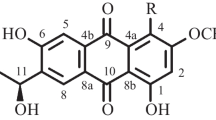
Abstract
The marine fungus Aspergillus versicolor was isolated from the inner tissue of the Red Sea green alga Halimeda opuntia. The fungus was identified by its morphology and 18s rDNA. Cultivation of this fungal strain led to a new metabolite named isorhodoptilometrin-1-methyl ether (1) along with the known compounds emodin (2), 1-methyl emodin (3), evariquinone (4), 7-hydroxyemodin 6,8-methyl ether (5), siderin (6), arugosin C (7), and variculanol (8). The structures were elucidated on the basis of NMR spectroscopic analysis and mass spectrometry. The biological properties of ethyl acetate extract and compounds 1–3 and 6–8 were explored for antimicrobial activity, anti-cancer activity and inhibition of Hepatitis C virus (HCV) protease.
Similar content being viewed by others
References
Ayer, W. A. and Trifonov, L. S., Anthraquinones and a 10-hydroxyanthrone from Phialophora alba. J. Nat. Prod., 57, 317–319 (1994).
Bacon, C. W. and White, J. F., Jr. (eds): Microbial Endophytes. Marcel Dekker, Inc., New York, Basel, p. 487, (2000).
Ballantine, J. A., Ferrito, V., Hassall, C. H., and Jenkins, M. L., The Biosynthesis of Phenols. Part XXIV. Arugosin C, a Metabolite of a Mutant Strain of Aspergillus rugulosus. J. Chem. Soc., 17, 1825–1830 (1973).
Belofsky, G. N., Jensen, P. R., Renner, M. K., and Fenical, W., New cytotoxic sesquiterpenoid nitrobenzoyl esters from a marine isolate of the fungus Aspergillus versicolor. Tetrahedron, 54, 1715–1724 (1998).
Bringmann, G., Lang, G., Steffens, S., Gunther, E., and Schaumann, K., Evariquinone, isoemericellin, and stromemycin from a sponge derived strain of the fungus Emericella variecolor. Phytochemistry, 63, 437–443 (2003).
Chomczynski, P. and Sacchi, N., Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem., 162, 156–159 (1987).
Cohen, P. A. and Towers, G. H. N., The anthraquinones of Heterodermia obscurata. Phytochemistry, 40, 911–915 (1995).
Fremlin, L. J., Piggott, A. M., Lacey, E., and Capon, R. J., Cottoquinazoline A and cotteslosins A and B, metabolites from an Australian marine-derived strain of Aspergillus versicolor. J. Nat. Prod., 72, 666–670 (2009).
Kumeda, Y. and Asao, T., Single-strand conformation polymorphism analysis of PCR-amplified ribosomal DNA internal transcribed spacers to differentiate species of Aspergillus section Flavi. Appl. Environ. Microbiol., 62, 2947–2952 (1996).
Lee, Y. M., Mansoor, T. A., Hong, J., Lee, C. O., Bae, K. S., and Jung, J. H., Polyketides from a sponge-derived fungus, Aspergillus versicolor. Nat. Prod. Sci., 13, 90–96 (2007).
Lee, Y. M., Li, H., Hong, J., Cho, H. Y., Bae, K. S., Kim, M. A., Kim, D.-K., and Jung, J. H., Bioactive metabolites from the sponge-derived fungus Aspergillus versicolor. Arch. Pharm. Res., 33, 231–235 (2010a).
Lee, Y. M., Dang, H., Hong, J., Lee, C., Bae, K. S., Kim, D. K., and Jung, J. H., A cytotoxic lipopeptide from the spongederived fungus Aspergillus versicolor. Bull. Korean Chem. Soc., 31, 205–208 (2010b).
Li, G. Y., Li, L. M., Yang, T., Chen, X. Z., Fang, D. M., and Zhang, G. L., Four new alkaloids, brevianamides O-R, from the fungus Aspergillus versicolor. Helv. Chim. Acta, 93, 2075–2080 (2010).
Lin, W., Brauers, G., Ebel, R., Wray, V., Sudarsono, B. A., and Proksch, P., Novel chromone derivatives from the fungus Aspergillus versicolor isolated from the marine sponge Xestospongia exigua. J. Nat. Prod., 66, 57–61 (2003).
Love, R. L., Parge, H. E., Wickersham, J. A., Hoastomsky, Z., Habiuka, N., Moomaw, E. W., Adachi, T., and Hostomska, Z., The crystal structure of hepatitis C virus NS3 protease reveals a trypsin-like fold and a structural zinc binding site. Cell, 87, 331–342 (1996).
Pelezer, M. J., Chan, E. C. S., and Krieg, N. R., Microbiology, 5th ed, McGraw-Hill, New York, p. 536, (1986).
Schmidt, H. W. and Moyer, A. J., Penicillin: I. Methods of assay. J. Bacteriology, 47, 199–209 (1944).
Singh, S. B., Reamer, R. A., Zink, D., Schmatz, D., Dombrowski, A., and Goetz, M. A., Variculanol: structure and absolute stereochemistry of a novel 5/12/5 tricyclic sesterterpenoid from Aspergillus variecolor. J. Org. Chem., 56, 5618–5622 (1991)
Strobel, G. A., Endophytes as sources of bioactive products. Microbes Infect., 5, 535–544 (2003).
Thomson, R. H., Naturally Occurring Quinones, III, Chap man and Hall, London, New York (1987).
Valeriote, F., Grieshaber, C. K., Media, J., Pietraszkiewicz, H., Hoffmann, J., Pan, M., and McLaughlin, S., Discovery and development of anticancer agents from plants. J. Exp. Ther. Oncol., 2, 228–236 (2002).
Wang, G. Y., Laidlaw, R. D., Marshall, J., and Keasling, J. D., Metabolic engineering of fungal secondary metabolic pathways. In: An, Z. Q. (ed.). Handbook of industrial mycology. Marcel Dekker, New York, p. 10016 (2003).
Wei, Y., Ma, C. M., and Hattori, M., Synthesis of dammaranetype triterpene derivatives and their ability to inhibit HIV and HCV proteases. Bioorg. Med. Chem., 17, 3003–3010 (2009).
Yurchenko, A. N., Smetanina, O. F., Kalinovsky, A. I., Pivkin, M. V., Dmitrenok, P. S., and Kuznetsova, T. A., A new meroterpenoid from the marine fungus Aspergillus versicolor (Vuill.) Tirab. Russ. Chem. Bull., 59, 852–856 (2010).
Zhuang, Y., Teng, X., Wang, Y., Liu, P., Li, G., and Zhu, W., New quinazolinone alkaloids within rare amino acid residue from coral-associated fungus, Aspergillus versicolor LCJ-5-4. Org. Lett., 13, 1130–1133 (2011).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hawas, U.W., El-Beih, A.A. & El-Halawany, A.M. Bioactive anthraquinones from endophytic fungus Aspergillus versicolor isolated from red sea algae. Arch. Pharm. Res. 35, 1749–1756 (2012). https://doi.org/10.1007/s12272-012-1006-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-012-1006-x