: 620–623.
Guo, B. H., Y. C. Wang, X. W. Zhou, K. Hu, F. Tan, Z. Q. Miao, and K. X. Tang (2006) An endophytic taxolproducing fungus BT2 isolated from Taxus chinensis var. mairei. Afr. J. Biotechnol. 5: 875–877.
Kumaran, R. S., J. Muthumary, and B. K. Hur (2008) Taxol from Phyllosticta citricarpa, a leaf spot fungus of the angiosperm Citrus medica. J. Biosci. Bioeng. 106: 103–106.
Kumaran, R. S., J. Muthumary, and B. K. Hur (2008) Production of taxol from Phyllosticta spinarum, an endophytic fungus of Cupressus sp. Eng. Life Sci. 8: 438–446.
Carroll, G. C. (1988) Fungal endophytes in stems and leaves: from latent pathogen to mutualistic symbiont. Ecology 69: 2–9.
Tan, R. X. and W. X. Zou (2001) Endophytes: a rich source of functional metabolites. Nat. Prod. Rep. 18: 448–459.
Yeung, T. K., C. Germond, X. Chen, and Z. Wang (1999) The mode of action of taxol: Apoptosis at low concentration and necrosis at high concentration. Biochem. Biophys. Res. Commun. 263: 398–404.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumaran, R.S., Muthumary, J., Kim, EK. et al. Production of taxol from Phyllosticta dioscoreae, a leaf spot fungus isolated from Hibiscus rosa-sinensis . Biotechnol Bioproc E 14, 76–83 (2009). https://doi.org/10.1007/s12257-008-0041-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-008-0041-4