Abstract
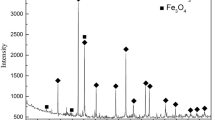
The reduction of ilmenite concentrate by hydrogen gas was investigated in the temperature range of 500 to 1200°C. The microstructure and phase transition of the reduction products were studied by X-ray diffraction (XRD), scanning electron microscopy (SEM), and optical microscopy (OM). It was found that the weight loss and iron metallization rate increased with the increase of reduction temperature and reaction time. The iron metallization rate could reach 87.5% when the sample was reduced at 1150°C for 80 min. The final phase constituents mainly consist of Fe, M3O5 solid solution phase (M=Mg, Ti, and Fe), and few titanium oxide. Microstructure analysis shows that the surfaces of the reduction products have many holes and cracks and the reactions take place from the exterior of the grain to its interior. The kinetics of reduction indicates that the rate-controlling step is diffusion process control with the activation energy of 89 kJ·mol−1.
Similar content being viewed by others
References
L. Zhang, L.N. Zhang, M.Y. Wang, G.Q. Li, and Z.T. Sui, Precipitation selectivity of perovskite phase from Ti-bearing blast furnace slag under dynamic oxidation conditions, J. Non Cryst. Solids, 353(2007), No.22–23, p.2214.
C.S. Kucukkaragoz and R.H. Eric, Solid state reduction of a natural ilmenite, Miner. Eng., 19(2006), No.3, p.334.
Y.M. Wang and Z.F. Yuan, Reductive kinetics of the reaction between a natural ilmenite and carbon, Int. J. Miner. Process., 81(2006), No.3, p.133.
R. Merk and C.A. Pickles, Reduction of ilmenite by carbon monoxide, Can. Metall. Q., 27(1988), No.3, p.179.
M. Pouraboli, S. Raygan, H. Abdizadeh, and K. Hanaei, Production of high titania slag by electro-slag crucible melting (ESCM) process, Int. J. Miner. Process., 78(2006), No.3, p.175.
M. Ma, D.H. Wang, W.G. Wang, X.H. Hu, X.B. Jin, and G.Z. Chen, Extraction of titanium from different titania precursors by the FFC Cambridge process, J. Alloys Compd., 420(2006), No.1–2, p.37.
G.Z. Chen, D.J. Fray, and T.W. Farthing, Direct electrochemical reduction of titanium dioxide to titanium in molten calcium chloride, Nature, 407(2000), No.6802, p.361.
U.B. Pal and A.C. Powell IV, The use of solid-oxide-membrane technology for electrometallurgy, JOM, 59(2007), No.5, p.44.
X.S. Ye, X.G. Lu, C.H. Li, W.Z. Ding, X.L. Zou, Y.H. Gao, and Q.D. Zhong, Preparation of Ti-Fe based hydrogen storage alloy by SOM method, Int. J. Hydrogen Energy, 36(2011), No.7, p.4573.
O.J.P. Birat, Addressing the climate change challenge: ULCOS breakthrough program, CAMP-ISIJ, 22(2009), No.1, p.258.
T. Usui, H.O. Nakazato, H. Konishi, and H. Kawabata, Effect of hydrogen on reduction of iron ore agglomerates with H2-CO mixture, CAMP-ISIJ, 22(2009), No.1, p.294.
J.R. Hino, S. Kubo, K. Onuki, Y. Tachibana, and M. Ogawa, Contribution of nuclear hydrogen to ironmaking, CAMP-ISIJ, 22(2009), No.1, p.290.
P.C. Pistorius and T. Motlhamme, Oxidation of high-titanium slags in the presence of water vapour, Miner. Eng., 19(2006), No.3, p.232.
J. Pesl and R.H. Eric, High temperature carbothermic reduction of Fe2O3-TiO2-MxOy oxide mixtures, Miner. Eng., 15(2002), No.11, p.971.
D. Bessinger, J.M.A. Geldenhuis, P.C. Pistorius, A. Mulaba, and G. Hearne, The decrepitation of solidified high titania slags, J. Non Cryst. Solids, 282(2001), No.1, p.132.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was financially supported by the Postgraduate Innovative Foundation of Shanghai University (SHUCX091031), the National Natural Science Foundation of China (No.51074105), and the National Basic Research Priorities Program of China (No.2007CB613606).
Rights and permissions
About this article
Cite this article
Si, Xg., Lu, Xg., Li, Cw. et al. Phase transformation and reduction kinetics during the hydrogen reduction of ilmenite concentrate. Int J Miner Metall Mater 19, 384–390 (2012). https://doi.org/10.1007/s12613-012-0568-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-012-0568-4