Abstract
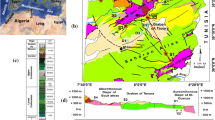
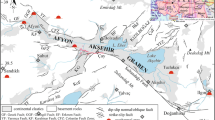
This paper deals with the results of a geothermal study on the thermal waters of Northeast Tunisia, which has remarkable thermal springs that have been used for bathing since many years. Water temperature at the emergency varies between 18 and 57 °C and total dissolved solids (TDS) range from 1,742 to 18,542 mg/l. Hydrochemical approach recognized three different water types: (1) the poorly mineralized Ca–HCO3–SO4 2+ type characterizing the carbonate aquifer, (2) the mixed type influenced by the evaporate rocks, and (3) the highly mineralized Na–Cl– type representing the deep thermal waters. Furthermore, the estimated temperatures at depth, using various chemical geothermometers, range between 60 and 110 °C. A synthetic hydrogeothermal approach, applied on the study area, including hydrogeological, geological, hydrochemical, and geothermometrical data shows that the thermal water of Northeast Tunisia is of meteoric origin. This water infiltrates deeply into the Jurassic dolomite and reaches a high temperature before rising upwards to supply the hot springs and well discharge zones. The upward movement of hot water from the deep aquifers to the surface is essentially due to the tectonic disharmonies which affected the area.










Similar content being viewed by others
References
Arnorsson S (1983) Chemical equilibria in icelandic geothermal systems—Implications for chemical geothermometry in investigations. Geothermics 12:119–128A
Ben Dhia H (1983) Les provinces géothermiuques de la Tunisie méridionale. Thèse de Doctorat Es-sciences naturelles. Univ. Bordeau I. 196 et 6 annexes
Ben Dhia H (1987) The geothermal gradient map of central Tunisia, comparison with structural, gravimetric and petroleum data. Tectonophysics 142:99–109
Bouri S (1994) Hydrothermie de surface et potentialites hydrogéothermiques du Nord tunisien. Thèse de 3eme cycle, ENI-Sfax, Tunisia
Bouri S, Ben Abdallah I, Zarhloule Y, Ben Dhia H (2007) Thermal regime, groundwater flow and petroleum occurrences in the Cap Bon region, northeastern Tunisia. Geothermics 36(4):362–381
Bouri S, Makni J, Ben Dhia H (2008) A synthetic approach integrating surface and subsurface data for prospecting deep aquifers: The Southeast Tunisia. Environ Geol 54:1473–1484
Brahim FB, Makni J, Bouri S, Dhia HB (2012) Properties of geothermal resources in Kebilli region, Southwestern Tunisia. Environ Earth Sci. doi:10.1007/s12665-012-2018-z
Burollet PF (1978) Mouvements quaternaires et récents aux îles Kerkennah (Tunisie orientale). Compte rendu de l ‘Académie des sciences 286:1133–1135
Calmbach L (1995) HYDROWIN computer programme. Version 3.0. Institut de Mine’ralogie BFSH 2, 1015 Lausanne
Can I (2002) A new improved Na/K geothermometer by artificial neural networks. Geothermics 31:751–760
Castany G (1955) Les extrusions jurassique en Tunisie. Ann. Min. et Géo., Tunis, n 14, 71 pp
D’Amore F, Fancelli R, Caboi R (1987) Observations on the application of chemical geothermometers to some hydrothermal systems in Sardinia. Geothermics 16(3):271–282
D’Amore F, Scandifio G, Panichi C (1983) Some observation on the chemical classification of ground waters. Geothermics 12(2/3):141–148
Durov SA (1948) Classification of natural waters and graphic representation of their composition. Dokl Akad Nauk SSSR 59:87–90
Fleury J (2005) PCA-CHEM new code using Matlab script for interpretative chemistry and microbiology data. Matlab pour les nuls no5
Fouillac C, Michard G (1977) Sodium potassium calcium relationship in hot springs of Massif Central. In: Proc. of the second int. symposium on water-rock interaction, Strasbourg, vol 3, pp 109–113
Fournier RO (1977) Chemical geothermometers and mixing models for geothermal systems. Geothermics 5(1/4):41–50
Fournier RO, Potter RW (1979) Magnesium correction to the Na–K–Ca chemical geothermometer. Geochim Cosmochim Acta 43:1543–1550
Fournier RO, Truesdell AH (1973) An empirical Na-K-Ca geothermometer for natural waters. Geochim Cosmochim Acta 37:1255–1275
Giggenbach WF (1986) Graphical techniques for the evaluation of water-rock equilibration condition by use of Na, K, Mg and Ca contents of discharge waters. In: Proceedings of 8th N.Z. geothermal workshop, pp 37–44. Labo. geology, ENS, Paris, ERAG on CNRS
Giggenbach WF (1988) Geothermal solute equilibria. Derivation of Na–K–Mg–Ca geoindicators. Geochim Cosmochim Acta 52:2749–2765
Giggenbach WF, Sheppard DS, Robinson BW, Stewart MK, Lyon GL (1994) Geochemical structure and position of the Waiotapu geothermal field, New Zealand. Geothermics 23:599–644
Jallouli A (2002) La Tunisie thermale, Office du thermalisme, 193 pp
Khomsi S, Ghazi Ben Jemia M, Frizon de Lamotte D, Maherssi C, Echihi O, Mezni R (2009) An overview of the Late Cretaceous–Eocene positive inversions and Oligo-Miocene subsidence events in the foreland of the Tunisian Atlas: Structural style and implications for the tectonic agenda of the Maghrebian Atlas system. Tectonophysics 475(1):38–58
Langelier W, Ludwig H (1942) Graphical methods for indicating the mineral character of natural waters. J Am Water Assoc 34:335–352
Lloyd WJ, Heathcotte JA (1985) Natural inorganic hydrochemistry in relation to groundwater. Clarendon, Oxford
Makni J, Bouri S, Ben Dhia H (2013) Hydrochemistry and geothermometry of thermal groundwater of southeastern Tunisia (Gabes region). Arab J Geosci. doi:10.1007/s12517-011-0510-5
Mamou A (1990) Caractérisation et évaluation des ressources en eau du Sud tunisien. Thèse 400 de doctorat d’état es sciences naturelles. Univ. Paris-Sud, centre d’Orsay, 426 pp
Meddeb N (1993) Potentialités géothermique de la Tunisie septentrionale. Thèse de 3ième cycle, Univ. Tunis II
Noda T, Shimada K (1993) Water mixing model calculation for evaluation of deep geothermal water. Geothermics 22(3):165–180
Pang ZH, Reed M (1998) Theoretical chemical thermometry on geothermal waters: Problems and methods. Geochim Cosmochim Acta 62:1083–1091
Piper AM (1944) A graphic procedure in the geochemical interpretation of water analysis. Amer Geophys Union Trans 25:914–923
Sadki O (1991) Géochimie des sources thermales de la Tunisie septentrionale: Relation hydrothermalisme - minéralisations. D.E.A. 97 p. F S T, Univ, Tunis II, Tunisie
Sadki O (1998) Etude des systèmes hydrothermaux du Nord de la Tunisie : Géochimie des interactions eaux- roches et circulation hydrothermale. Thèse de Doctorat 246 p. Faculté des Sciences de Tunis, Univ, Tunis II, Tunisie
Simler R (2007) D I A G R A M M E S : Logiciel d’hydrochimie multilangage en distribution libre. Version 4. Laboratoire d’Hydrogéologie d’Avignon
Soussi M (2002) Le Jurassique de la Tunisie atlasique, stratigraphie, dynamique sédimentaire, paléogéographie, et intérêt pétrolier. Documents n°157–2002, stu, Lyon, 363 pp
Tonani F (1980) Some remarks on the application of geochemical techniques exploration. In: Proc. of the adv. Eur. geoth. res., 2nd symp., Strasbourg, pp 428–443
Truesdell AH, Fournier RO (1977) Procedure for estimating the temperature of a hot water, component in a mixed water using a plot of dissolved silica vs enthalpy. J Res US Geological Survey 5(1):49–52
Turki MM (1985) Polycin6matique et contrô1e sédimentaire associé sur la cicatrice Zaghouan-Nebhana. Thèse de Doctorat en sciences, Univ. Tunis II
Turki MM, Soussi M (2002) Etude des réservoirs jurassiques et des structures géologiques de la région Zaghouan-Zriba-Medeker. Rapport final, Projet, ENIS-Office du thermalisme
Verma SP, Santoyo E (1997) New improved equations Na/K, Na/Li and SiO2 geothermamometers, by outlier detection and rejection. J Volcanol Geotherm Res 79:9–23
Zouiten S (1999) Application de la géothermométrie aux eaux des sources thermales du Nord de la Tunisie. Thèse de Doctorat 221 p. Faculté des Sciences de Tunis, Université Tunis II, Tunisie
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trabelsi, S., Makni, J., Bouri, S. et al. Hydrochemistry of thermal waters in Northeast Tunisia: water–rock interactions and hydrologic mixing. Arab J Geosci 8, 1743–1754 (2015). https://doi.org/10.1007/s12517-014-1293-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12517-014-1293-2